Author: Agnieszka Radziszewska, MD (Advocate Christ Medical Center, EM Resident Physician); Thaer Ahmad, MD (Advocate Christ Medical Center, EM Attending Physician) // Reviewed by: Sophia Görgens, MD (EM Resident Physician, Zucker – Northwell NS/LIJ, NY); Cassandra Mackey, MD (Assistant Professor of Emergency Medicine, UMass Chan Medical School); Brit Long, MD (@long_brit)
Welcome to EM@3AM, an emDOCs series designed to foster your working knowledge by providing an expedited review of clinical basics. We’ll keep it short, while you keep that EM brain sharp.
A 3-year-old male with G6PD deficiency presents to your ED by ambulance with shortness of breath, found to be hypoxic with SpO2 80% on 15L NRBM. He appears pale and dyspneic. The child is placed on HFNC with no improvement in saturation.
His remaining vital signs include T 36.6℃, BP 95/55, HR 160, RR 24.
Given his persistent hypoxia on noninvasive methods, a decision is made to intubate. Bedside CXR is unremarkable, and in spite of full ventilatory support with FiO2 of 100%, the patient’s saturation remains at 85%. You later learn he had eaten a full box of blueberries earlier that day. You quickly obtain an arterial blood gas which shows the following:
Carboxyhemoglobin 3.3
Hemoglobin: 6.0
Methemoglobin: 20
HCO3: 20.8
PCO2: 36
PaO2 :85
PH: 7.37
O2 Sat: 99%
PaO2 :85
Based on the blood gas, what is the most likely cause of the patient’s presentation? What other tests would you like to order to confirm your suspicion?
Answer: Acute Hemolytic Anemia with Methemoglobinemia 1-16
Epidemiology
- Anemia is one of the most frequently encountered lab abnormalities in clinical practice
- Definition of Anemia in pediatric population1:
- children 6 months – 6 years when hemoglobin level is < 11g/dL
- children 6 – 14 years when hemoglobin level is < 12 g/dL
- Anemia is the result of reduction in the circulating red blood cell mass which can be caused by one of three major mechanisms2:
- Decreased production of RBCs
- Increased destruction of RBCs
- Blood loss
- The normal lifespan of a red blood cell is typically 120 days3,4
- The response to anemia is a release of reticulocytes from bone marrow as old ones are removed from circulation
- Normal reticulocyte count is between 1-2%; 1% of RBCs are replaced daily
- If anemia is present reticulocyte count will increase to >3%
Hemolytic Anemia5, 6
- Red blood cell destruction termed “Hemolysis” can occur in the extravascular space (as part of the spleen, liver, or lymph nodes) or intravascular space (within vessels)
- Extrinsic Hemolytic Anemia: hemolysis due to factors outside the RBC
- Examples: Autoimmune hemolytic (AIHA), Microangiopathies (HUS, TTP, DIC), liver disease, drugs/toxins
- Coombs test positive
- Intrinsic Hemolytic Anemia: hemolysis due to factors within the RBC
- Typically normocytic with MCV range of 80-100fL
- increased reticulocyte count
- Coombs test negative
- Examples:
- Enzymopathies: G6PD deficiency
- Hemoglobinopathies: Sickle cell disease
- Membranopathies: Hereditary Spherocytosis
- Microangiopathic hemolysis
Quick Look: G6PD Deficiency7,8
- Affects ~400 million people worldwide particularly of African, Mediterranean or Middle Eastern descent
- Most common enzymatic disorder of red blood cells
- G6PD enzyme generates NADPH
- Management: Prevent hemolysis by avoidance of certain medications or oxidative stressors
- Fluoroquinolones, Dapsone, sulfonylureas, Fava beans, and even blueberries18
- Treatment: removal of offending agent, supportive care (IVF, transfuse if needed)
Quick look: Acquired Methemoglobinemia9-11
- Methemoglobin: oxidized form of hemoglobin unable to carry oxygen
- Oxidant chemicals such as topical benzocaine and sulfonamides induce methemoglobin
- Signs and symptoms: vary but can include cyanosis, dyspnea, fatigue
- Methemoglobin levels over 70% are considered lethal
- Diagnosis: Hypoxemia with SpO2 level ~85% unresponsive to oxygen supplementation
- Co-oximetry is the gold standard test
- Patient will have normal arterial oxygen concentration
- “Chocolate brown” blood
- Treatment: Methylene blue for severe signs and symptoms and/or MetHgb level >30%
- Methylene blue 1-2 mg/kg 11
- Poison control should be notified prior to administration
- Do NOT give to patients with G6PD deficiency given they lack NADPH for reduction and this may cause methemoglobin production
Hemolytic Anemia Clinical Presentation12-14
- Patient presentation can vary depending on whether the process is acute, chronic, or acute on chronic
- Symptoms are the result of the decreased oxygen carrying capacity which include: fatigue, weakness, shortness of breath, headache
- Severe cases of anemia may present as congestive heart failure
- Exam findings in hemolytic anemia may include:
- Cyanosis if hemolysis is rapid in onset
- Conjunctival, tongue, palmar or nailbed pallor
- Splenomegaly
- Jaundice from unconjugated bilirubin
- Dark/pink tinged urine

- Labs suggestive of hemolysis: 2,5
- Decreased Hgb, reticulocytosis
- Elevated indirect bilirubin, LDH, or haptoglobin
- Peripheral smear may show spherocytes or schistocytes
- Urinalysis may show bilirubinuria or hemoglobinuria
- Bottom line: Patients with reticulocytosis and indirect bilirubinemia have hemolysis
Treatment:
ABCs:
- Airway: Assess the airway given severe anemia can result in altered mental status
- Breathing: Dyspnea, Tachypnea, or hypoxia may be present and oxygen supplementation may be needed
- Circulation: Transfuse if symptomatic and Hgb level <6-7 g/dL16
- Pediatric transfusion is usually 10-15mL/kg
- This raises Hgb by 2-3 g/dL
- Packed red blood cells contain 250-300mL per unit
- In adults, 1 unit raises Hgb by 1g/dL and hematocrit by 3%
- In pediatrics, give over 4 hours to avoid circulatory overload which is equivalent to a rate of about 2.5 mL/kg/hr
- If there is profound active bleeding massive transfusion protocol can be initiated – this entails transfusing an infant’s/child’s total blood volume within 24 hours and using rapid infuser17
- Hematology Consultation
- Pediatric transfusion is usually 10-15mL/kg
Disposition:
- Admission for patients with hypoxia, acidosis, hemodynamic instability or newly diagnosed anemia
- Obtain hematology consultation in patients with bleeding disorders with anemia of unknown etiology and need for transfusion
Pearls:
- An ABG or VBG is a fast way to assess the patient’s true oxygenation status and obtain electrolyte, Co-oximetry, and hemoglobin levels.
- Some facilities can only run co-oximetry on ABG, otherwise can use VBG.
- Provide supportive care in the case of G6PD deficiency, including oxygen, IVF, and removal of the offending agent.
- If the patient’s hypoxia is refractory to maximal oxygen support, obtain co-oximetry.
- If transfusion is required in pediatric patients, it is weight based at 10-15cc/kg and administered at 2.5 cc/kg/hr.
- Do NOT give methylene blue to a patient with G6PD deficiency as it may precipitate further hemolysis.

A 35-year-old man with a history of HIV and recurrent anemia presents to the ED with increasing fatigue after being started on dapsone. He is noted to have scleral icterus on physical exam. Which of the following laboratory findings would be expected?
A) Heinz bodies on peripheral smear
B) Low lactate dehydrogenase
C) Positive direct Coombs test
D) Positive indirect Coombs test
Answer: A
Patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency will have a low hemoglobin concentration level and Heinz bodies on peripheral smear when exposed to oxidative stress. G6PD deficiency is an inherited disorder found mainly in patients with African, Asian, orMediterranean ancestry. Glutathione is responsible for protecting red blood cells from harmful oxidative metabolites. Impairment in this protective mechanism results in hemolytic anemia when red blood cells are exposed to oxidative stress. Certain drugs, such as dapsone, phenazopyridine,nitrofurantoin, primaquine, rasburicase, and methylene blue, have been implicated in precipitating acute hemolysis in patients with G6PD deficiency. Patients can present with symptoms related to anemia, such as fatigue or dyspnea. They may also present with jaundice from an increase in indirect bilirubin from the breakdown of red blood cells.
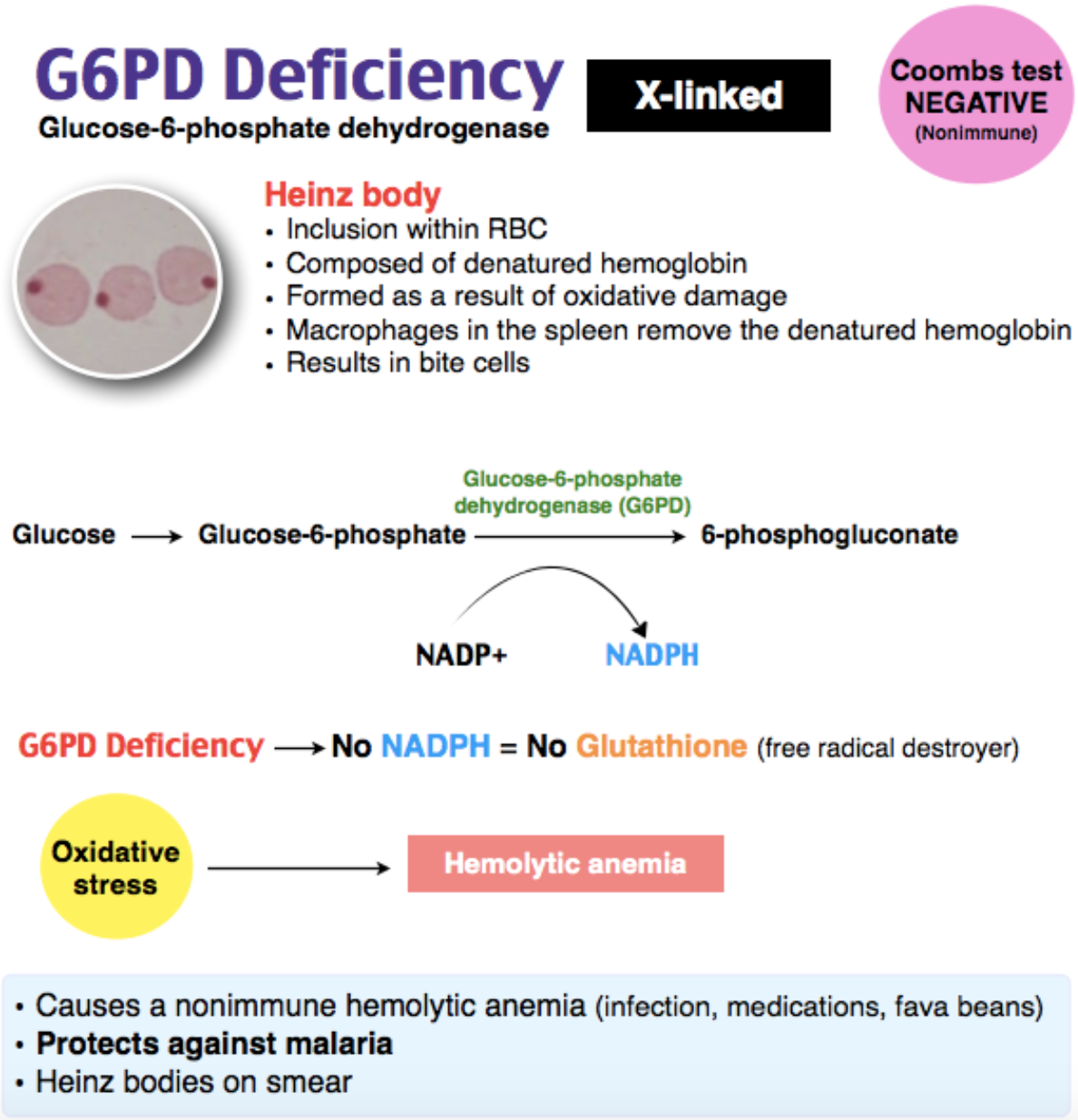
A low lactate dehydrogenase (LDH) (B) is not consistent with hemolytic anemia. Lab findings consistent with acute hemolytic anemia include an elevated LDH, low haptoglobin, and elevated indirect bilirubin. The Coombs tests are used to identify patients with autoimmune hemolytic anemia. A positive direct Coombs test (C) identifies autoantibodies attached to red blood cells, while a positive indirect Coombs test (D) confirms autoantibodies in the serum. G6PD causes nonimmune hemolytic anemia and thus has a negative Coombs test.
Further Reading
- Conrad ME. Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd edition. Boston: Butterworths; 1990. Chapter 147. Available from: https://www.ncbi.nlm.nih.gov/books/NBK254/
- Tintinalli’s Emergency Medicine: A Comprehensive Study Guide. McGraw-Hill Education; 2019.
- Franco RS. Measurement of red cell lifespan and aging. Transfus Med Hemother. 2012;39(5):302-307. doi:10.1159/000342232
- ALLISON AC. Turnovers of erythrocytes and plasma proteins in mammals. Nature. 1960;188:37-40. doi:10.1038/188037a0
- Cline D. Tintinalli’s Emergency Medicine: Just the Facts. McGraw-Hill; 2013.
- Sattar HA. Fundamentals of Pathology: Medical Course and Step 1 Review. Pathoma.com; 202
- Cappellini MD, Fiorelli G. Glucose-6-phosphate dehydrogenase deficiency. Lancet. 2008;371(9606):64-74. doi:10.1016/S0140-6736(08)60073-2
- Luzzatto L, Seneca E. G6PD deficiency: a classic example of pharmacogenetics with on-going clinical implications. Br J Haematol. 2014;164(4):469-480. doi:10.1111/bjh.12665
- Ashurst J, Wasson M. Methemoglobinemia: a systematic review of the pathophysiology, detection, and treatment. Del Med J. 2011;83(7):203-208
- Wright RO, Lewander WJ, Woolf AD. Methemoglobinemia: etiology, pharmacology, and clinical management. Ann Emerg Med. 1999;34(5):646-656. doi:10.1016/s0196-0644(99)70167-8
- Kearney TE, Manoguerra AS, Dunford JV Jr. Chemically induced methemoglobinemia from aniline poisoning. West J Med. 1984;140(2):282-286
- Benseñor IM, Calich AL, Brunoni AR, et al. Accuracy of anemia diagnosis by physical examination. Sao Paulo Med J. 2007;125(3):170-173. doi:10.1590/s1516-31802007000300008
- Kalantri A, Karambelkar M, Joshi R, Kalantri S, Jajoo U. Accuracy and reliability of pallor for detecting anemia: a hospital-based diagnostic accuracy study. PLoS One. 2010;5(1):e8545. Published 2010 Jan 1. doi:10.1371/journal.pone.0008545
- Walker, Patricia Frye, and Elizabeth D. Barnett. “Chapter 46: Anemia and Red Blood Cell Disorders.” Immigrant Medicine, Elsevier Mosby, St. Louis, MO, 2007.
- Lee, A.Q., Aronowitz, P. Conjunctival and Palmar Pallor. J GEN INTERN MED 36, 3575–3576 (2021). https://doi.org/10.1007/s11606-021-06981-5
- Davies P, Robertson S, Hegde S, Greenwood R, Massey E, Davis P. Calculating the required transfusion volume in children. Transfusion. 2007;47(2):212-216. doi:10.1111/j.1537-2995.2007.01091.
- Diab YA, Wong EC, Luban NL. Massive transfusion in children and neonates. Br J Haematol. 2013;161(1):15-26. doi:10.1111/bjh.12247
- Babu T, Panachiyil GM, Sebastian J, Ravi MD. Probable blueberry-induced haemolysis in a G6PD deficient child: A case report. Nutr Health. 2019;25(4):303-305. doi:10.1177/0260106019885290






1 thought on “EM@3AM: Acute Hemolytic Anemia”
Pingback: Quiz 174, January 6th, 2023 – The FOAMed Quiz