Authors: Colin J. O’Shea, MD, MPH (EM Assistant Professor, Vanderbilt University Medical Center) and Matthew Pirotte, MD (EM Program Director, Vanderbilt University Medical Center) // Reviewed by: Summer Chavez, DO, MPH, MPM; Brit Long, MD (@long_brit); Alex Koyfman, MD (@EMHighAK)
Case #1
Your first patient is a 67-year-old male who presents with sudden, painless vision loss of the right eye. He describes total vision loss in all fields that began 12 hours prior to presentation. Four days ago, the patient had 20 minutes of complete vision loss of his right eye like a curtain descending that resolved spontaneously. He has a history of hypertension, type 2 diabetes mellitus, and hyperlipidemia. On exam, his acuity in the affected eye is limited to hand motion only. He has full extraocular movements, and you note that he has asymmetric pupillary response on the right when light is shined back and forth between his eyes.
Case #2
Your second patient is a 65-year-old female who presents with vision loss of the left eye with associated headache. She describes looking through a haze that was noticed when she awoke this morning – around 8 hours ago. The patient notes that she has had intermittent, brief vision loss over the past week and endorses recent headaches, fever, and muscle weakness. On exam, her visual acuity is 20/100 in the left eye and 20/25 in the right eye. Her extraocular movements are intact, and her intraocular pressure is normal.
Introduction
Vision loss can be a challenging ED chief complaint to diagnose and manage because of the difficulty determining which causes are neurological and which are related to intrinsic ocular pathology. This website has previously covered an overview of the causes of vision loss. This article will help to differentiate central (CNS) versus peripheral (ocular) causes of vision loss.
Definitions
The causes of vision loss are vast and include processes in the CNS, the orbit, and systemic toxicities. For this article, we will define central vision loss as causes that arise from the CNS – specifically, the optic nerve, the chiasm, and the retrochiasm. Peripheral etiologies are those that originate in the orbit or systemic processes.
Historical Features
When a patient presents with vision loss, there are important historical features that can help distinguish central versus peripheral causes.
- Quality of the vision loss – is it a loss of acuity or a field cut?
- Determine if the vision change is monocular versus binocular.
- Pain – ask if there is associated globe pain and/or associated headache
- Determine the time course of the vision change
- Redness
- Trauma
- Associated neurologic symptoms
It is important to qualify the vision loss and determine if it is a field cut, scotoma, or loss of acuity, for example. It is also crucial to determine if the vision change is monocular or binocular – occurring in one eye or both. Binocular diplopia resolves with covering one eye, whereas monocular diplopia persists with covering one eye. In general, any binocular process should increase your suspicion for a central process. Patients may not think to look through one eye at a time before presenting, so this may only become clear through history and exam.
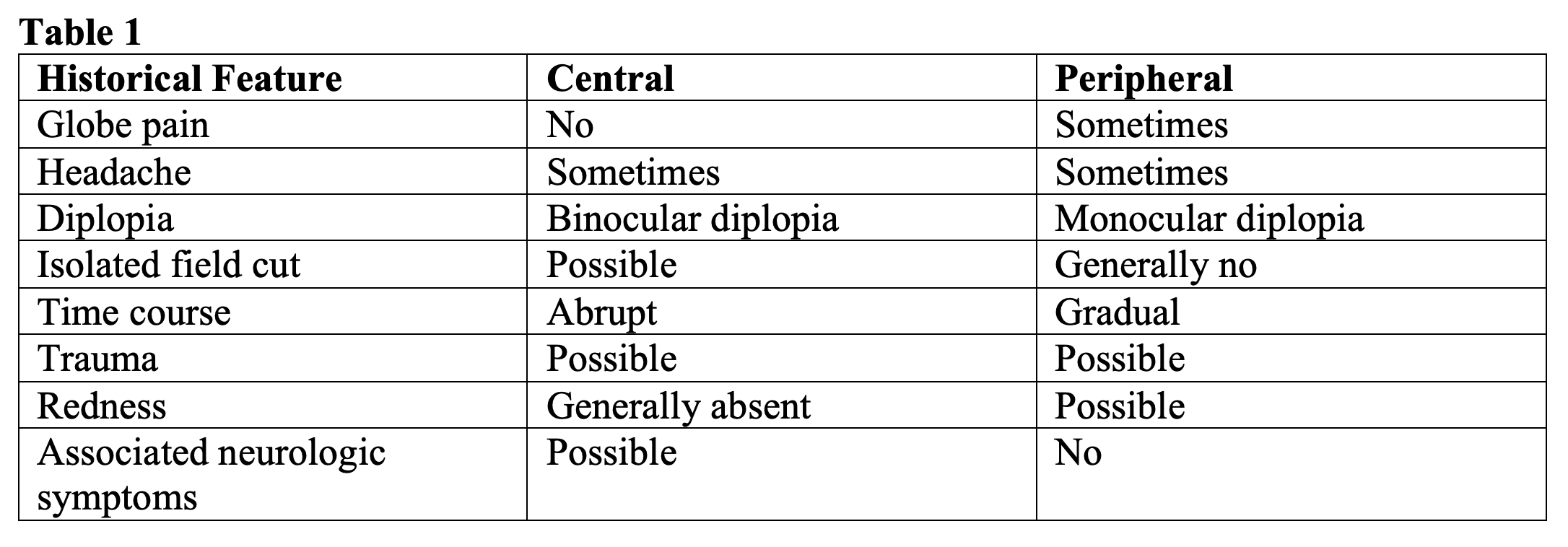
Pain – in the absence of trauma, most central causes of vision loss are painless. The few exceptions to this are optic neuritis, cavernous sinus thrombosis, and idiopathic intracranial hypertension (IIH).1-5 There are many painful causes of peripheral vision loss, from angle closure glaucoma to endophthalmitis.
Diplopia – although patients with diplopia may not have true vision loss, they may perceive diplopia as blurry vision,6 so this may be their chief complaint in the ED. Monocular diplopia persists with closing one eye and usually relates to a refractive error in the eye.6
Field cut – the one peripheral cause of a field cut is a retinal detachment. Branch retinal artery occlusion (BRAO) will have a monocular field cut, while optic pathway tumor or ischemia may have binocular field cuts.7,8
Time course – any sudden vision loss, particularly without trauma, should increase concern for a central cause.9-11 Vascular pathologies of the retina, such as central retinal artery occlusion (CRAO) and BRAO, or vascular insults of the cerebrum tend to cause acute visual deficits. Many inflammatory or infectious peripheral processes tend to cause more subacute vision loss.
Trauma – trauma can cause vision loss related to both central and peripheral etiologies.12 Central causes include cervical artery dissection and occipital lobe hemorrhage, while there are multiple peripheral causes of traumatic vision loss.
Redness – associated scleral injection occurs often with many peripheral causes, from iritis to acute glaucoma. Cavernous sinus thrombosis, however, can also cause redness and chemosis of the eye.3,4
Neurologic symptoms – any associated neurologic symptom raises concern for a central process.2,6 Cranial nerve palsies with vision loss can be seen in cavernous sinus thromboses and IIH. Optic neuritis may be associated with extremity or facial numbness or weakness if associated with multiple sclerosis.
The Eye Exam
The fundamental eye exam consists of the following: general inspection, visual acuity, pupils, confrontation visual fields, extraocular movements, external exam, eye surface (woods lamp/slit lamp, fluorescein staining), intraocular pressure, and posterior chamber evaluation. The posterior chamber is often evaluated with fundoscopy, which can be difficult to perform in the ED. Ocular ultrasound can be used.

General inspection – evaluate the lids and sclera closely for erythema, tearing, and proptosis.
Visual acuity – remember that if the patient cannot make out the letters on the Snellen chart, acuity is determined by ability to count fingers, detect hand motion, and detect light perception.
Confrontation visual fields – any bilateral field cut is concerning for a central process. As mentioned above, tumors along the optic pathway and strokes involving the occipital lobe can affect visual fields of both eyes.
Pupils – assess for shape, symmetry, and reactivity. Tear drop shaped pupils are classically seen in open globes.13 Patients with anisocoria, unequal pupillary sizes, may or may not have true vision loss. However, it is an important feature to note because of the wide differential for this finding.14 A relative afferent pupillary defect (RAPD) is concerning for optic nerve dysfunction, either from compression, ischemia, or demyelination.6, 14 Large retinal detachments and vitreous hemorrhage can also cause a RAPD if severe.
Extraocular movements – diseases affecting vision can also affect extraocular movements. Aside from trauma resulting in ocular muscle entrapment, the one peripheral cause of ophthalmoplegia is orbital cellulitis.15 More central causes are associated with ophthalmoplegia. Intranuclear ophthalmoplegia (INO) is a disorder of ocular movement causing an inability to perform conjugate lateral gaze and is due to damage at the medial longitudinal fasciculus. There are multiple possible causes of this damage and INO can occur unilaterally or bilaterally, but if INO is found, it should increase your suspicion for multiple sclerosis and optic neuritis.2
Eye Surface – in additional to evaluating for scleral erythema, use of a fluorescein stain can help in the evaluation of corneal abrasions, ulcerations, and keratopathy.
Intraocular pressure – acutely elevated intraocular pressure (IOP) can be seen in multiple peripheral causes of vision loss: angle closure glaucoma, retrobulbar hematoma, endophthalmitis, uveitis, orbital cellulitis.15-19 Cavernous sinus thrombosis, however, can also cause elevated IOP.4 Any measurement above 20 mmHg is abnormal. A measurement above 40 mmHg is an indication to start emergent therapy for acute angle closure glaucoma.13 There is not a specific cutoff for diseases other than glaucoma.
Posterior Chamber – bedside ocular ultrasound can be used to evaluate the posterior chamber and is particularly useful in the diagnoses of retinal detachments, vitreous detachments, and vitreous hemorrhage. Point of care ultrasound is fairly sensitive for retinal detachment and vitreous hemorrhage (97% and 88% respectively), but is less sensitive for vitreous detachment (43%).20
Clinical Presentation Differentials
Knowing the initial history and fundamental eye exam can narrow the differential for vision loss. The differentials for the primary complaints of field cut and vision loss have been broken down below using proposed step by step processes. Remember that each patient should have a complete history and a complete ophthalmologic physical exam, however, specific parts of the exam are helpful in distinguishing a central versus a peripheral process. A more complete list of diseases is outlined below (Table 3), but here we present an initial approach to certain chief complaints.
Field Cut
If a patient presents with an isolated field cut without loss of acuity, the differential changes whether the field cut affects one eye or both. Any binocular field cut is concerning for a central process, ischemia or tumor anywhere along the optic tract to the visual cortex.1,8 If a patient notes a monocular field cut, one helpful adjunct is ultrasound. Ultrasound can detect retinal detachment and vitreous detachment, which may cause a field cut with associated flashes and floaters. If there are no abnormal ultrasound findings, this should increase concern for a central process, such as BRAO. Vision loss in CRAO tends to be profound and involve all fields, however, central vision may be spared if the cilioretinal artery is spared.21 Keep in mind that CRAO is considered a form of ischemic stroke and patients with CRAO and BRAO should undergo vascular imaging.22

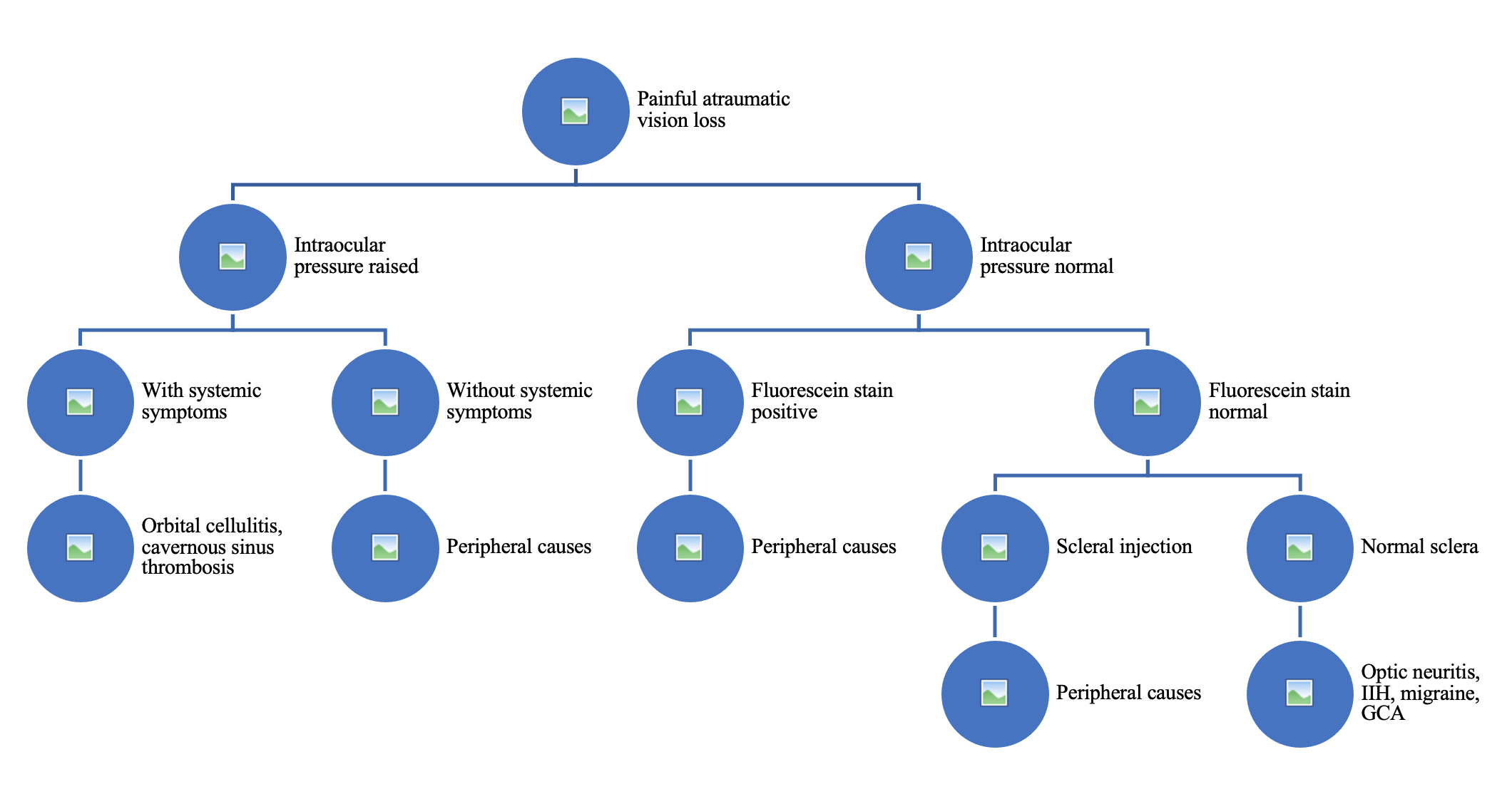
Painful Atraumatic Vision Loss
For patients who describe painful vision loss, it is important to note their intraocular pressure (IOP). As mentioned above, elevated IOP is more often seen in peripheral causes of vision loss. If a patient has elevated IOP, painful vision loss, and systemic symptoms, this should increase concern for orbital cellulitis or cavernous sinus thrombosis. There are many peripheral causes of painful vision loss that can be seen with fluorescein stain or close examination of the sclera.
Optic neuritis and IIH are two central causes of painful vision loss, however, these patients will have a normal fluorescein stain, normal intraocular pressure, and normal sclera on exam. Ultrasound may be an additional adjunct to evaluate for papilledema with fair sensitivity.23 Anterior ischemic optic neuropathy (AION) is a cause of painless vision loss; however, when it is associated with giant cell arteritis (GCA), can cause headaches and other body pain in addition to vision loss. Patients with GCA may have elevated inflammatory markers, jaw claudication, joint pain, and fever, which are unlikely to be seen in optic neuritis or IIH. It is reasonable to order an ESR and CRP on any patient over 50 who presents with the complaint of visual changes, especially if accompanied by a unilateral headache or temporal tenderness to palpation. Under this age, GCA is virtually unheard of.1
Painless Atraumatic Vision Loss
One helpful tool to use with patients with painless vision loss is ultrasound, which can quickly detect a large retinal detachment or vitreous hemorrhage. Around 25% of patients with GCA will present with painless vision loss without other symptoms;8 elevated inflammatory markers can help to make this diagnosis. CRAO and retinal vein occlusion can both present similarly – sudden onset vision loss with an associated APD and otherwise unremarkable exam. Retinal vein occlusion, however, has a different fundoscopic appearance of multiple hemorrhages throughout the retina – so called “blood and thunder”.12

Pearls
- For any patients over 50, it is reasonable to obtain an ESR and CRP to evaluate for GCA.
- A RAPD should increase your concern for optic nerve pathology.
- Monocular diplopia is almost always related to a peripheral process, while binocular diplopia should increase your concern for a central process.
- Any binocular field cut is concerning for a central process.
- CRAO should be considered a stroke equivalent.
Case Resolutions
Patient 1: On exam, you note that the patient has a RAPD. Additionally, he describes amaurosis fugax – transient loss of vision. ESR, CRP, and bedside ultrasound are normal. Your fundoscopic exam does not reveal any significant retinal hemorrhages. The patient undergoes CT/CTA of the head and neck, and neurology is consulted given the concern for CRAO. The patient is found to have a large plaque at the internal carotid artery and is admitted to neurology.
Patient 2: This is a patient presenting with vision loss with associated constitutional symptoms. Her intraocular pressure is normal. You send inflammatory markers for this patient and stain with fluorescein, which appears normal. ESR and CRP are both elevated. Given the concern for GCA causing AION, the patient is started on high dose steroids and is admitted for temporal artery biopsy.
Table 3:
References
- Kidd DP. Neuro-Ophthalmology: Illustrated Case Series. Springer-Verlag London Limited;2017.
- Hickman SJ, Raoof N, McLean RJ, Gottlob I. Vision and multiple sclerosis. Mult Scler Relat Disord. 2014;3(1):3-16.
- Frank GS, Smith JM, Davies BW, Mirsky DM, Hink EM, Durairaj VD. Ophthalmic manifestations and outcomes after cavernous sinus thrombosis in children. J AAPOS. 2015;19(4):358-62.
- Visvanathan V, Uppal S, Prowse S. Ocular manifestations of cavernous sinus thrombosis. BMJ Case Rep. 2010;3doi:10.1136
- Wall M, Kupersmith MJ, Kieburtz KD, et al. The idiopathic intracranial hypertension treatment trial: clinical profile at baseline. JAMA Neurol. 2014;71(6):693-701.
- Lemos J, Eggenberger E. Neuro-Ophthalmological emergencies. Neurohospitalist. 2015;5(4):223-33.
- Mason JO, III, Shah AA, Vail RS, Nixon PA, Ready EL, Kimble JA. Branch retinal artery occlusion: visual prognosis. Am J Ophthalmol. 2008;146(3):455-7.
- Lawlor M, Perry R, Hunt BJ, Plant GT. Strokes and vision: The management of ischemic arterial disease affecting the retina and occipital lobe. Surv Ophthalmol. 2015;60(4):296-309.
- Wray SH. Acute visual loss. Semin Neurol. 2016;36:425-432.
- Gordon LK, Levin LA. Visual loss in giant cell arteritis. JAMA. 1998;280(4):385-6.
- Heckman JG, Vachalova I, Lang CJG, Pitz S. Neuro-Opthalmology at the Bedside: A Clinical Guide. J Neurosci Rural Pract. 2018;9(4):561-573.
- Atkins EJ, Newman NJ, Biousse V. Post-Traumatic vision loss. Rev Neurol Dis. 2008;5(2):73-81.
- Go S. Ocular emergencies. In: Cline DM, Ma OJ, Cydulka RK, Meckler GD, Handel DA, Thomas SH, eds. Tintinalli’s Emergency Medicine Manual 7th Edition. McGraw Hill Medical; 2012:739-51:chap 149.
- Wilhelm H. Disorders of the pupil. Handb Clin Neurol. 2011;102:427-66.
- Hamed-Azzam S, AlHashash I, Briscoe D, Rose GE, Verity DH. Common orbital infections – state of the art – part I. J Ophthalmic Vis Res. 2018;13(2):175-182.
- Lipke KJ, Gümbel HOC. Emergency treatment of ocular trauma. Facial Plast Surg. 2015;31:345-350.
- Botting AM, McIntosh D, Mahadevan M. Paediatric pre- and post-septal peri-orbital infections are different diseases: A retrospective review of 262 cases. Int J Pediatr Otorhinolaryngol. 2008;72(3):377-83.
- Prum BE, Jr, Herndon LW, Jr, Moroi SE, et al. Primary angle closure preferred practice patern guidelines. Ophthalmology. 2016;123(1):1-40.
- Taban M, Behrens A, Newcomb RL, et al. Acute endophthalmitis following cataract surgery: a systematic review of the literature. Arch Ophthalmol. 2005;123(5):613-20.
- Lahham S, Shniter I, Thompson M, et al. Point-of-Care Ultrasonography in the Diagnosis of Retinal Detachment, Vitreous Hemorrhage, and Vitreous Detachment in the Emergency Department. JAMA Netw Open. 2019 Apr; 2(4): e192162.
- Grory BM, Lavin P, Kirshner H, Schrag M. Thrombolytic therapy for acute central retinal artery occlusion. Stroke. 2020;51(2):687-695.
- Sacco RL, Kasner SE, Broderick JP, et al. An updated definition of stroke for the 21st century: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013;44(7):2064-89.
- Teismann N, Lenaghan P, Nolan R, et al. Point-of-care ocular ultrasound to detect optic disc swelling. Acad Emerg Med. 2013;20(9):920-5.







