Authors: Ioana Rider, MD (EM Resident Physician – Aventura Hospital & Medical Center); Scarlet Benson, MD (Assistant Clerkship Director, Assistant Clinical Professor – Emergency Medicine Residency – Aventura Hospital & Medical Center) // Reviewed by: Edward Lew, MD (@elewMD); Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit)
Case:
72-year-old male with past medical history of HTN, IDDM, CAD, presents through triage with worsening shortness of breath, orthopnea, and bilateral lower extremity swelling for the past week. He denies fever, cough, chest pain and ROS is otherwise negative. Vitals are as follows: BP 167/78, HR 93, RR 18, O2 Sat 94% on RA. Exam remarkable for rales in all fields and pitting edema to mid-shin in bilateral lower extremities.
Question:
Which heart failure patients need to be admitted, and which patients can be discharged home?
Background:
Patients in acute heart failure (AHF) can experience significant morbidity and mortality and therefore the rapid evaluation, management and proper disposition of AHF is very important to the Emergency Physician. More than 90% of patients in acute heart failure present to the ED (1) and in the U.S. this leads to an estimated 650,000 ED visits annually with 80% of these patients being hospitalized (2). This leads to more than 70 billion dollars spent annually on AHF in U.S. hospitals (3). Despite the frequency of these visits there have been few studies focusing on the disposition of these patients from the ED. A large proportion of these admissions are not necessarily based on clinical status at time of disposition but rather on the uncertain nature of the progression of clinical deterioration in AHF and the patient’s ability to self-manage underlying comorbidities (1, 4, 5).
Less than 10% of AHF patients are severely ill (5). It has been proposed that 50% of AHF patients could be safely discharged from the ED after a period of observation (5). A large barrier to this is the fact that very few EDs have dedicated Observation Units where patients could be observed for up to 23 hours for clinical deterioration (3). In most practice environments, disposition decisions need to be made much more quickly, without the possibility for observation. Part of the challenge in determining a disposition for AHF patients is that there are few risk stratification tools for emergency physicians to use when determining who is safe to be discharged and who needs to be admitted.
New Onset Heart Failure:
One third of acute heart failure patients have no prior history of HF (6). Patients who are presenting with new onset acute heart failure should be admitted regardless of their clinical status in the ED as they need an expanded workup that will give insight into their long-term prognosis (7). All these patients should have a formal echocardiogram to assess for left ventricular dysfunction and any underlying abnormalities such as valvular dysfunction (aortic stenosis, mitral regurgitation, etc.) (7). Cases with reduced EF and severe aortic stenosis will need a stress test to evaluate for underlying inducible ischemia (7). Patients presenting with chest pain in addition to new onset heart failure, if stable, benefit from urgent coronary angiography (7). New onset AHF patients also need to be monitored for arrhythmias as sudden cardiac death is a significant occurrence even in patients with mild symptoms (7). If these patients are found to have reduced EF and arrythmia they will need AICD placement (7). Furthermore, these patients need to be started on the recommended medication regiment for HF management such as ACE inhibitors, beta blockers, and diuretics and to establish care with cardiology (7).
Pearl:
- Admit new onset HF patients for further evaluation.
Risk Stratification:
Patients requiring NIPPV, continuous infusion of diuretics or vasodilators, inotropes or vasopressors require admission due to high 7-day mortality (up to 3.9% in this high-risk group) (2, 4, 5, 8, 9).
High risk factors are hypotension with SBP<90, high BNP >1000, elevated troponin, elevated BUN/Cr, hyponatremia (2). The absence of these high-risk factors does not, however mean the patient has no risk, but places the patient in a low-risk category that may be better suited for discharge from the ED (2, 9, 10).
A few risk stratification tools have been developed for patients in AHF that EPs can employ to determine disposition: the Ottawa Heart Failure Risk Score, the Emergency Heart Failure Mortality Risk Grade (EHFMRG) and the Multiple Estimation of Risk Based on Spanish Emergency Department Score (MEESSI) (1,11).
Pearls:
- High risk features in AHF are hypotension, elevated BNP, troponin, Cr, and hyponatremia.
- Patients who require continuous infusions, NIPPV, or are initially treatment-resistant generally require ICU admission.
The Ottawa Heart Failure Risk Score:
OHFRS has been validated to evaluate the risk of 14- and 30-day adverse events by Stiell et al and was developed using a prospective observational cohort study conducted across 6 tertiary care EDs in Canada involving 1,100 patients who presented with shortness of breath in the setting of acute heart failure (2, 12). Adverse events were defined as death within 30 days, ICU admission, NIPPV, intubation, MI, or 14-day readmission (12). The study found that 15.5% of patients experienced adverse events and a concerning proportion of those were in patients who had been discharged home from the ED (12). In addition, 39% of the deaths occurred in patients who had been discharged home (12).
Based on this study, the authors developed a score using the criteria shown below. Patients with a score less than 1 can be considered appropriate for discharge. Admitting patients with OHFRS of 1 significantly increased the sensitivity for adverse events compared to actual practice (71.8% vs 91.8%) (12). Using a score of 2 or greater to admit had similar sensitivity to the current practice amongst the EPs in the study and also resulted in significantly fewer admissions (12).

One weakness of the OHFRS is that despite its objective scoring system, there is still some subjectivity involved on the part of the emergency physician when choosing the 1-point versus the 2-point threshold for admission. Patients with OHFRS of 1 are more challenging in terms of disposition, as their risk of adverse events is higher but they can also be discharged home using the OHFRS (12). For example, not all AHF patients with prior history of TIA require admission if they have no points assigned for other criteria in the score (such as NT-ProBNP>5000). However, if a patient scores 1 point for O2 sat <90% on arrival, an EP should strongly consider admitting these patients more as it is a real-time measurement of how the patient is doing and serves as a better predictor of their immediate risk (12). Another weakness of this tool is the walk test performed after ED treatment. The Stiell study found that a common concern among physicians in the study was forgetting to arrange for the walk test post intervention (12).
Pearls:
- Patients with OHFRS less than 1 can be discharged.
- Patients with OHFRS above 1 or 2 require admission (at the EP’s discretion regarding which score to use based on comorbidities and available follow-up).
- If a patient has OHFRS of 1 and the point is for O2 sat <90% on arrival, admit.
The Emergency Heart Failure Mortality Risk Grade (EHFMRG):
The EHFMRG has been developed and validated retrospectively in a study of 12,591 Canadian patients presenting to the ED with acute heart failure and again in a follow-up Canadian retrospective cohort study across 9 hospital EDs involving 6708 patients (13). This risk-stratification tool calculates a percentage for 7-day mortality risk (ranging from very low to very high) based on scoring for the following factors (2, 13, 14):
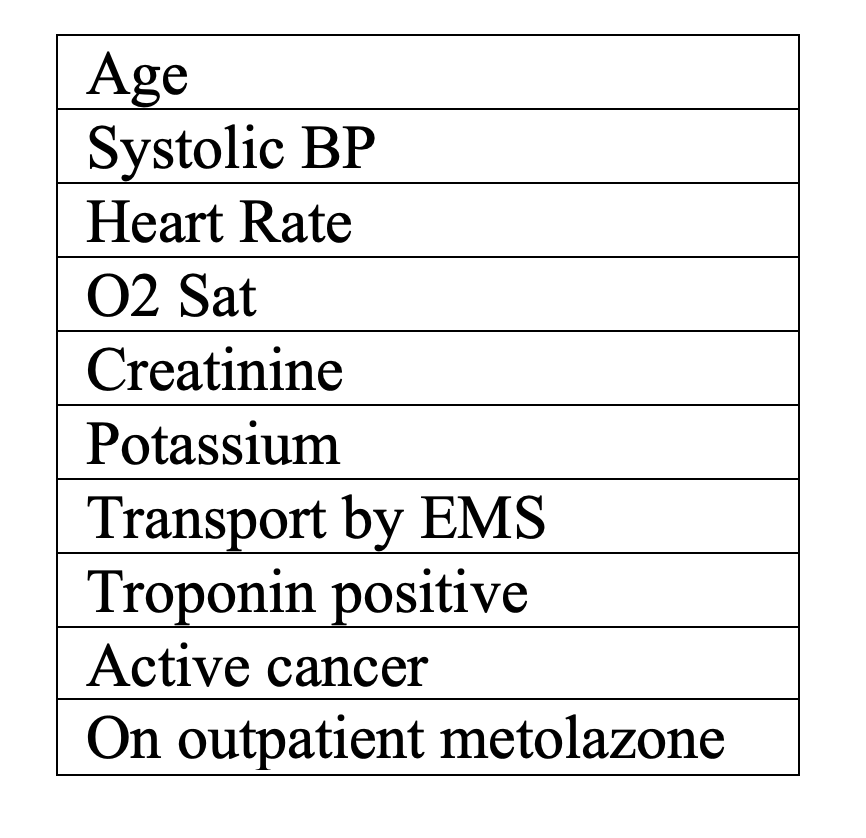
The calculator can be found on MDCalc (https://www.mdcalc.com/emergency-heart-failure-mortality-risk-grade-ehmrg) and at https://ehmrg.ices.on.ca/#/ (2).
The 7-day mortality risk across the 5 categories is as follows: 0.0%, 0.8%, 1.6%, 4.0%, 4.2%, and 12.0% for very low, low, intermediate, high, and very high risk, respectively (13).
A major disadvantage of this risk stratification tool is the fact that it awards 60 points to the overall risk calculation for EMS transport to the ED. There are many reasons why a patient would call EMS, ranging from severity of symptoms and fear of impending decompensation, to no available method of transportation to the hospital. A patient could potentially increase their calculated mortality risk significantly, simply because EMS brought that patient to the ED. Another disadvantage of this score is that there are 5 categories of risk, which makes determining disposition slightly more complicated.
A more simplified and straight-forward algorithm for following the EHFMRG has been proposed (15):
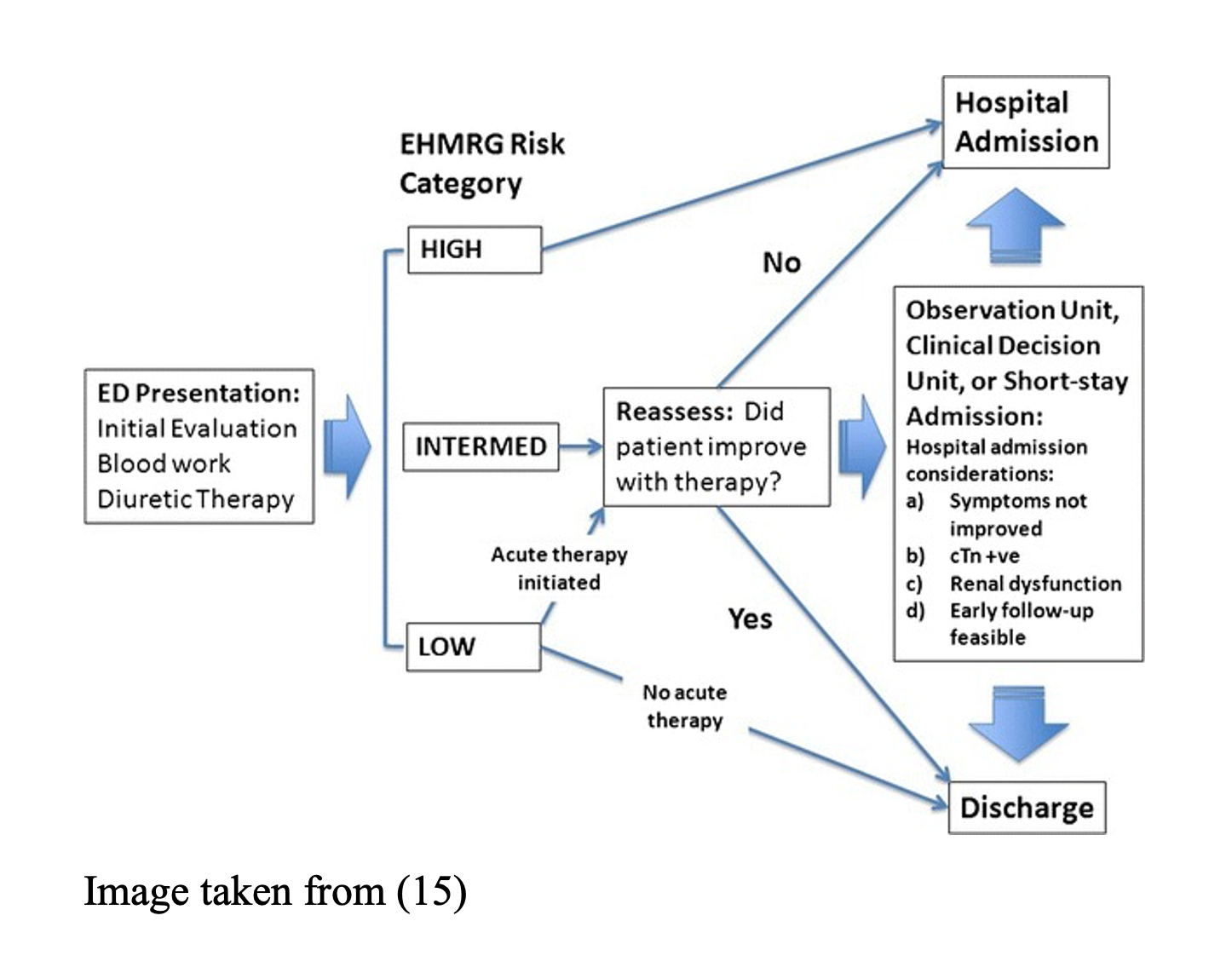
Pearl:
- EHFMRG is an internally and externally validated tool to predict a percentage risk in 7-day mortality for AHF patients.
The Multiple Estimation of Risk Based on Spanish Emergency Department Score (MEESSI):
The MEESSI score was successfully internally validated to predict 30-day mortality in ED AHF patients in a cohort of 4,711 Spanish ED patients and was also externally validated in a Swiss prospective cohort study of 1,572 patients with AHF (1, 16). The MEESSI score gives the following distribution of patients according to risk categories: 35.5% low risk, 42.9% intermediate risk, 11.3% high risk and 10.3% very high risk, with a 30-day mortality of 2.0%, 7.8%, 17.9%, and 41.4%, respectively (1).
This risk stratification tool can be used to predict 30-day mortality in ED AHF patients based on the following variables:
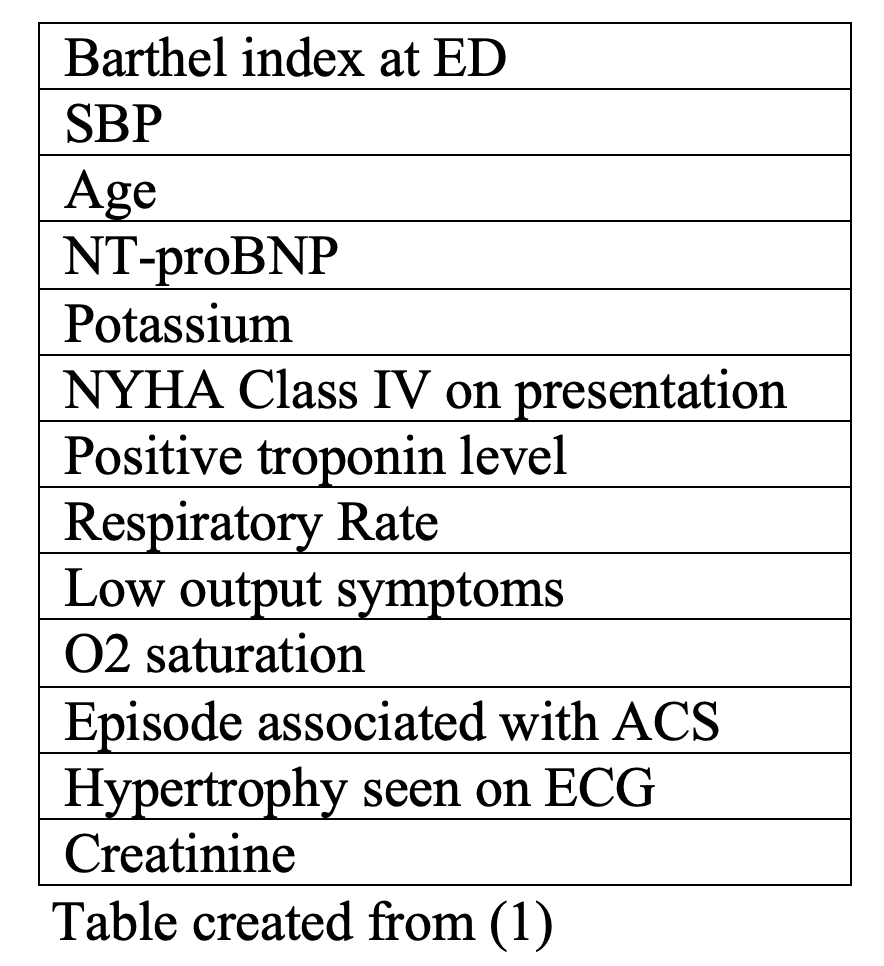
The calculator can be found at: http://meessi-ahf.risk.score-calculator-ica-semes.portalsemes.org/ (1).
This score has several disadvantages. Although it is important to be able to determine a 30-day risk of mortality for AHF patients, it would be more useful to an EP to determine a more immediate risk, such a 7-day or 14-day risk level. In addition, this risk calculator tool uses some variables that may be largely unknown or that take a significantly longer time to determine, such as the Barthel index and the NYHA class of the patient.
Pearls:
- Low-risk MEESSI patients carry a <2% mortality and may be appropriate to discharge home from the ED (1).
- Some patients in AHF will be in the MEESSI higher risk categories with a significant increase in 30-day mortality compared to the low-risk groups.
Data on Outcomes Between Patients Discharged from ED vs Hospitalized Patients:
Very little data exist on differences in outcome for AHF patients discharged home versus those who were hospitalized due to many confounding factors between the groups. One study by Brar et al compared death at 30 days, hospitalization, and ED revisit among the two groups and found that these outcomes were higher for patients who were discharged home from the ED (9). Return visits to the ED occurred in 20-25% of discharged patients from the ED and eventual rehospitalization at 30 days was 75% (9). Another study by Levy et al showed that 7- and 30-day mortality was similar among those discharged from the ED versus those who were hospitalized although the discharged group overall fared worse in terms of morbidity (9).
Observation Units:
Appropriate Observation Unit candidates for EDs with such capabilities are patients with the following: SBP>120 mmHg, RR <32, BUN<40 mg/dl, Cr <3 mg/dl, no ischemic changes on EKG and no elevation in troponin (3). Patients in an OBS unit should undergo cardiology consult and planning for expedited follow-up.
Pearl:
- Observation units are appropriate only for hemodynamically stable patients who are responding to diuresis, without significant elevations in BUN/Cr with no evidence of ischemia on ECG or labs.
Case Conclusion:
The patient in our case remained hemodynamically stable throughout the ED visit. The workup revealed the following pertinent findings: EKG with LVF, no arrhythmia, CXR showing interstitial edema and small bilateral pleural effusions, troponin 0.099 ng/mL, NT-ProBNP 13,500 (pg/mL), K 4.6 mEq/L, Cr 2.1 mg/dL, BUN 24 mg/dL. He was given loop diuretics in the ED as treatment for pulmonary edema due to new onset heart failure.
The patients risk stratification calculations are as follows:
OHFRS – 1 point – medium risk
EHMRG – 38.2 points (2%) – intermediate risk of 7-day mortality
MEESSI – 3.344% risk of 30-day mortality – low risk (2nd quartile)
Based on the risk stratification calculated above with the OHFRS and EHMRG scores the recommended disposition for this patient is admission. Based on the MEESSI calculation, the patient could be discharged home. However, given the patient has been diagnosed with new onset HF with a positive troponin, he would benefit for admission for further investigations as discussed above. This patient was admitted with continued diuretics; his troponin trended down, and he was discharged the next day at his request to follow-up with cardiology for outpatient echocardiogram.
Which Risk Stratification Tool We Recommend:
After reviewing the literature, the authors of this article prefer the EHFRMG risk stratification tool. This is due to several reasons: 1) it gives a 7-day mortality risk, thus helping the provider make decisions with a more immediate short-term outcome as a goal; 2) the variables involved in the calculation are more straight-forward and the calculator is easy to use and interpret; 3) the tool was validated on a large number of patients across several studies. We have not used any validation tool in our ED to date, mainly because these risk stratification tools are not widely used in the U.S. We do suggest the incorporation of these tools as they would benefit disposition decisions in the ED in the U.S. as well.
Editor Note: As you can see, there are a variety of tools available, but they should not replace clinical judgement. They only function as a tool to assist you. It is also helpful to have an agreed upon tool among the emergency medicine, internal medicine, and cardiology clinicians if possible. A walk of life test can make a big difference in your disposition decision, and respect abnormal lab values like elevated Cr, BUN, and troponin in these patients.
Conclusion:
Acute heart failure is a presentation commonly seen in the ED that carries high morbidity and mortality. The emergency physician’s role involves rapid identification, management and disposition of these patients based on clinical presentation, response to therapy and risk of adverse events. Several risk stratification tools can be used to risk stratify patients. A wide majority of AHF patients require admission and all require close follow-up with cardiology and primary care.
Pearls:
- High risk features in AHF are hypotension, elevated BNP, troponin, Cr, and hyponatremia.
- Patients who require continuous infusions, NIPPV, or are initially treatment-resistant generally require ICU admission.
- New onset HF patients should be admitted for a broader workup.
- Patients with OHFRS less than 1 can be discharged.
- Patients with OHFRS above 1 or 2 require admission (at the EP’s discretion regarding which score to use based on comorbidities and follow-up).
- Patients with OHFRS of 1 due to O2 saturation <90% on arrival to ED should be admitted.
- EHFMRG is an externally validated tool to predict a % risk in 7-day mortality for AHF patients.
- Low-risk MEESSI patients carry a <2% mortality and may be appropriate to discharge home from the ED.
- Some patients in AHF will be in the MEESSI higher risk categories with a significant increase in 30-day mortality compared to the low-risk groups.
- Observation units are appropriate for hemodynamically stable patients with good response to therapy who are in very low-risk categories and are reliable to follow-up.
References:
- Miró, Ò, Rossello, X., Gil, V., Martín-Sánchez, F. J., Llorens, P., Herrero-Puente, P., . . . Ferrer, E. S. (2019, August). Analysis of How Emergency Physicians’ Decisions to Hospitalize or Discharge Patients With Acute Heart Failure Match the Clinical Risk Categories of the MEESSI-AHF Scale. Annals of Emergency Medicine, 74(2), 204-215. doi:10.1016/j.annemergmed.2019.03.010
- Long, Brit, Koyfman, A., & Gottlieb, M. (2018). Management of heart failure in the emergency department setting: An evidence-based review of the literature. The Journal of Emergency Medicine, 55(5), 635-646. doi:10.1016/j.jemermed.2018.08.002
- Collins, Storrow, A. B., Albert, N. M., Butler, J., Ezekowitz, J., Felker, G. M., . . . Lenihan, D. J. (2015). Early management of patients with acute heart failure: State of the art and future directions. a consensus document from the Society for Academic Emergency Medicine/Heart Failure Society of America Acute Heart Failure Working Group. Journal of Cardiac Failure, 21(1), 27-43. doi:10.1016/Aj.cardfail.2014.07.003
- Lee, D. S., Lee, J. S., Schull, M. J., Borgundvaag, B., Edmonds, M. L., Ivankovic, M., . . . Tu, J. V. (2019). Prospective validation of the emergency heart failure mortality risk grade for acute heart failure. Circulation, 139(9), 1146-1156. doi:10.1161/circulationaha.118.035509
- Horton, C. F., & Collins, S. P. (2013). The role of the emergency department in the patient with acute heart failure. Current Cardiology Reports, 15(6). doi:10.1007/s11886-013-0365-3
- Nieminen, M. S., Brutsaert, D., Dickstein, K., Drexler, H., Follath, F., Harjola, V., . . . Tavazzi, L. (2006). EuroHeart Failure Survey II (EHFS II): A survey on hospitalized acute heart failure patients: Description of population. European Heart Journal, 27(22), 2725-2736. doi:10.1093/eurheartj/ehl193
- Mcmurray, J. J., Adamopoulos, S., Anker, S. D., Auricchio, A., Bohm, M., Dickstein, K., . . . Ponikowski, P. (2012). ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. European Heart Journal, 33(14), 1787-1847. doi:10.1093/eurheartj/ehs104
- Lee, D. S., & Ezekowitz, J. A. (2014). Risk Stratification in Acute Heart Failure. Canadian Journal of Cardiology. doi:10.1007/springerreference_225162
- Miró, Levy, P. D., Möckel, M., Pang, P. S., Lambrinou, E., Bueno, H., . . . Collins, S. P. (2017). Disposition of emergency department patients diagnosed with acute heart failure. European Journal of Emergency Medicine,24(1), 2-12. doi:10.1097/mej.0000000000000411
- Blood, A. J., Fraiche, A. M., & Eapen, Z. J. (2017). Is an admission for decompensated heart failure inevitable? Progress in Cardiovascular Diseases, 60(2), 171-177. doi:10.1016/j.pcad.2017.07.003
- Russell, F. M., & Pang, P. S. (2019). Acute heart failure risk stratification in the emergency department: Are we there yet? Revista Española De Cardiología (English Edition), 72(3), 190-191. doi:10.1016/j.rec.2018.09.008
- Stiell G., Perry, J. J., Clement, C. M., Brison, R. J., Rowe, B. H., Aaron, S. D., . . . Wells, G. A. (2017). Prospective and explicit clinical validation of the OTTAWA heart failure Risk scale, with and without use of quantitative NT ‐pro BNP. Academic Emergency Medicine, 24(3), 316-327. doi:10.1111/acem.13141
- Lee S., Lee, J. S., Schull, M. J., Borgundvaag, B., Edmonds, M. L., Ivankovic, M., . . . Tu, J. V. (2019). Prospective validation of the emergency heart failure mortality risk grade for acute heart failure. Circulation, 139(9), 1146-1156. doi:10.1161/circulationaha.118.035509
- Sepehrvand, Youngson, E., Bakal, J. A., Mcalister, F. A., Rowe, B. H., & Ezekowitz, J. A. (2019). External validation and refinement of emergency heart failure mortality risk grade risk model in patients with heart failure in the emergency department. CJC Open, 1(3), 123-130. doi:10.1016/j.cjco.2019.03.003
- Ho C., Schull, M. J., & Lee, D. S. (2012). The challenge of heart failure discharge from the emergency department. Current Heart Failure Reports, 9(3), 252-259. doi:10.1007/s11897-012-0100-1
- Wussler, D., Kozhuharov, N., Sabti, Z., et al. (2019). External Validation of the MEESSI Acute Heart Failure Risk Score: A Cohort Study. Annals of internal medicine, 170(4), 248–256. https://doi.org/10.7326/M18-1967
This research was supported (in whole or in part) by HCA Healthcare and/or an HCA Healthcare affiliated entity. The views expressed in this publication represent those of the author(s) and do not necessarily represent the official views of HCA Healthcare or any of its affiliated entities.






