Author: Amber Cibrario, DO (EM Attending Physician, San Antonio, TX), Zachary Baker, DO (EM Attending Physician, San Antonio, TX), Rachel Bridwell, MD (@rebridwell, EM Resident Physician, SAUSHEC / San Antonio, TX) // Reviewed by: Alex Koyfman, MD (@EMHighAK) and Brit Long, MD (@long_brit)
Welcome to EM@3AM, an emDOCs series designed to foster your working knowledge by providing an expedited review of clinical basics. We’ll keep it short, while you keep that EM brain sharp.
A 3-day full term male who was discharged on day 2 after birth presents with mom with poor feeding and lethargy that has become worse, looking grey. He is breastfed every 2 hours, but he becomes diaphoretic with feeds and breaths very heavily. Mom denies fevers, URI symptoms, cough, vomiting, diarrhea, blood in stool. He has not gained weight since birth. Review of systems is otherwise unremarkable.
Triage vital signs (VS): BP 52/41, HR: 178, RR 62, SpO2 94% on room air, rectal temp 37.1. He is lethargic with weak cry, sunken fontanelle, prolonged capillary refill, hepatomegaly, cool extremities, mottled skin, and poor tone.
What should you consider?
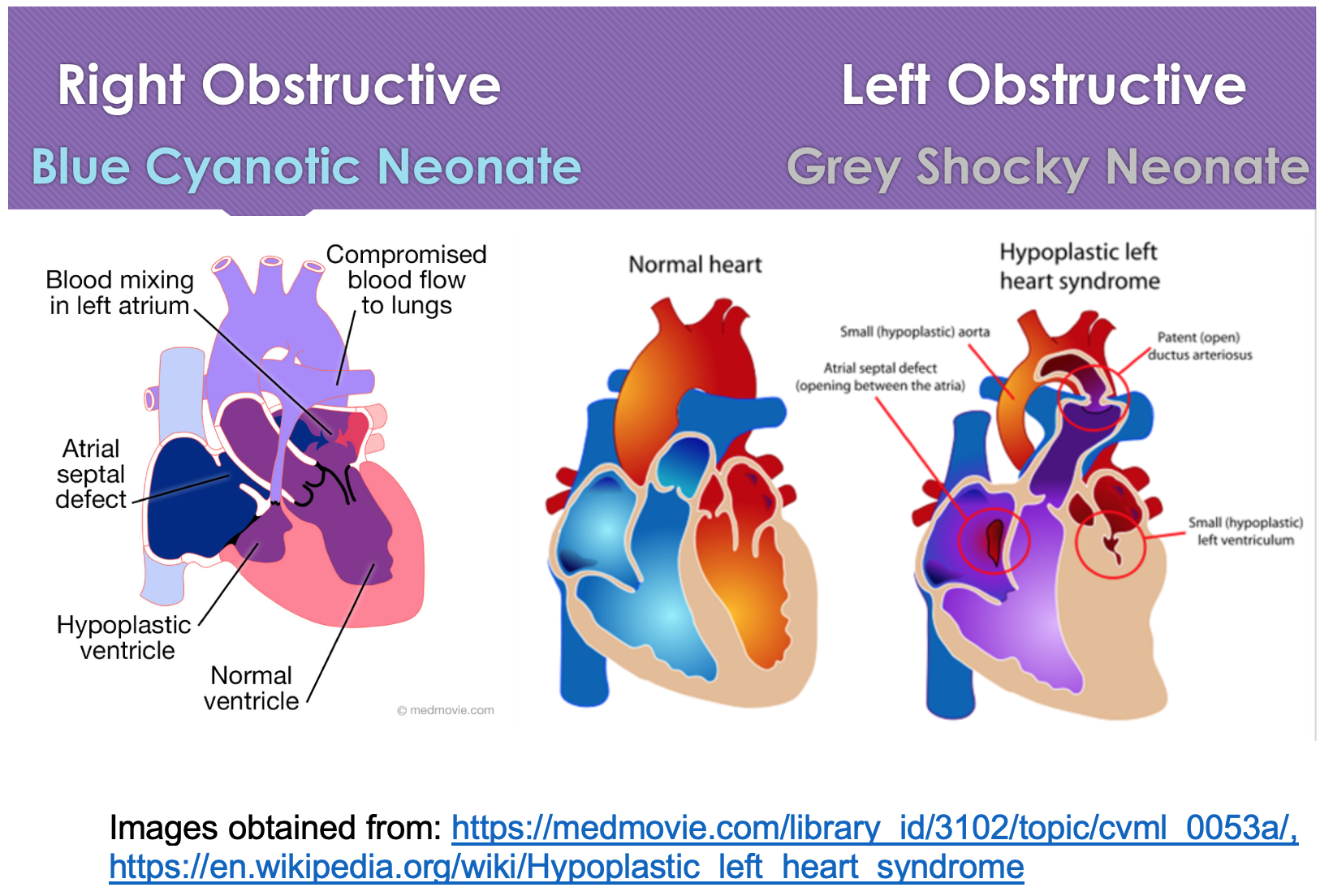
Answer: Left Obstructive Ductal Dependent Pathology1-13
Epidemiology:
- CHD is the most common major congenital anomaly and is the most common cause of mortality from birth defects in infants.1,2
- Prevalence
- 1-9:1000 live births Worldwide.3,4
- 2:1000 of live births in the US ~ 40,000 births/year have hemodynamically significant CHD.3,4
- 50% are discharged from the nursery with missed congenital heart lesion.3,4
- 25% have critical lesions that end up in the ED shortly after.3
- These critical congenital lesions are often missed both at discharge from delivery and in the ED leading to death.3–5
- The number of congenital heart lesions is increasing.3,4
- Prevalence when compare that to hemodynamically significant septic shock.
- 01:1000 sick neonates have significant septic shock.
- If you have a neonate poorly perfused and in shock (<30 days), CHD should be higher on index of suspicion than sepsis unless a measured fever is present.
- Neonates in shock < 2 weeks of life are often misdiagnosed as neonatal sepsis, incurring morbidity and mortality.
- Risk Factors:6–8
- Genetics: Family history especially siblings, chromosomal abnormalities (e.g. Trisomy 13, 18, Turner Syndrome, DiGeorge, CHARGE, VACTERL)
- Environmental:
- Maternal factors: phenylketonuria, advanced maternal age, febrile illness during pregnancy
- Infections: Influenza, rubella
- Medications: NSAIDs, Lithium, Sulfasalazine, TMP-SMX, phenytoin
- Toxins: Marijuana, Organic Solvents
Anatomy and Pathophysiology: Right Obstructive Ductal Dependent Lesions
- Common Causes
- Critical Coarctation of the Aorta = most common9
- Hypoplastic Left Ventricle = most severe9
- Critical Aortic Stenosis10
- Pathophysiology:
- Lesions above have some type of structural systemic blood flow obstruction + some type of septal defect; however, early on the PDA helps provide more blood to the body to compensate.10
- When the PDA closes, the systemic blood supply is cut off, causing hypoperfusion, shock, and acidosis.11
Clinical Presentation:
- Utilize pediatric assessment triangle: Appearance, work of Breathing, Color.12
- On history: fussiness, tachypnea, and poor feeding.9
- While this case is a left obstructive lesion, a broad differential should be considered in neonatal patients. Please see this fantastic post for the MISFITS and other great pearls: The Sick Neonate.
- Physical Exam:9
- General: Toxic appearing neonate in shock
- HEENT: Dry oral mucosa
- Respiratory: Tachypnea> 60 bpm with normal lungs sounds
- Cardiac: Tachycardic, may have a murmur but difficult to detect with elevated HR
- Abdomen: Hepatomegaly
- Skin: Mottled, cool extremities, poor capillary refill (>5 secs)
Evaluation:
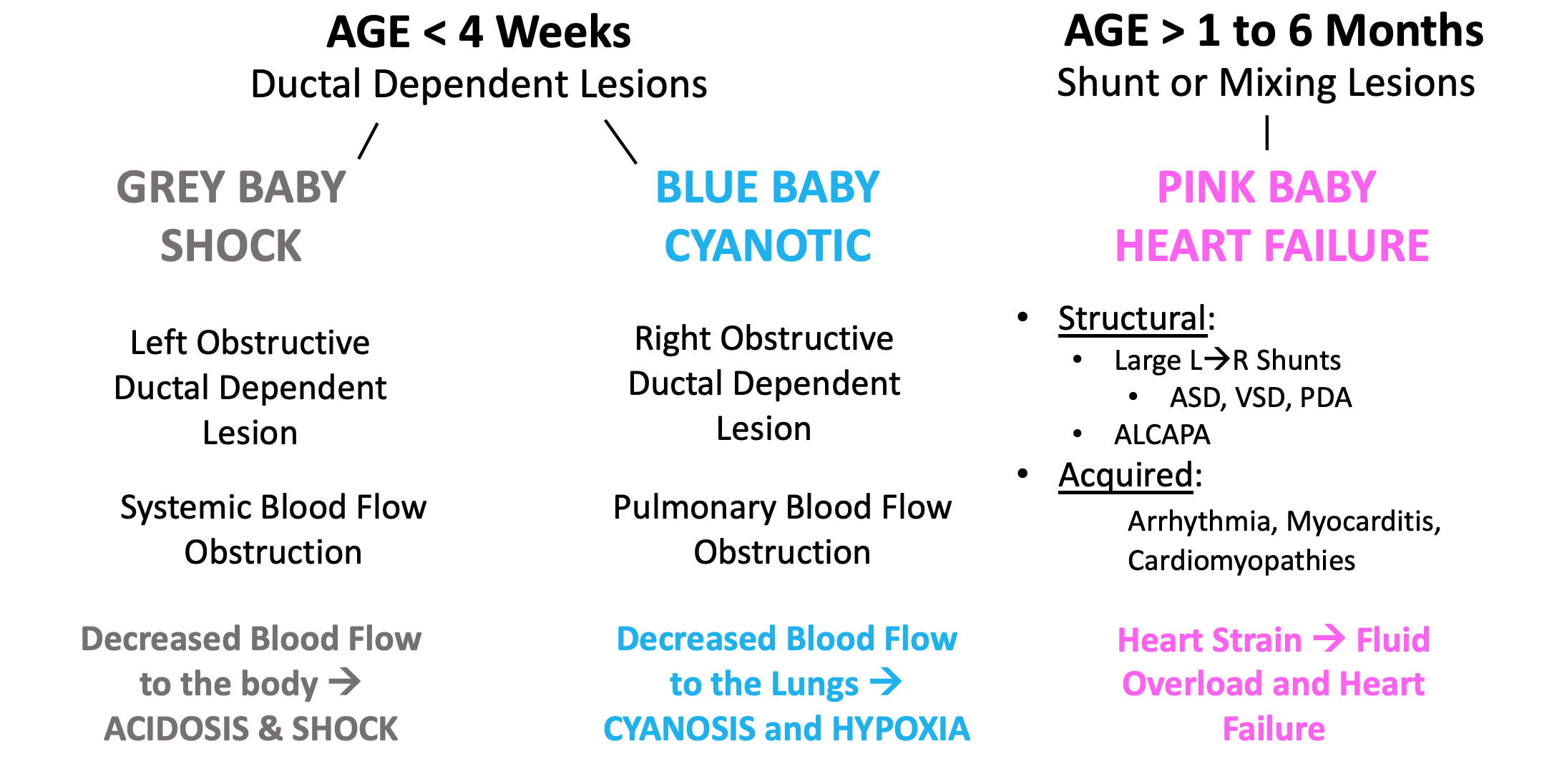
- Grouping congenital heart diseases based on several factors simplifies recognition and management.13
- Age
- Color
- 4 Extremity BPs, 4 Extremity pulse Oxs, Hyperoxia test, CXR
- Initial critical actions
- Glucose:
- Critically ill neonates have limited glycogen stores and thus are often hypoglycemic.15
- IV/IO – 2 IV attempts then place an IO.
- Need 2 access points if CHD is suspected.
- Monitor
- 4 extremity BPs
- Blood pressure differential:Delta of preductal (right arm) BP and lower extremity blood pressure of > 10 mmHg, consider obstructive process.16
- 4 extremity pulse ox
- O2 saturation differential:17
- Difference in O2 saturation > 3% between preductal (right upper) and post ductal (right lower extremity)
- <94% in lower extremities
- < 90% in any extremity require further investigation.
- Differentiates a systemic obstructive process (e.g. critical coarctation) expediently.
- O2 saturation differential:17
- What if you are not sure if this is Pulmonary vs. Cardiac cause?13
- Hyperoxia test for ~5 min
- Pulse Ox or ABG looking at PaO2
- Place infant on ~100% FiO2 using NRB for ~5-10min
- If SpO2 or PaO2 improves, likely respiratory cause.
- If SpO2 or PaO2 does not improve, likely cardiac cause.
- iSTAT or Cap Gas can help immediately narrow THE MISFITS differential, especially electrolyte derangements and congenital adrenal hyperplasia if normal glucose, K, and Na.15
- Hyperoxia test for ~5 min
- Glucose:
- What diagnostic test should we order?
- Labs13
- Full Sepsis Evaluation – CBC, CMP, lactate, Blood Cultures, UA, Urine CX, CSF (LP once stabilized).
- Ketones, Ammonia, Lactate – help us determine if there is inborn error of metabolism to rule out THEMISFITS
- Serum and urine drug screen
- Caregivers often rub winter green oil on infants, precipitating salicylate toxicity, which may present with tachypnea and poor feeding
- BNP:18
- <100 pg/mL 100% sensitivity and 98% specificity for detection of significant congenital heart disease.
- <132.5 pg/mL to detect physiologically significant L to R shunts, 93% SN and 100% SP.
- >40 pg/mL accuracy of 84% to differentiate cardiac from pulmonary etiologies of respiratory distress.
- TSH – helps determine endocrine abnormalities.
- ECG
- Newborn ECGs are different:
- Newborns are right heart dominant so normal ECG can demonstrate RAD.
- Signs of LVH in a newborn are never normal.
- Think Right Obstructive Ductal Dependent Lesion
- Normal Peds ECG can have persistent juvenile T wave inversions in V1-V3.
- Persistent RVH after 2-4 weeks is abnormal.
- Fatal arrhythmia, especially Prolonged QT > 500 ms
- ALCAPA17,18
- Big deep Q waves in lateral leads, ST segment elevation, T-wave inversion in lateral leads.
- Severe RVH13
- Upright T wave in V1 after 1 week is abnormal, suggestive of left obstructive ductal dependent lesion.
- Newborn ECGs are different:
- Chest X-ray
- Abnormal findings
- Cardiomegaly13,14,19
- Boot Shaped Heart
- Eggs on a String Sign, not always present
- Snowman Sign
- Clear black lungs19
- Obstructive pulmonary blood flow
- Cardiomegaly13,14,19
- Abnormal findings
- POCUS20
- Assess for the following features:
- Number of cardiac chambers
- Pericardial effusion
- Poor global cardiac function
- Septum present
- Plethoric IVC (if not on positive pressure)
- B lines suggestive of pulmonary edema
- Assess for the following features:
- Labs13
Treatment:
- Oxygen
- Oxygen is a potent pulmonary vasodilator, which worsen shunting in CHD patients, decreasing PVR and increasing pulmonary blood flow, generating pulmonary over-circulation and clinical deterioration.13
- Goal oxygen ~88-90%. If congenital heart disease is suspected, do not empirically place on 100% oxygen.13
- IVFs
- Judicious fluid resuscitation is key.
- 5ml/kg is the starting recommended fluid bolus in patients with CHD to balance their metabolic demands with their obstructive physiology.
- POCUS with a plethoric IVC or obvious hepatomegaly should clue the provider that a relatively fluid overloaded state is present.
- Prostaglandin is the crucial intervention.
- Alprostadil 0.05-0.1 mcg/kg/min (titrate slowly to effect or a maximum of 0.4 mcg/kg/min).21,22
- Do not bolus, as a bolus with precipitate side effects.23
- Complications/adverse effects:23
- Apnea – prepare for intubation
- Hypotension – have vasopressors ready
- Hyperthermia
- Complications may not be immediate
- Contraindications:
- TAPVR: worsens shunt physiology with patency of ductus arteriosus
- Give in any neonate in first 30 days of life with cyanosis or circulatory collapse.22
- Alprostadil 0.05-0.1 mcg/kg/min (titrate slowly to effect or a maximum of 0.4 mcg/kg/min).21,22
- RSI for Intubation
- Intubate with caution and be deliberate with medication selection as the wrong choice can precipitate cardiac collapse.
- Induction:
- While pretreatment with atropine remains controversial, consider in neonates with cardiac etiology.
- Avoid ketamine in this population21
- Increase SVR worsens left-to-right shunting and systemic flow with subsequent cardiac collapse.
- Etomidate 0.3 mg/kg13,21
- Fentanyl 1-2mcg/kg13,21
- High doses can cause cardiac depression, so be cautious
- Avoid benzodiazepines21
- Adverse effects of cardiac depressant
- Paralytics: Better to paralyze to improve 1st pass success
- Rocuronium or succinylcholine13
- Induction:
- Intubate with caution and be deliberate with medication selection as the wrong choice can precipitate cardiac collapse.
- Push dose vasopressors
- Push dose epinephrine ready for intubation, if preintubation blood pressure is hypotensive, or have vasopressors already on board.
- Avoid phenylephrine
- The elevated SVR increases afterload and worsens obstruction
- Ventilator Settings13
- Lung protective tidal volume
- Titrate O2 ~88-90% to avoid pulmonary recirculation.
- Low PEEP = 3
- Positive pressure ventilation increases PVR, decreasing preload and SVR which can affect shunting flow.
- RR 30-40 (normal for neonates)
- Vasopressors, inotropes, chronotropes in their preferred order
- Milrinone – Reduces afterload and increases contractility21
- Dose: continuous IV or intraosseous infusion; infusion dose range: 0.25 to 0.75 mcg/kg/minute
- Some experts say to start at 0.01mcg/kg/min but will likely need to titrate quickly for effect to 0.5mcg/kg/min.
- Not usually available in the ED
- Dobutamine – Readily available in ED in crash carts
- Dose: 5-1 mcg/kg/min IV continuous infusion initially and titrate21
- Titrate 5-10 mcg/kg/min for beta effects
- Do NOT exceed 40 mcg/kg/min
- Dose: 5-1 mcg/kg/min IV continuous infusion initially and titrate21
- Epinephrine – This medication can increase SVR, especially at high doses. Lower dose can be used if other agents are unavailable, as this has more beta effect.
- Dose:01–0.5 mcg/kg/min start low and titrate
- Remember higher doses -> alpha effect -> increase SVR
- 01-1mcg/kg/min
- Avoid phenylephrine norepinephrine – increase SVR due to alpha agonism dominance
- Avoid dopamine – difficult to titrate and can increase SVR
- Still controversial
- Recommend discussion with receiving PICU
- Milrinone – Reduces afterload and increases contractility21
- Antibiotics
- Critically ill neonate requires empiric sepsis coverage.
- IV Ampicillin 50 mg/kg/Gentamicin 4 mg/kg or Ampicillin 50 mg/kg/Cefotaxime 25 mg/kg
- Antivirals
- Cover for HSV with IV Acyclovir 20mg/kg
Disposition:
- PICU with Peds Cardiothoracic Surgery and Peds Cardiology consults often requires transfer to tertiary facility with above consultants available.
- Critical things the PICU and Cardiothoracic Surgeon need to know at minimum:
- Age
- Shock or Cyanotic
- 4 extremity blood pressures
- 4 extremity pulse ox
- CXR findings
- Interventions
Pearls:
- With a neonate in shock, consider all etiologies in THEMISFITS and do not anchor on sepsis.
- A sick neonate that presents ashen, in shock, and afebrile is more likely to have left obstructive ductal dependent lesion than sepsis.
- In every sick neonate need to obtain: POC glucose, 4 extremity BPs, 4 extremity pulse ox, CXR, ECG, Beside US, Labs to assess for obstructive lesions.
- You need a reason NOT to give prostaglandins in a sick neonate (< 30 days old).
- Be prepared for apnea, hypotension, hyperthermia.
- 2 IVs or IOs are critical.
- All sick neonates need full septic work up and treatment even if likely CHD.
- Broad spectrum ABX, antivirals, labs, cultures, etc.
- Empiric sepsis resuscitation can kill those with cardiac obstructive pathology so carefully evaluate the sick neonate.

From Dr. Katy Hanson at Hanson’s Anatomy:

A 7-day-old boy presents to the emergency department with poor feeding. The infant was born at term with a routine prenatal course and delivery. He was feeding well prior to discharge but has been worsening over the last few days. On exam, his heart rate is 190 beats per minute, right arm blood pressure is 62/36 mm Hg, and capillary refill is 5 seconds. He has a systolic murmur and an absent femoral pulse. He is diaphoretic and has poor respiratory effort. Positive pressure ventilation and intravenous access are obtained. His blood glucose and bedside electrolytes are normal. The patient’s vital signs do not change after boluses or inotropic infusions. Which of the following is the most likely diagnosis?
A) Bacterial sepsis
B) Coarctation of the aorta
C) Congenital adrenal hyperplasia
D) Galactosemia
Answer: B
Many congenital heart diseases depend on the ductus arteriosus to maintain appropriate blood flow. Ductal dependent lesions can be divided into those that have restriction in pulmonary blood flow (e.g., pulmonary atresia, tetralogy of Fallot, tricuspid atresia), systemic blood flow (e.g., coarctation of the aorta, interrupted aortic arch, left ventricle hypoplasia), and those that require mixing (e.g., transposition). The degree of restriction of blood flow and exact location of the coarctation determines whether the patient can tolerate closure of the ductus arteriosus. Patients with mild postductal coarctation of the aorta often go undiagnosed until found to be hypertensive at a routine physician visit later in life. Patients with severe coarctation of the aorta will lose all systemic blood flow as the ductus arteriosus closes, developing metabolic acidosis, shock, and circulatory collapse. The ductus arteriosus usually closes within the first 2 weeks of life, so ductal dependent cardiac defects should be considered for infants presenting in cardiovascular collapse in this timeframe. This is less common than sepsis and easily missed in this age group. Clues to the diagnosis of a critical coarctation of the aorta include weak or absent lower extremity pulses, a radial-femoral delay in pulse (while palpated simultaneously), and a differential cyanosis (a higher right arm or preductal oxygen saturation compared to the leg). Treatment is with a prostaglandin (e.g., alprostadil), which can cause apnea in some infants.

Bacterial sepsis (A) is a common cause of a septic-appearing infant, often from a urinary tract infection, but this typically responds to fluid boluses and inotropic medication when needed. Congenital adrenal hyperplasia (C) can also cause a septic-appearing infant and is easily missed in boys, but patients will have hypoglycemia and hyponatremia when presenting in shock. Galactosemia (D) can also present with a septic-appearing infant within the first few weeks of life, but this is usually from Escherichia coli sepsis. It should respond as bacterial sepsis and more commonly presents with jaundice, vomiting, hepatomegaly, and failure to thrive.
Further Reading:
FOAM Resources
Emergency Medicine Cases: Congenital Heart Disease
PEM PLAYBOOK: Undifferentiated Sick Neonate
References
- Van Der Bom T, Zomer AC, Zwinderman AH, Meijboom FJ, Bouma BJ, Mulder BJM. The changing epidemiology of congenital heart disease. Nat Rev Cardiol. 2011;8(1):50-60. doi:10.1038/nrcardio.2010.166
- Van Der Linde D, Konings EEM, Slager MA, et al. Birth prevalence of congenital heart disease worldwide: A systematic review and meta-analysis. J Am Coll Cardiol. 2011;58(21):2241-2247. doi:10.1016/j.jacc.2011.08.025
- Chang RKR, Gurvitz M, Rodriguez S. Missed diagnosis of critical congenital heart disease. Arch Pediatr Adolesc Med. 2008;162(10):969-974. doi:10.1001/archpedi.162.10.969
- CDC. Data and Statistics on Congenital Heart Defects. https://www.cdc.gov/ncbddd/heartdefects/data.html. Accessed September 15, 2020.
- Savitsky E, Alejos J, Votey S. Emergency department presentations of pediatric congenital heart disease. J Emerg Med. 2003;24(3):239-245. doi:10.1016/S0736-4679(02)00753-9
- Jenkins KJ, Correa A, Feinstein JA, et al. Noninherited risk factors and congenital cardiovascular defects: Current knowledge – A scientific statement from the American Heart Association Council on Cardiovascular Disease in the Young. Circulation. 2007;115(23):2995-3014. doi:10.1161/CIRCULATIONAHA.106.183216
- Fung A, Manlhiot C, Naik S, et al. Impact of prenatal risk factors on congenital heart disease in the current era. J Am Heart Assoc. 2013;2(3). doi:10.1161/JAHA.113.000064
- Miller A, Riehle-Colarusso T, Siffel C, Frías JL, Correa A. Maternal age and prevalence of isolated congenital heart defects in an urban area of the United States. Am J Med Genet Part A. 2011;155(9):2137-2145. doi:10.1002/ajmg.a.34130
- Okada PJ, Hicks B. Neonatal surgical emergencies. Clin Pediatr Emerg Med. 2002;3(1):3-13. doi:10.1016/S1522-8401(02)90012-1
- Brown K. The infant with undiagnosed cardiac disease in the Emergency Department. Clin Pediatr Emerg Med. 2005;6(4):200-206. doi:10.1016/j.cpem.2005.09.006
- Brown K. The infant with undiagnosed cardiac disease in the Emergency Department. Clin Pediatr Emerg Med. 2005;6(4):200-206. doi:10.1016/j.cpem.2005.09.006
- Horeczko T, Gausche-Hill M. The paediatric assessment triangle: a powerful tool for the prehospital provider. J Paramed Pract. 2011;3(1):20-25. doi:10.12968/jpar.2011.3.1.20
- Strobel AM, Lu LN. The Critically Ill Infant with Congenital Heart Disease. Emerg Med Clin North Am. 2015;33(3):501-518. doi:10.1016/j.emc.2015.04.002
- Brousseau T, Sharieff GQ. Newborn emergencies: The first 30 days of life. Pediatr Clin North Am. 2006;53(1):69-84. doi:10.1016/j.pcl.2005.09.011
- Crossland DS, Furness JC, Abu-Karb M, Sadagopan SN, Wren C. Variability of four limb blood pressure in normal neonates. Arch Dis Child Fetal Neonatal Ed. 2004;89(4). doi:10.1136/adc.2003.034322
- Maher KO, Reed H, Cuadrado A, et al. B-type natriuretic peptide in the emergency diagnosis of critical heart disease in children. Pediatrics. 2008;121(6):e1484-e1488. doi:10.1542/peds.2007-1856
- Arunamata A, Buccola Stauffer KJ o., Punn R, Chan FP, Maeda K, Balasubramanian S. Diagnosis of Anomalous Aortic Origin of the Left Coronary Artery in a Pediatric Patient. World J Pediatr Congenit Heart Surg. 2015;6(3):470-473. doi:10.1177/2150135114558689
- Rodriguez-Gonzalez M, Tirado AM, Hosseinpour R, de Soto JS. Anomalous origin of the left coronary artery from the pulmonary artery: Diagnoses and surgical results in 12 pediatric patients. Texas Hear Inst J. 2015;42(4):350-356. doi:10.14503/THIJ-13-3849
- Ferguson EC, Krishnamurthy R, Oldham SAA. Classic imaging signs of congenital cardiovascular abnormalities. Radiographics. 2007;27(5):1323-1334. doi:10.1148/rg.275065148
- Pershad J, Chin T. Early detection of cardiac disease masquerading as acute bronchospasm: the role of bedside limited echocardiography by the emergency physician. Pediatr Emerg Care. 2003;19(2):466. doi:10.1097/00006565-200304000-00023
- Costello JM, Almodovar MC. Emergency Care for Infants and Children with Acute Cardiac Disease. Clin Pediatr Emerg Med. 2007;8(3):145-155. doi:10.1016/j.cpem.2007.06.005
- Danford DA, Gutgesell HP, McNamara DG. Application of information theory to decision analysis in potentially prostaglandin-responsive neonates. J Am Coll Cardiol. 1986;8(5):1125-1130. doi:10.1016/S0735-1097(86)80391-6