Author: Brit Long, MD (@long_brit, EM Attending Physician, San Antonio, TX) and Michael Gottlieb, MD (EM Attending Physician, Rush Medical Center) // Edited by: Alex Koyfman, MD (@EMHighAK, EM Attending Physician, UTSW / Parkland Memorial Hospital)
Case
You are caring for a 28-year-old male transferred to your trauma center with severe abdominal and pelvic trauma after an MVC. The patient was found to have a grade IV hepatic laceration and open book pelvic fracture. He was given 3 units of pRBCs and 3 units of FFP before transfer, which stabilized him for transfer. His current vital signs are HR 112 bpm, BP 108/58 mm Hg, RR 26, oxygen saturation 90% on room air, and oral temperature 96F. On your exam, the patient is in distress, and his abdomen is significantly distended and tense to palpation.
What is abdominal compartment syndrome?
Abdominal compartment syndrome (ACS) is defined by sustained intra-abdominal pressure (IAP) > 20 mm Hg with associated organ injury. The condition was first described in 1863, but not significantly discussed until the 1990s (1-3). Intra-abdominal hypertension (IAH), or elevated pressures within the abdomen in the absence of end organ injury, has been associated with increased mortality and lengths of stay, but just because the patient has IAH does not mean he/she has ACS. Approximately 3-13% of patients will develop ACS in the setting of IAH (4-7).
ACS possesses a large number of associated risk factors, but all of these are related to 4 components: diminished abdominal wall compliance, increased intraluminal contents, increased abdominal contents, or capillary leak/fluid resuscitation (7-9).

Unfortunately, ACS is associated with a mortality ranging from 25-100% (1-5, 7). However, it is often missed in the ED. While one of our primary roles is to resuscitate and manage acute life-threatening conditions, we are finding ourselves boarding critical patients in the ED. With this increased boarding, emergency clinicians must recognize, diagnose, and manage ACS.
What is the underlying pathophysiology?
The abdomen is a compartment defined by the abdominal wall anteriorly and posteriorly, the diaphragm superiorly, and the pelvic floor inferiorly. A normal intra-abdominal pressure ranges from 2-5 mm Hg, which can increase to 7 mm Hg in critically ill patients (8-10). However, other conditions can also increase this baseline level, such as obesity (7-16 mm Hg at baseline) and pregnancy (up to 14 mm Hg) (11-13).
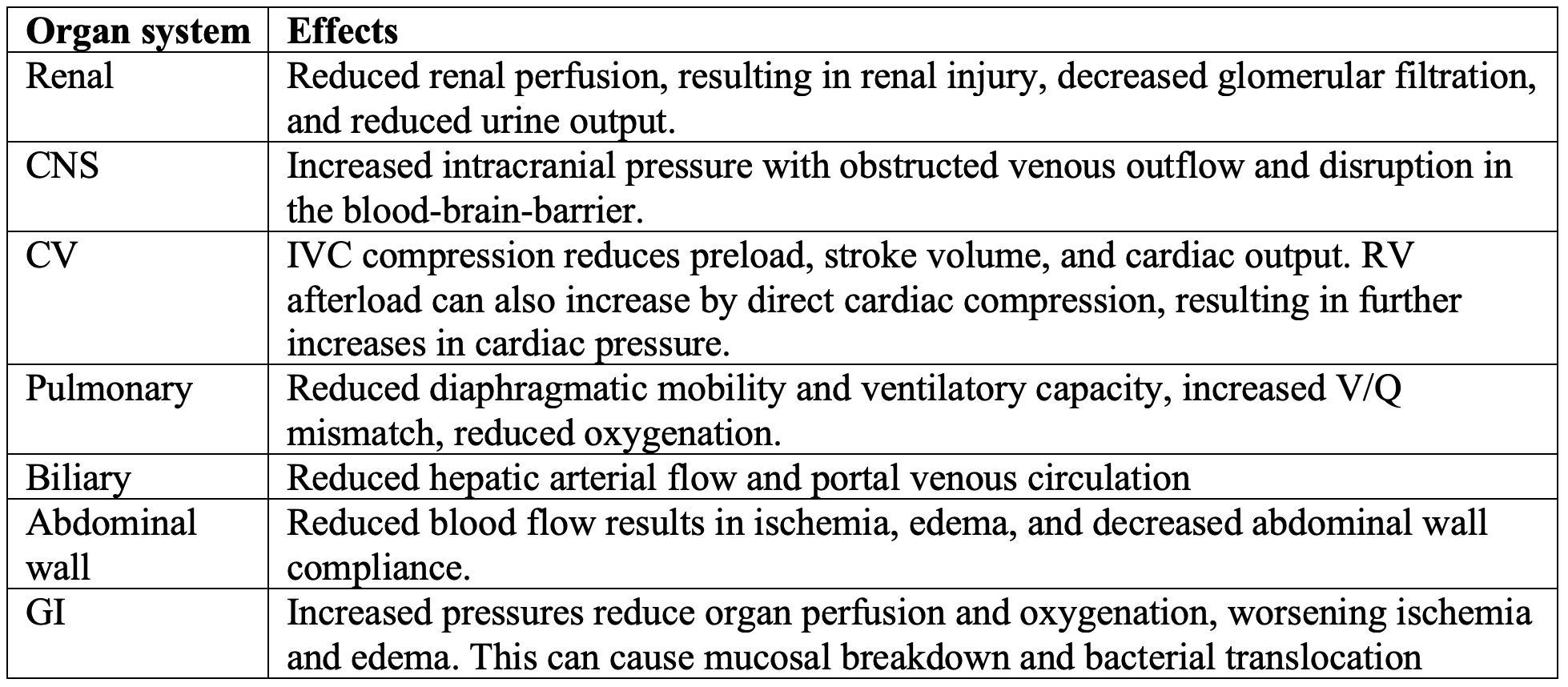
ACS is just like any other form of compartment syndrome, with decreased tissue perfusion and oxygenation resulting from elevated pressure within a fixed space (8,14). ACS increases bowel edema and intestinal ischemia, allowing for bacterial translocation. However, ACS does not just affect the abdominal organs (1,3,4,15-18). Other systems affected include the renal, CNS, cardiovascular, pulmonary, and biliary systems.
There are three categories of ACS, based on the cause and duration (1,2).
- Primary: Intrinsic etiology resulting from an intra-abdominal injury or disease
- Secondary: Extrinsic etiology that does not come from the abdominopelvic region (sepsis, fluid resuscitation, burns)
- Recurrent: ACS redevelops following a prior episode of either primary or secondary ACS.
What can you find on history and exam?
You cannot diagnose what you do not consider. Most patients with ACS will be critically ill, with many intubated or with poor mental status. Thus, keep this condition in mind in patients who are critically ill with the risk factors from Table 1. Other risks factors include prior abdominal surgeries or known intra-abdominal adhesions (1,2,19), which reduce abdomen’s ability to compensate for increased pressures. Patients who are awake may have worsening pain, distension, and difficulty breathing (10,20). Orthopnea may be present (10,20). Worsening or refractory hypotension with new organ failure are major clues to ACS. Oliguria and renal dysfunction are two of the earliest findings in ACS (21,22).
Exam in ACS possesses major limitations and cannot exclude the diagnosis with a sensitivity of 40-60%, though specificity for several findings range from 80% to 94% (16,17). Increased abdominal distension, elevated abdominal wall tension, and absence of bowel soundsare associated with diagnosis (23,24). While it seems measuring the amount of abdominal distension would work, literature suggests this is not a reliable predictor (23-25). Diffuse edema, increasing abdominal tension with palpation, and frequent high-pressure alarms on a ventilated patient suggest the disease (10,20).
What role do labs and imaging play?
Labs play an important role in ACS, as organ dysfunction is a key part of the diagnosis. CBC, renal and liver function, VBG (pH and lactate), electrolytes, and urinalysis are recommended. Severe organ dysfunction may be represented by increased creatinine, troponin, liver function tests, or lactate, as well as reduced glomerular filtration rate (1,2,24,26,27). Metabolic acidosis is present in severe ACS with elevated lactate. D-lactate is a produce of intestinal bacterial metabolism and may be an early detector of intestinal ischemia, but this test may not be obtainable. Intestinal fatty acid binding protein (I-FABP) and interleukins have also been evaluated but are not ready for primetime (20,28-30).
Just like your exam, imaging may demonstrate findings associated with ACS, but normal imaging cannot exclude the diagnosis. Abdominal x-ray may reveal free air or intestinal dilation, and chest x-ray may show pleural effusions, diaphragm elevation, and atelectasis (1,2,31-33). CT of the abdomen/pelvis can reveal many findings suggesting ACS: increased intestinal AP diameter, IVC collapse, diaphragm elevation, pneumoperitoneum, renal vessel compression, thickened bowel wall, and inguinal hernias bilaterally (32-34). Narrowed vasculature and organ compression on CT strongly suggest ACS. There are several other findings on CT cited in the literature concerning ACS: the round belly sign (RBS) is an increased ratio of anteroposterior to transverse diameter of more than 0.80, and the peritoneal-to-abdominal height ratio (PAR) is the ratio of the anteroposterior peritoneal compartment diameter (distance from linea alba to posterior part of duodenum) and anteroposterior abdomen diameter (distance from linea alba to posterior fascia) measured along the midline. A PAR > 0.52 suggests IAH, while values < 0.45 possess a 100% negative predictive value (1,2,32-36). Other values of CT include evaluating for the underlying etiology of ACS.

What about intra-abdominal pressure? When should you measure IAP, and how is it measured?
While history, exam, labs, and imaging have roles in the diagnosis of ACS, measuring IAP is imperative. Measure IAP in any critically ill patient with a risk factor for ACS and new/worsening organ failure (1,2,10,20). You can measure IAP through direct or indirect measurement. Direct measurement includes placing a catheter into the peritoneal cavity, while indirect measurements are obtained through the bladder, rectum, stomach, or vagina. In the ED, bladder pressures are the easiest and most reliable means of assessing IAP (1,3,4,7,37-39). However, obtaining bladder pressures is contraindicated in those with traumatic bladder injury, pelvic packing, or history of cystectomy (37).
Intermittent IAP assessment every 4-6 hours is recommended by the World Society of the Abdominal Compartment Syndrome (37,40). Assessing more frequently than 4 hours is not recommended. In the ED, a single measurement in a patient with risk factors is recommended (10,20,37). However, if the patient remains in the ED for longer than 4 hours, you may need to obtain a repeat measurement. There are means of continuous measurement, but data suggest that continuous and intermittent assessments are equally reliable. Plus, continuous assessment needs a 3-way catheter for measurement (3,7,37).
To measure bladder pressures, you need the following: Foley catheter kit, Kelly forceps, 60-mL syringe with sterile saline, 3-way stopcock, arterial line pressure transducing tubing, 1 Liter of intravenous fluids, and a pressure bag (1,3,37-43).

Make sure the patient is placed supine and relaxed. Coughing and inadequate sedation can increase bladder pressure. While most patients with ACS will be mechanically ventilated, you can measure IAP in awake, spontaneously breathing patients, but just ensure these patients are provided appropriate analgesia. Obtain the pressure at end-expiration with an appropriately placed Foley catheter (1,2,27,37,39)). Zero the pressure transducer at the level of the bladder in the mid-axillary line. Be sure to install a maximum of 25 mL of warm saline (cold saline will increase the pressures) (3,37,43,44). Once you have placed 25 mL into the bladder, wait 30 seconds to allow the detrusor muscle to relax. Another measurement technique includes urine drainage manometry with 50 mL of sterile saline and the Foley catheter tubing as the device for measurement. The catheter tube is placed perpendicular at the pubic symphysis level, with a ruler to measure the fluid height. Use a ruler to obtain pressure readings, which conversion of cm H20 into mm Hg by multiplying by 0.74 (45-49).
What are pitfalls in IAP measurement?
Obesity, pelvic pathology, and radiation cystitis can elevate IAP (1,2,20,31,37).Falsely low readings may occur with a leak in the transducer system. IAH is defined by values > 12 mm Hg. Keep in mind there are several grades of IAH and ACS (1,2,20,31,37,48,49).

Remember, diagnosis includes IAP > 20 mm Hg with organ dysfunction. However, patients at risk for ACS are critically ill and may have several contributors to organ failure (7,9,37-39). This is one of those classic “chicken or the egg” situations, as some patients may have organ dysfunction from the primary illness or injury (1,2,9,20,37). Timing of injury can assist. ACS is likely when IAP > 20 mm Hg with organ dysfunction that occurs in a time-dependent association (1,2,9,20,37). Patients with oliguria and IAP > 20 mm Hg likely have ACS as well (21,22,50).
Ok, you have diagnosed ACS. What’s the management?
Management targets improving end-organ perfusion and reducing elevated IAH. The WSACS management algorithm includes evacuating intraluminal contents and intraabdominal space-occupying lesions, improving compliance of the abdominal wall, optimizing intravascular fluid therapy, and increasing systemic perfusion (20,37,41).

Abdominal perfusion pressure (APP) is calculated by mean arterial pressure (MAP) minus IAP (37,51,52). APP is an assessment of the intra-abdominal organs and is a better predictor of survival than IAP or MAP alone (37,51,52). While a goal APP > 60 mm Hg is generally recommended, there is no evidence-based goal for a specific APP (9,22,37). Optimize APP first by ensuring adequate intravascular volume, but avoid hypervolemia (41,53). Fluid administration should be minimal, as third spacing increases IAH (1-3,37,54,55). In hypotensive patients, early vasopressor and inotropic support may be needed (1-3,9,37,53-55). Norepinephrine and dobutamine possess the greatest literature support for use in ACS (56-59).
Treat pain and agitation, as these increase IAP (1,2,9,37,41). Though many will be intubated, mechanical ventilation should be avoided if possible, as this converts negative-pressure system to positive pressure, increasing IAP (41,60). In the mechanically ventilated patient, target as low PEEP and plateau pressures as oxygen saturation allows (9,37,41,60). Paralysis can also be used while awaiting adequate decompression (61-63). However, prolonged paralysis does not appear to result in persistent reduction in IAP (61-63).
Abdominal compartment decompression is essential, with reducing intraluminal and intra-abdominal contents. Ileus and luminal distension are common due to intestinal ischemia (10,20,37,41). Metoclopramide and erythromycin (prokinetic agents) can reduce intraluminal contents. NG or OG tube placement can also reduce gastric contents and pressure, and with stimulant laxatives, neostigmine, rectal tube drainage, and/or colonoscopy can reduce colonic distension (37,41). Ascites, blood, abscess, and free air can increase IAP, and if any of these are present, they should be removed (10,20,37,41). Therapeutic paracentesis is recommended if ascites is present, and percutaneous drainage may be used to remove extraluminal air (64-66). Thoracic decompression is recommended for pleural effusion or pneumothorax, and escharotomy is recommended for patients with severe burns that are causing increased IAP (37,41).
Patients may fail to respond to the above measures, necessitating surgical decompression via laparotomy. This is the definitive intervention, reducing IAP and improving patient hemodynamic status and end-organ prefusion (37,41,67-69). Surgical decompression improves patient mortality, and the earlier the intervention, the better (improved survival and reduced morbidity with earlier decompression) (70).
Disposition and Prognosis
Most patients with ACS require ICU level care (1,2,9,37). Just keep in mind repeat assessments in patients who are boarding may be needed. Prognosis primarily depends on rapid diagnosis and early management. Patients with older age, comorbidities, greater volume received in resuscitation, and major organ dysfunction have higher mortality rates (10,20,37,71-73).
Key Points
- ACS is a potentially deadly condition caused by increased pressure within the abdominal compartment.
- ACS is defined by IAP > 20 mm Hg with organ dysfunction.
- Four underlying causes contribute: decreased abdominal wall compliance, increased intraluminal contents, increased abdominal contents, or capillary leak/fluid resuscitation.
- History and physical examination may suggest the diagnosis, but they should not be used to exclude ACS. Labs and imaging can also assist in diagnosis.
- The gold standard for diagnosis is intra-abdominal pressure measurement, typically through a Foley catheter.
- Management includes increasing abdominal wall compliance, evacuating gastrointestinal contents, avoiding excessive fluid resuscitation, draining intraperitoneal contents, and decompressive laparotomy in select cases. Patients typically require admission to a critical care unit.
References/Further Reading
- Papavramidis TS, Marinis AD, Pliakos I, et al. Abdominal compartment syndrome – Intra-abdominal hypertension: Defining, diagnosing, and managing. J Emerg Trauma Shock. 2011 Apr;4(2):279-91.
- Sosa G, Gandham N, Landeras V, Calimag AP, Lerma E. Abdominal compartment syndrome. Dis Mon. 2019 Jan;65(1):5-19.
- Cheatham ML. Abdominal compartment syndrome: pathophysiology and definitions. Scand J Trauma Resusc Emerg Med. 2009 Mar 2;17:10.
- Vidal MG, Ruiz Weisser J, Gonzalez F, et al. Incidence and clinical effects of intra-abdominal hypertension in critically ill patients. Crit Care Med. 2008 Jun;36(6):1823-31.
- Murphy PB, Parry NG, Sela N, Leslie K, Vogt K, Ball I. Intra-Abdominal Hypertension Is More Common Than Previously Thought: A Prospective Study in a Mixed Medical-Surgical ICU. Crit Care Med. 2018 Jun;46(6):958-964.
- Malbrain ML, Chiumello D, Pelosi P, et al. Incidence and prognosis of intraabdominal hypertension in a mixed population of critically ill patients: a multiple-center epidemiological study. Crit Care Med. 2005 Feb;33(2):315-22.
- Maluso P, Olson J, Sarani B. Abdominal Compartment Hypertension and Abdominal Compartment Syndrome. Crit Care Clin. 2016 Apr;32(2):213-22.
- Sugrue M, De Waele JJ, De Keulenaer BL, Roberts DJ, Malbrain ML. A user’s guide to intra-abdominal pressure measurement. Anaesthesiol Intensive Ther. 2015;47(3):241-51.
- Malbrain ML, De Laet IE, De Waele JJ, Kirkpatrick AW. Intra-abdominal hypertension: definitions, monitoring, interpretation and management. Best Pract Res Clin Anaesthesiol. 2013 Jun;27(2):249-70.
- Carr JA. Abdominal compartment syndrome: a decade of progress. J Am Coll Surg. 2013 Jan;216(1):135-46.
- De Keulenaer BL, De Waele JJ, Powell B, Malbrain ML. What is normal intra-abdominal pressure and how is it affected by positioning, body mass and positive end-expiratory pressure? Intensive Care Med. 2009 Jun;35(6):969-76.
- Fuchs F, Bruyere M, Senat MV, et al. Are standard intra-abdominal pressure values different during pregnancy? PLoS One. 2013 Oct 25;8(10):e77324.
- Staelens AS, Van Cauwelaert S, Tomsin K, Mesens T, Malbrain ML, Gyselaers W. Intra-abdominal pressure measurements in term pregnancy and postpartum: an observational study. PLoS One. 2014 Aug 12;9(8):e104782.
- Long B, Koyfman A, Gottlieb M. Evaluation and Management of Acute Compartment Syndrome in the Emergency Department. J Emerg Med. 2019 Apr;56(4):386-397.
- Malbrain ML, Chiumello D, Cesana BM, et al; WAKE-Up! A systematic review and individual patient data meta-analysis on intra-abdominal hypertension in critically ill patients: the wake-up project. World initiative on Abdominal Hypertension Epidemiology, a Unifying Project (WAKE-Up!). Minerva Anestesiol. 2014 Mar;80(3):293-306.
- Kirkpatrick AW, Brenneman FD, McLean RF, Rapanos T, Boulanger BR. Is clinical examination an accurate indicator of raised intra-abdominal pressure in critically injured patients? Can J Surg. 2000 Jun;43(3):207-11.
- Sugrue M, Bauman A, Jones F, et al. Clinical examination is an inaccurate predictor of intraabdominal pressure. World J Surg. 2002 Dec;26(12):1428-31.
- Diebel LN, Dulchavsky SA, Wilson RF. Effect of increased intra-abdominal pressure on mesenteric arterial and intestinal mucosal blood flow. J Trauma. 1992 Jul;33(1):45-8.
- Popescu GA, Bara T, Rad P. Abdominal Compartment Syndrome as a Multidisciplinary Challenge. A Literature Review. J Crit Care Med (Targu Mures). 2018 Oct 1;4(4):114-119.
- Hecker A, Hecker B, Hecker M, Riedel JG, Weigand MA, Padberg W. Acute abdominal compartment syndrome: current diagnostic and therapeutic options. Langenbecks Arch Surg. 2016 Feb;401(1):15-24.
- Bozdağ E, Gündeş E, Aday U, et al. Postrenal acute kidney injury and abdominal compartment syndrome associated with bladder pressure: type III rectus sheath hematoma. Arch Med Sci Atheroscler Dis. 2017 Mar 13;2(1):e1-e2.
- Mohmand H, Goldfarb S. Renal dysfunction associated with intra-abdominal hypertension and the abdominal compartment syndrome. J Am Soc Nephrol. 2011 Apr;22(4):615-21.
- Reintam Blaser A, Regli A, De Keulenaer B, et al; Incidence, Risk Factors, and Outcomes of Intra-Abdominal (IROI) Study Investigators. Incidence, Risk Factors, and Outcomes of Intra-Abdominal Hypertension in Critically Ill Patients-A Prospective Multicenter Study (IROI Study). Crit Care Med. 2019 Apr;47(4):535-542.
- Iyer D, Rastogi P, Åneman A, D’Amours S. Early screening to identify patients at risk of developing intra-abdominal hypertension and abdominal compartment syndrome. Acta Anaesthesiol Scand. 2014 Nov;58(10):1267-75.
- Malbrain ML, De laet I, Van Regenmortel N, Schoonheydt K, Dits H. Can the abdominal perimeter be used as an accurate estimation of intra-abdominal pressure? Crit Care Med. 2009 Jan;37(1):316-9.
- An G, West MA: Abdominal compartment syndrome: a concise clinical review. Crit Care Med 2008, 36(4):1304–1310.
- Balogh Z, De Waele JJ, Kirkpatrick A, et al; World Society of the Abdominal Compartment Syndrome. Intra-abdominal pressure measurement and abdominal compartment syndrome: the opinion of the World Society of the Abdominal Compartment Syndrome. Crit Care Med. 2007 Feb;35(2):677-8; author reply 678-9.
- Nielsen C, Kirkegård J, Erlandsen EJ, et al. D-lactate is a valid biomarker of intestinal ischemia induced by abdominal compartment syndrome. J Surg Res. 2015 Apr;194(2):400-4.
- Duzgun AP, Gulgez B, Ozmutlu A, et al. The relationship between intestinal hypoperfusion and serum d-lactate levels during experimental intra-abdominal hypertension. Dig Dis Sci 2006;51:2400.
- Strang SG, Van Waes OJF, Van der Hoven B et al. Intestinal fatty acid binding protein as a marker for intra-abdominal pressurerelated complications in patients admitted to the intensive care unit; study protocol for a prospective cohort study (I-Fabulous study). Scand J Trauma Resusc Emerg Med 2015;23:6.
- Blaser AR, Björck M, De Keulenaer B, Regli A. Abdominal compliance: A bench-to-bedside review. J Trauma Acute Care Surg. 2015 May;78(5):1044-53.
- Sugrue G, Malbrain MLNG, Pereira B, et al. Modern imaging techniques in intra-abdominal hypertension and abdominal compartment syndrome: a bench to bedside overview. Anaesthesiol Intensive Ther. 2018;50(3):234-242.
- Patel A, Lall CG, Jennings SG, Sandrasegaran K. Abdominal compartment syndrome. AJR Am J Roentgenol. 2007 Nov;189(5):1037-43.
- Bouveresse S, Piton G, Badet N, Besch G, Pili-Floury S, Delabrousse E. Abdominal compartment syndrome and intra-abdominal hypertension in critically ill patients: diagnostic value of computed tomography. Eur Radiol. 2019 Feb 8. doi: 10.1007/s00330-018-5994-x. [Epub ahead of print]
- Al-Bahrani AZ, Abid GH, Sahgal E, et al. A prospective evaluation of CT features predictive of intra-abdominal hypertension and abdominal compartment syndrome in critically ill surgical patients. Clin Radiol. 2007; 62(7): 676–682.
- Pickhardt PJ, Shimony JS, Heiken JP, et al. The abdominal compartment syndrome: CT findings. AJR Am J Roentgenol. 1999; 173(3): 575–579.
- Kirkpatrick AW, Roberts DJ, De Waele J, et al. Intra-abdominal hypertension and the abdominal compartment syndrome: updated consensus definitions and clinical practice guidelines from the World Society of the Abdominal Compartment Syndrome. Intensive Care Med. 2013;39:1190–1206.
- Cheatham ML, Safcsak K. Intra-abdominal hypertension and abdominal compartment syndrome: the journey forward. Am Surg. 2011 Jul;77 Suppl 1:S1-5.
- Milanesi R, Caregnato RC. Intra-abdominal pressure: an integrative review. Einstein (Sao Paulo). 2016;14(3):423–430.
- Starkopf J, Tamme K, Blaser AR: Should we measure Intra-abdominal pressures in every intensive care patient? Ann Intensive Care 2012, 2 (Suppl 1): S9.
- Hunt L, Frost SA, Hillman K, et al. Management of intra-abdominal hypertension and abdominal compartment syndrome: a review. J Trauma Manag Outcomes. 2014;8(1):2.
- Cheatham ML, Malbrain ML, Kirkpatrick A, et al. Results from the International Conference of Experts on intra-abdominal hypertension and abdominal compartment syndrome. II. Recommendations. Intensive Care Med 2007;33:951–62.
- Ali M. Abdominal compartment syndrome: the importance of urinary catheter placement in measuring intra-abdominal pressure. BMJ Case Rep. 2018 Oct 21;2018. pii: bcr-2018-226786
- Soler Morejón Cde D, Lombardo TA, Tamargo Barbeito TO, Sandra BG. Effects of zero reference position on bladder pressure measurements: an observational study. Ann Intensive Care. 2012 Jul 5;2 Suppl 1:S13.
- Harrahill M. Intra-abdominal pressure monitoring. J Emerg Nurs 1998;24:465–466.
- Sugrue M, Balogh Z, Malbrain M. Intra-abdominal hypertension and renal failure. ANZ J Surg 2004;74:78.
- Otto J, Binnebösel M, Junge K, et al. Harrahill’s technique: a simple screening test for intra-abdominal pressure measurement. Hernia. 2010 Aug;14(4):415-9.
- Al-Abassi AA, A Sadi AS, Ahmed F. Is intra-bladder pressure measurement a reliable indicator for raised intra-abdominal pressure? A prospective comparative study. BMC Anesthesiology. 2018l18:69,
- Malbrain MLNG, Cheatham ML, Kirkpatrick A, et al. Results from the international conference of experts on intra-abdominal hypertension and abdominal compartment syndrome. I. definitions. Intensive Care Med. 2006;32(11):1722–1732.
- Patel DM, Connor MJ Jr. Intra-Abdominal Hypertension and Abdominal Compartment Syndrome: An Underappreciated Cause of Acute Kidney Injury. Adv Chronic Kidney Dis. 2016 May;23(3):160-6.
- De Waele JJ, De Laet I, Kirkpatrick AW, Hoste E. Intra-abdominal Hypertension and Abdominal Compartment Syndrome. Am J Kidney Dis. 2011 Jan;57(1):159-69.
- Cheatham ML, White MW, Sagraves SG, Johnson JL, Block EF. Abdominal perfusion pressure: a superior parameter in the assessment of intra-abdominal hypertension. J Trauma. 2000 Oct;49(4):621-6; discussion 626-7.
- Regli A, De Keulenaer B, De Laet I, et al. Fluid therapy and perfusional considerations during resuscitation in critically ill patients with intra-abdominal hypertension. Anaesthesiol Intensive Ther. 2015;47(1):45-53.
- Joseph B, Zangbar B, Pandit V, et al. The conjoint effect of reduced crystalloid administration and decreased damage-control laparotomy use in the development of abdominal compartment syndrome. J Trauma Acute Care Surg. 2014 Feb;76(2):457-61.
- Kula R, Szturz P, Sklienka P, Neiser J, Jahoda J. A role for negative fluid balance in septic patients with abdominal compartment syndrome? Intensive Care Med. 2004 Nov;30(11):2138-9.
- Zhang H, Smail N, Cabral A, Rogiers P, Vincent JL.Effects of norepinephrine on regional blood flow and oxygen extraction capabilities during endotoxic shock. Am J Respir Crit Care Med. 1997 Jun;155(6):1965-71.
- Peng ZY, Critchley LA, Joynt GM, Gruber PC, Jenkins CR, Ho AM. Effects of norepinephrine during intra-abdominal hypertension on renal blood flow in bacteremic dogs. Crit Care Med. 2008 Mar;36(3):834-41.
- Krejci V, Hiltebrand LB, Sigurdsson GH. Effects of epinephrine, norepinephrine, and phenylephrine on microcirculatory blood flow in the gastrointestinal tract in sepsis. Crit Care Med. 2006 May;34(5):1456-63.
- Agustí M, Elizalde JI, Adàlia R, Cifuentes A, Fontanals J, Taurà P. Dobutamine restores intestinal mucosal blood flow in a porcine model of intra-abdominal hyperpressure. Crit Care Med. 2000 Feb;28(2):467-72.
- Verzilli D, Constantin J-M, Sebbane M, et al. Positive end expiratory pressure affects the value of intra-abdominal pressure in acute lung injury/acute respiratory distress syndrome patients: a pilot study. Crit Care. 2010;14:R137.
- Deeren DH, Dits H, Malbrain ML. Correlation between intraabdominal and intracranial pressure in nontraumatic brain injury. Intensive Care Med. 2005;31:1577–1581.
- De Laet I, Hoste E, Verholen E, De Waele JJ. The effect of neuromuscular blockers in patients with intra-abdominal hypertension. Intensive Care Med. 2007 Oct;33(10):1811-4.
- De Waele JJ, Benoit D, Hoste E, Colardyn F. A role for muscle relaxation in patients with abdominal compartment syndrome? Intensive Care Med. 2003 Feb;29(2):332.
- Corcos AC, Sherman HF. Percutaneous treatment of secondary abdominal compartment syndrome. J Trauma 2001;51:1062–4.
- Savino JA, Cerabona T, Agarwal N, et al. Manipulation of ascitic fluid pressure in cirrhotics to optimize hemodynamic and renal function. Ann Surg 1988;208:504–11.
- Liang YJ, Huang HM, Yang HL, et al. Controlled peritoneal drainage improves survival in children with abdominal compartment syndrome. Ital J Pediatr. 2015 Apr 8;41:29.
- Van Damme L, De Waele JJ. Effect of decompressive laparotomy on organ function in patients with abdominal compartment syndrome: a systematic review and meta-analysis. Crit Care. 2018 Jul 25;22(1):179.
- Muresan M, Muresan S, Brinzaniuc K, et al. How much does decompressive laparotomy reduce the mortality rate in primary abdominal compartment syndrome: A single-center prospective study on 66 patients. Medicine (Baltimore). 2017;96(5):e6006.
- Strang SG, Van Lieshout EMM, Verhoeven RA, et al. Recognition and management of intra‑abdominal hypertension and abdominal compartment syndrome; a survey among Dutch surgeons. Eur J Trauma Emerg Surg. 2017;43:85–98.
- Mentula P, Hienonen P, Kemppainen E, et al. Surgical decompression for abdominal compartment syndrome in severe acute pancreatitis. Arch Surg. 2010 Aug;145(8):764-9.
- García P, Santa-Teresa P, Muñoz J, et al. Incidence and prognosis of intra-abdominal hypertension in critically ill medical patients: a prospective epidemiological study. Ann Intensive Care. 2012, 2(1):S3.
- Aik-Yong C, Ye-Xin K, Yi NS, Hway WT. Abdominal compartment syndrome: Incidence and prognostic factors influencing survival in Singapore. Indian J Crit Care Med. 2014;18(10):648–652.
- Balogh Z, McKinley BA, Cox CS, et al. Abdominal compartment syndrome: The cause or effect of postinjury multiple organ failure. Shock. 2003;20:483–92.






