Authors: Kristina Eastman, MD (EM Resident Physician, Aventura Hospital); Eva Ryder, MD (EM Resident Physician, Aventura Hospital); Scarlet Benson, MD (EM Attending Physician, Aventura Hospital) // Reviewed by: Andrew Grock, MD (Faculty Physician, Division of Emergency Medicine, Greater Los Angeles VA Healthcare System and Assistant Clinical Professor of Emergency Medicine David Geffen School of Medicine, UCLA); Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit)
Clinical Cases
#1: A 66-year-old male with no past medical history presents with pain, swelling, and redness to the left gluteal region for seven days. He reports progressively worsening symptoms since shaving in that area a few days prior. The patient denies fever, chills, or malaise. In the ED, his vitals were HR 86, BP 143/81, RR 17, T 98.2. The patient’s exam was benign except for his rectal and buttock exam. To his left gluteal cleft, adjacent to the anus, he had extensive induration with surrounding erythema and multiple open pustules without fluctuance, active draining, or discharge. Additionally, he had marked tenderness to palpation on digital rectal exam (DRE). Labs were all within normal limits. A computer tomography (CT) of the abdomen and pelvis showed a peripherally enhancing left ischioanal fossa abscess measuring 2.2 x 2.9 x 3.6 cm. Surgery was consulted and performed surgical drainage in the operating room (OR).
#2: A 30-year-old male with no past medical history presented with four days of progressively worsening pain and redness to the right gluteal region. The patient reports several episodes of painful bowel movements but denies fever, chills, or malaise. In the ED, the patient’s vitals are HR 102, BP 137/75, RR 18, T 98.7. Exam reveals a right perianal 4×6 cm area of fluctuance with surrounding erythema but no induration, active draining, or discharge. Labs within normal limits except for an elevated WBC (12.8 103/uL). Computed tomography (CT) of the abdomen and pelvis shows a superficial 2.8 x 1.8 x 2.0 cm abscess posterior and to the right of the anus. After appropriate anesthesia with subcutaneous lidocaine, an incision and drainage (I&D) is performed with a cruciate incision over the fluctuant part of the abscess. Afterwards, the patient is discharged home with Amoxicillin-Clavulanate 875-125mg q12h for seven days, instructions for frequent sitz baths, and instructions to follow-up urgently in the general surgery clinic.
Introduction
Anorectal complaints are a common presentation in the Emergency Department. While significantly less common than hemorrhoids, there are an estimated 68,000 to 96,000 cases of anorectal abscesses per year in the United States. (1) This number is thought to be grossly underestimated by the misdiagnosis of abscesses as hemorrhoids by both patients and clinicians. (1) Although the diagnosis would seem straightforward, one study found that only 50% of physicians diagnosed anorectal conditions accurately, suggesting that there is a need for better understanding of the history and clinical findings associated with these conditions. (3) If not correctly diagnosed and drained, anorectal abscesses can progress and result in a generalized systemic infection often referred to as anal sepsis.
Anatomy and Pathophysiology
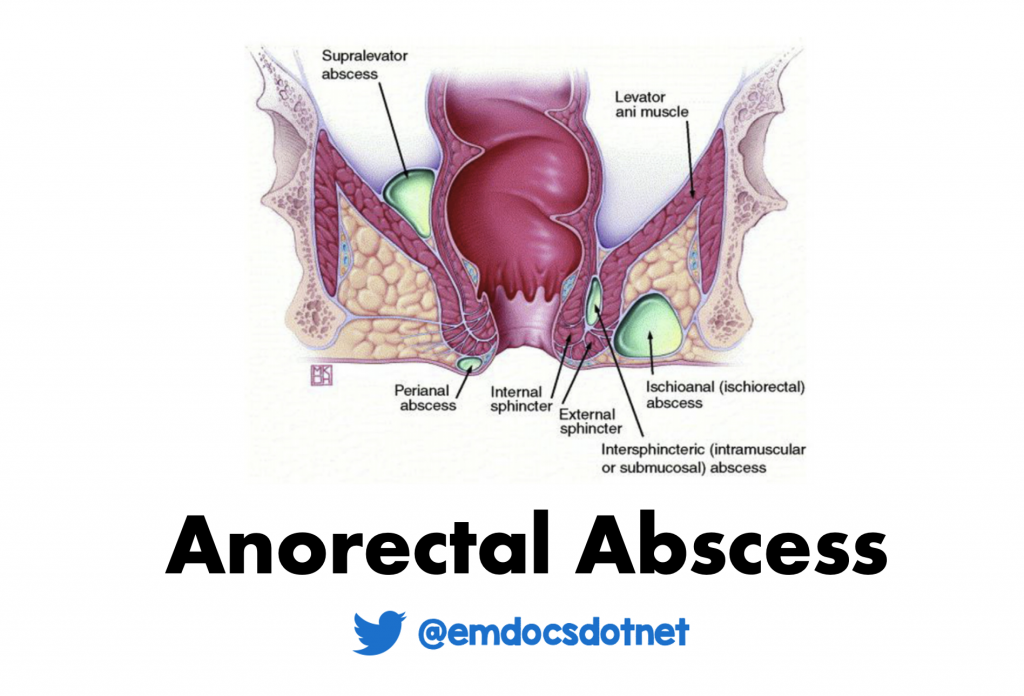
The anorectal area starts at the dentate line, where the rectosigmoid colon ends, and continues to the skin at the anal verge. The area just distal to the dentate line is the anal canal, which continues distally and ends at the anal verge (see figure 1). (1) Anorectal abscesses originate as infections in the anal glands, at or just distal to the dentate line. The anal glands are located within the internal sphincter and drain at the anal crypts into the anal canal. (1) When debris obstruct the anal crypt, bacteria proliferate, resulting in growing purulence within the gland. These abscesses are typically polymicrobial with both aerobic and anaerobic bacteria (i.e. Staphylococcus aureus, Streptococcus, Enterococcus species, Escherichia coli, Proteus, and Bacteroides). Once formed, the abscess can track into the surrounding tissues to form varying types of anorectal abscesses, each of which require specific treatment.
Classification and Presentation
Anorectal abscesses are classified based on their relationship to the anal sphincter from superficial to deep (3). These typically breakdown into perianal and perirectal (see figure 1). These can be differentiated based on presenting symptoms, location, defining characteristics on exam, and if obtained, imaging. Physical examination requires proper positioning and a DRE.
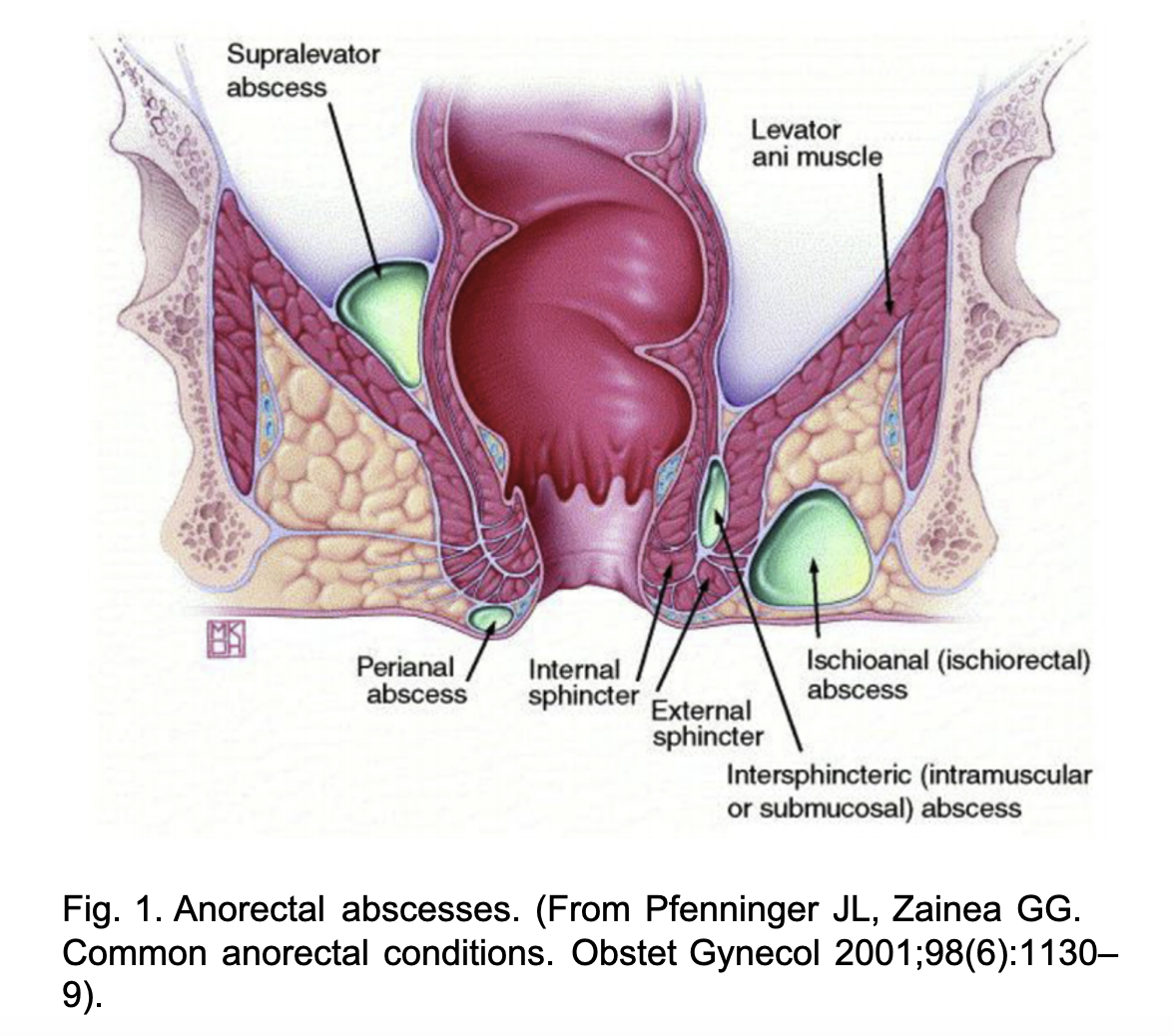
When the infection is isolated to the crypt, it is often referred to as cryptitis. (2) When the infection overwhelms the crypt space, it extends into the local surrounding tissues. Perianal abscesses are the most common comprising up to 60-70% of anorectal abscesses. This abscess results from the infection invading laterally and distally between the internal and external sphincter, also known as the intersphincteric groove, to the skin (see figure 2). (1) Perianal abscesses typically present as dull, aching, throbbing pain that is worse with defecation. (3) On exam, they are palpable, tender, fluctuant masses lateral to the anal verge within the submucosa. Perianal abscesses are not usually associated with fever or leukocytosis. (2)
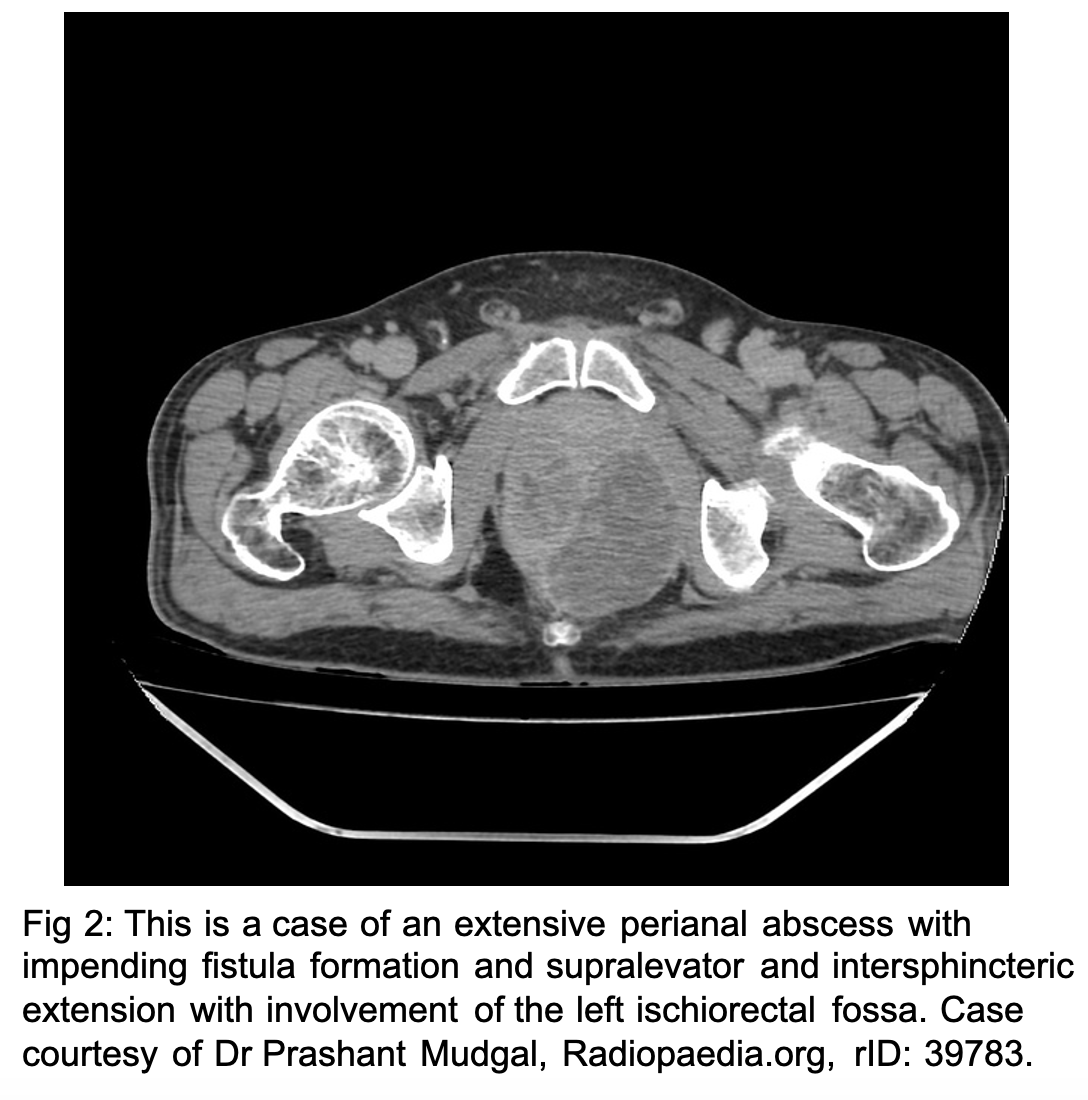
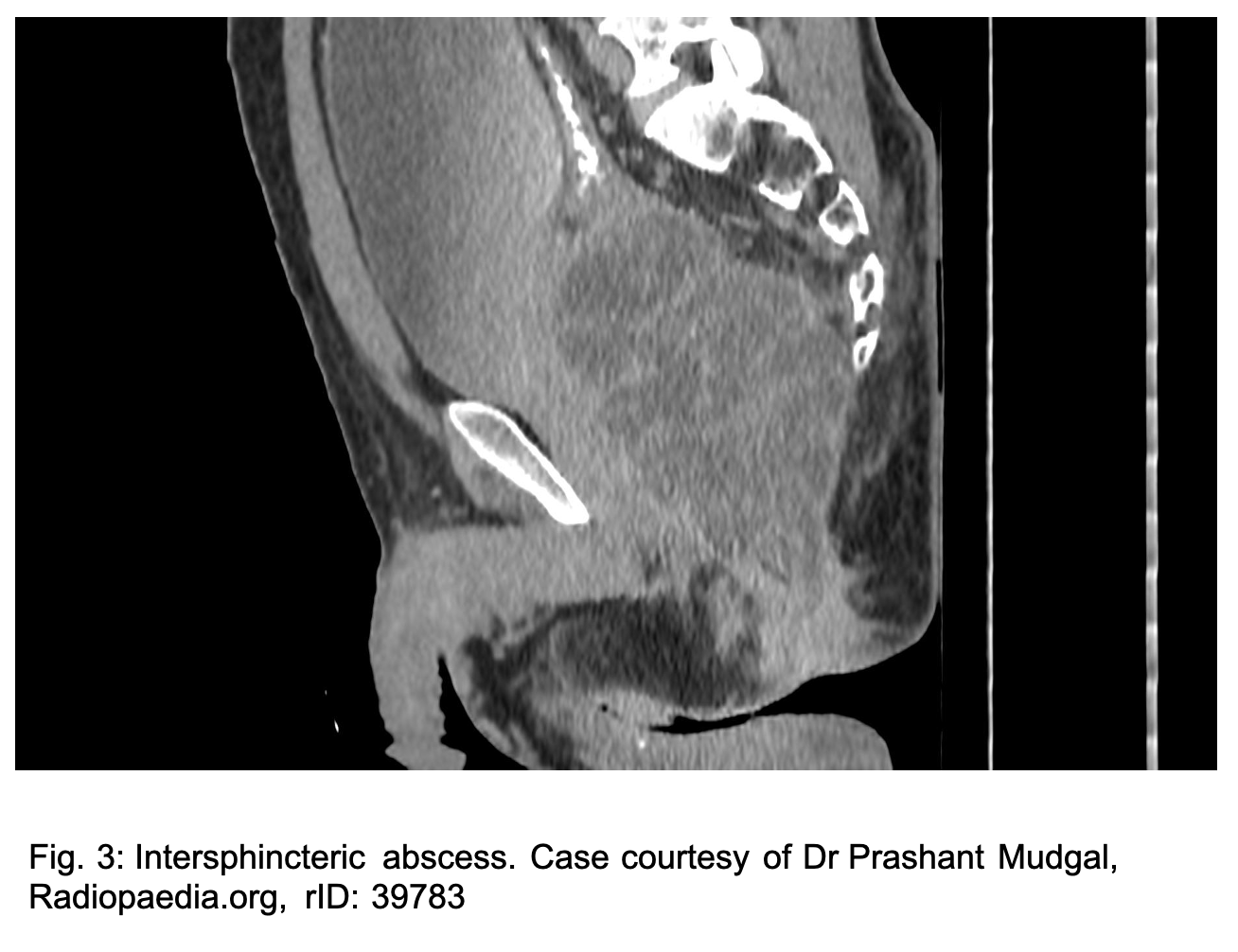
Perirectal abscesses include ischiorectal abscesses, intersphincteric abscesses, supralevator abscesses and horseshoe abscess. (2) Patients with anorectal abscesses will generally present with throbbing anorectal pain, often exacerbated by defecation. Systemic symptoms will usually present later in the course of the disease. (2) The average age of incidence is 40 in both men and women with a 2:1 male to female predominance. (1) Accounting for 20-25% of perirectal abscesses, ischiorectal abscesses develop from lateral extension of purulence through the external sphincter to the ischiorectal region. These diffusely tender, indurated, and fluctuant masses occur deeper and more lateral, within the buttock which results in pain on DRE. (2) Intersphincteric abscesses, when the infection tracks into the intersphincteric groove, and supralevator abscesses, superior to the levator ani muscles, are estimated to account for 2-5% of anorectal abscesses. (1) Intersphincteric abscesses (see figure 3) present with pain on defecation, sometimes with rectal discharge and can be palpable on DRE posterior to the midline. Supralevator abscesses result from either crypt infection tracking superiorly or secondary to an inflammatory or malignant condition of the colon such as diverticular perforation. Supralevator abscesses typically present with generalized perirectal pain, malaise, leukocytosis without external skin findings. (1,2) Horseshoe abscesses are complicated perirectal abscesses forming posterior to the anal canal and extending semicircumferentially around the internal anal sphincter in the intersphincteric plane or in ischiorectal fossa (see figure 4). They are most commonly seen in the context of Crohn’s disease and perianal fistula. (4)

Diagnostic Studies
The majority of patients presenting with superficial abscesses without systemic signs of illness can undergo I&D in the ED without further imaging. However, in patients with systemic signs of illness or concern for deeper infection, computed tomography (CT) is the most usefully imaging modality. Ultrasonography (transperineal, transperitoneal, or endorectal) can identify more superficial abscesses, such as perirectal and ischiorectal abscesses, but deeper infections require a CT scan for evaluation. (10) If the CT scan is negative and there is still a high suspicion for deep space infection, MRI should be used for evaluation. (11)
Management
Management depends upon the type of abscess. In the emergency department, uncomplicated, isolated, fluctuant perianal abscesses can be drained using local anesthetics and occasionally procedural sedation. Drainage is accomplished by either a linear or cruciate incision. If a linear incision is used, the abscess cavity should be loosely packed with iodoform gauze to prevent premature closure and proper drainage. When a cruciate incision is performed, trimming the flaps is suggested, and packing is not required. All patients should be instructed to take frequent sitz baths and follow up with a surgeon. The addition of antibiotics after routine I&D of uncomplicated anorectal abscess has not been shown to improve healing time or reduce recurrences. (13) The American Society of Colorectal Surgery (ASCRS) guidelines suggest a course of antibiotics after drainage of an anorectal abscess only in patients meeting the following criteria: extensive perianal/perineal cellulitis, signs of systemic infection, diabetes, valvular heart disease, and immunosuppression. (13) However, studies have shown that antibiotic therapy after incision and drainage have been associated with 36% lower odds of fistula formation in all patients, regardless of associated conditions. (14) All perirectal abscesses require surgical drainage in the operating room because of the complex nature of the abscesses and risk of complications such as fistula formation.
Immunocompromised
Risk factors for developing anorectal abscesses include diabetes, inflammatory bowel disease, smoking, HIV, and other causes of immunosuppression. (1) Unfortunately, immunosuppressed patients may exhibit a lack of systemic symptoms or external skin changes, and CT scan has been shown to be sensitive only 77% of the time when identifying perirectal abscesses in immunocompromised patients. (11) As such, emergency providers should have a low threshold to consider the diagnosis in immunosuppressed patients presenting with sepsis without a source or rectal pain despite a normal exam. In addition, immunocompromised patients with any evidence of sepsis or cellulitis require urgent drainage of abscess in < 24 hours due to the high risk of developing deep infection, sepsis, and necrotizing soft tissue infection. (4) As stated above, once an abscess has undergone surgical incision and drainage, many healthy patients do not require antibiotics. However, the recommendation for immunocompromised patients after I&D is usually a 5-7 day course of either amoxicillin-clavulanate or ciprofloxacin 500mg PO BID and metronidazole 500mg PO TID. (13, 15)
Pearls
- Suspect anorectal abscesses in patients with severe anorectal pain, especially when accompanied by fever or malaise.
- Superficial anorectal abscesses can be diagnosed on physical examination by perianal erythema and a palpable, fluctuant, tender mass.
- Perirectal abscesses can occasionally present with a tender, fluctuant mass on DRE, but may have limited physical exam findings and require imaging to diagnose.
- For patients with evidence of deep infection, CT is the imaging modality of choice. Transperineal ultrasound has recently been recognized as a quick, easy, noninvasive, and cost-effective technique to diagnose and monitor for resolution of anorectal abscesses.
- Perianal abscesses can be drained at the bedside in the ED while perirectal abscesses require surgical intervention in the operating room.
- Immunocompromised patients can rapidly decompensate secondary to severe infection from anorectal abscesses. They require urgent surgical intervention.
- After I&D of anorectal abscesses, immunocompromised patients require antibiotics.
References / Further Reading
- Abcarian H. Anorectal infection: abscess-fistula. Clin Colon Rectal Surg. 2011;24(1):14-21. doi:10.1055/s-0031-1272819 .
- Tupe CL, Pham TV. Anorectal Complaints in the Emergency Department. Emerg Med Clin North Am. 2016;34(2):251-270. doi:10.1016/j.emc.2015.12.013
- Grucela A, Salinas H, Khaitov S, Steinhagen RM, Gorfine SR, Chessin DB. Prospective analysis of clinician accuracy in the diagnosis of benign anal pathology: comparison across specialties and years of experience. Dis Colon Rectum. 2010;53(1):47-52. doi:10.1007/DCR.0b013e3181bbfc89.
- Sahnan K, Adegbola SO, Tozer PJ, Watfah J, Phillips RK. Perianal abscess. BMJ. 2017;356:j475. Published 2017 Feb 21. doi:10.1136/bmj.j475
- Rizzo JA, Naig AL, Johnson EK. Anorectal abscess and fistula-in-ano: evidence-based management. Surg Clin North Am. 2010;90(1):. doi:10.1016/j.suc.2009.10.001
- Guniganti, P., Lewis, S., Rosen, A. et al. Imaging of acute anorectal conditions with CT and MRI. Abdom Radiol 42, 403–422 (2017). https://doi.org/10.1007/s00261-016-0982-6
- Lavazza A, Maconi G. Transperineal ultrasound for assessment of fistulas and abscesses: a pictorial essay. J Ultrasound. 2019;22(2):241-249. doi:10.1007/s40477-019-00381-6
- Henderson PK, Cash BD. Common anorectal conditions: evaluation and treatment. Curr Gastroenterol Rep. 2014;16(10):408. doi:10.1007/s11894-014-0408-y
- Hyder JW, MacKeigan JM. Anorectal and colonic disease and the immunocompromised host. Dis Colon Rectum. 1988;31(12):971-976. doi:10.1007/BF02554899
- Plaikner, M., Loizides, A., Peer, S. et al. Transperineal ultrasonography as a complementary diagnostic tool in identifying acute perianal sepsis. Tech Coloproctol 18, 165–171 (2014).
- Glauser J, Katz J. Anorectal emergencies. Emerg Med Rep 2014;35(10):113–22.
- Caliste X, Nazir S, Goode T, et al. Sensitivity of computed tomography in detection of perirectal abscess. Am Surg. 2011;77(2):166-168.
- Vogel JD, Johnson EK, Morris AM, et al. Clinical Practice Guideline for the Management of Anorectal Abscess, Fistula-in-Ano, and Rectovaginal Fistula. Dis Colon Rectum. 2016;59(12):1117-1133. doi:10.1097/DCR.0000000000000733.
- Mocanu V, Dang JT, Ladak F, et al. Antibiotic use in prevention of anal fistulas following incision and drainage of anorectal abscesses: A systematic review and meta-analysis. Am J Surg. 2019;217(5):910-917. doi:10.1016/j.amjsurg.2019.01.015.
- Sawyer RG, Claridge JA, Nathens AB, et al. Trial of short-course antimicrobial therapy for intraabdominal infection [published correction appears in N Engl J Med. 2018 Jan 25;:null]. N Engl J Med. 2015;372(21):1996-2005. doi:10.1056/NEJMoa1411162.