Authors: Alexander Garrett, MD (@alexgarrettMD, EM Resident Physician, Harbor-UCLA Medical Center) and Manpreet Singh, MD (@MprizzleER, EM Attending Physician, Harbor-UCLA Medical Center) // Reviewed by: Michael Yoo, MD (EM Physician; San Antonio, TX); Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit)
Case:
A 66-year-old female with atrial fibrillation on warfarin presents with hematochezia. She is passing bright red blood intermittently but is not actively bleeding. She has no vomiting, hematemesis, melena, or abdominal pain. Her vitals are normal and your abdominal exam is benign. Upon digital rectal exam, you find trace bright red blood. Her hemoglobin is 8 g/dL, and her INR is therapeutic at 2.4.
Do you consider imaging? Will you transfuse her? Should you stop, or even reverse, her warfarin? What is her disposition?
Background:
Lower GI bleeding (LGIB) is traditionally defined as bleeding from a GI source distal to the ligament of Treitz and usually presents as hematochezia.1 The most common etiology of lower GI bleeds is diverticular disease, followed by hemorrhoids and ischemic colitis (see Table 1).2,3 Bleeding from a lower GI source, which accounts for 20% of GI bleeding, is less common than upper GI bleeds (UGIB) and carries a much lower mortality rate (about 4%).2-5 Clinically significant lower GI bleeds are more common in older adults, with the incidence increasing 200-fold between the 2nd and 9th decades of life.2,6 This is likely due to hemorrhoids being a common cause of LGIB presentations in younger patients and diverticular disease more commonly affecting older patients.2,7,8 Notably, about 15% of UGIBs present with hematochezia, and lower GI bleeds may present with melena (usually from a right-sided bleed with low stool transit time).3,5
History:
Most lower GI bleeds will resolve spontaneously and can be further evaluated and managed on an outpatient basis.3-5History, therefore, should specifically focus on identifying hematochezia that may represent a brisk UGIB and risk stratifying patients with true lower GI sources. As always, ensure patient stabilization prior to detailed history-taking in critical patients. We highlight key components of the history below:
- History of present illness
- Severity, timing, and frequency
- Bleeding from hemorrhoids and infectious colitis will most likely be seen with bowel movements, while other causes of bleeding may cause passage of frank blood without stool.
- Appearance and color
- Patient-reported melena is associated with a likelihood ratio of 5.1-5.9 for an UGIB.9
- The presence of clots is less likely to be from an upper GI source (likelihood ratio 0.05).9
- Associated symptoms
- A history of constipation raises concern for hemorrhoids and diverticulosis.
- Associated abdominal pain is seen with ischemic colitis, infectious colitis, and inflammatory bowel disease. Painless bleeding is more typical in diverticular bleeding and arteriovenous malformations.5,10
- Medications
- Medications that may increase risk for bleeding include non-steroidal anti-inflammatory drugs, aspirin and other anti-platelet agents, and other anticoagulant medications. Consider reversal of agents if the patient is unstable.11-16
- Past medical history and other risk factors
- Patients with alcohol abuse or cirrhosis may present with an upper GI source with rapid transit.
- Atrial fibrillation, coagulopathy, and atherosclerotic disease can predispose to ischemic colitis.17,18
- Past surgical history
- A history of abdominal aortic aneurysm repair should raise suspicion for an aortoenteric fistula, an uncommon but incredibly dangerous etiology of hematochezia, which requires immediate surgical consultation.
- Severity, timing, and frequency
Exam:
The priority in the physical exam is to establish stability. Any unstable patient should be evaluated by the ABCs (airway, breathing, circulation). After stabilizing a patient, or in a patient who is initially stable, focus on these key features:
- Vitals: consider using the shock index to assess stability: heart rate divided by the systolic blood pressure, e.g., a patient with a HR of 120 and a SBP of 80 has a shock index of 1.5. A shock index >1 correlates with occult bleeding, need for blood products, and/or need for vasopressors.4
- Skin
- Pale, cool, and/or clammy
- Jaundice or other secondary signs of cirrhosis (spider angioma, palmar erythema) should raise suspicion for a concurrent or primary UGIB.
- Abdomen
- Tenderness can suggest ischemic colitis. If you are considering ischemic colitis and the patient has peritonitis or signs of instability, early surgical consultation should be obtained.
- Rectum
- Perform an external exam to assess for fissures and hemorrhoids.
- Digital rectal exam to check for color of stool and presence of melena
- Stool guaiac testing, once commonplace to evaluate LGIB, has become controversial.19 The test was designed to assess for occult blood in the stool, and it requires strict dietary and medication restrictions prior to testing to achieve the best specificity and sensitivity.20 Subsequently, use in the ED can lead to a higher rate of false-positives and false-negatives. Further, the test has a poor sensitivity for lower GI lesions (24.3%).21 Currently, the literature does not support the routine use of stool guaiac testing in the ED.19
- External hemorrhoids and fissures are easily identifiable with a visual exam. However, in the absence of external findings and with a high suspicion for internal hemorrhoids or fissures, anoscopy can be used to confirm the diagnosis.7,22
Evaluation:
- To reiterate, any unstable patient, especially in one with suspected bleeding, focus on stabilization. This includes obtaining multiple large bore IVs and consideration for empiric blood transfusion.
- Labs
- CBC, type and screen (type and cross if unstable), renal and liver function panels, electrolytes, and coagulation panel
- ECG
- Assess for ischemia from acute blood loss in patients at risk (older than 40 or history of CAD).
- Risk Stratification
- A risk stratification tool may be used to guide further evaluation.4,5 The most recent British Society of Gastroenterology guidelines recommend calculating the shock index, as described above.4 Stable patients (shock index <1) can be further risk stratified using the Oakland score, which relies on history, exam, and hemoglobin level (of note: Dr. Oakland is the primary author of the British guidelines), Table 2.4,13

-
- Using a cutoff of ≤8 identifies patients safe for discharge with a sensitivity of 98.4%. Extending the cutoff to ≤10 carries a sensitivity of 96%.13
- As always, decision making and scoring tools should never replace or override clinical judgement.
- Imaging
- Younger patients with obvious anorectal etiologies of LGIB and who are otherwise low risk (shock index <1, Oakland score ≤8-10) will likely not require imaging.4,8
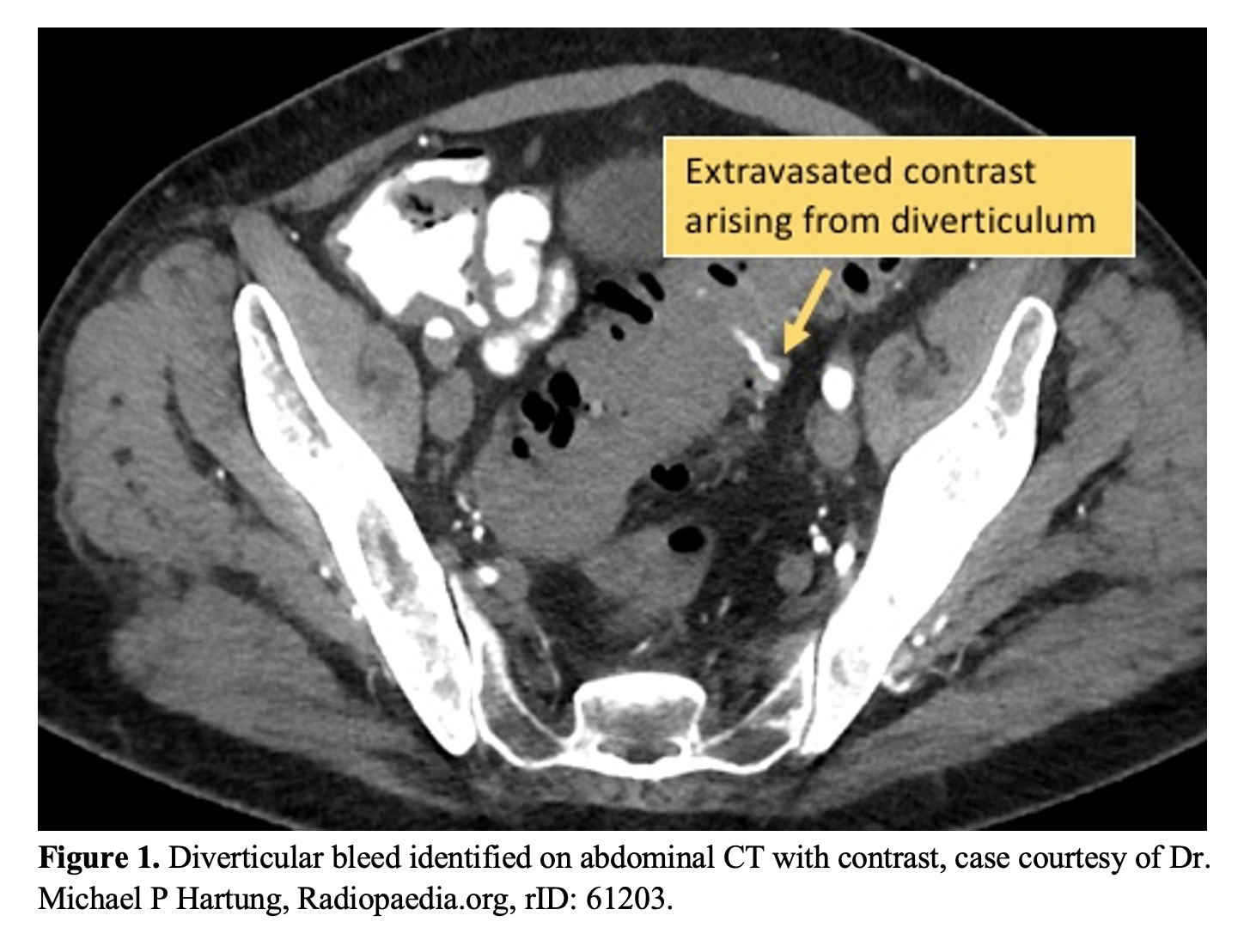
- Abdominal CT may show signs of ischemic colitis, especially later in the disease presentation (Figure 1).17,23,24 CT with contrast may be able to locate areas of diverticular bleeding, with a sensitivity of only 57.6%, but specificity of 91.2%. The sensitivity is increased if the scan is obtained within 4 hours of the last bleed, up to 64.7%.25
-
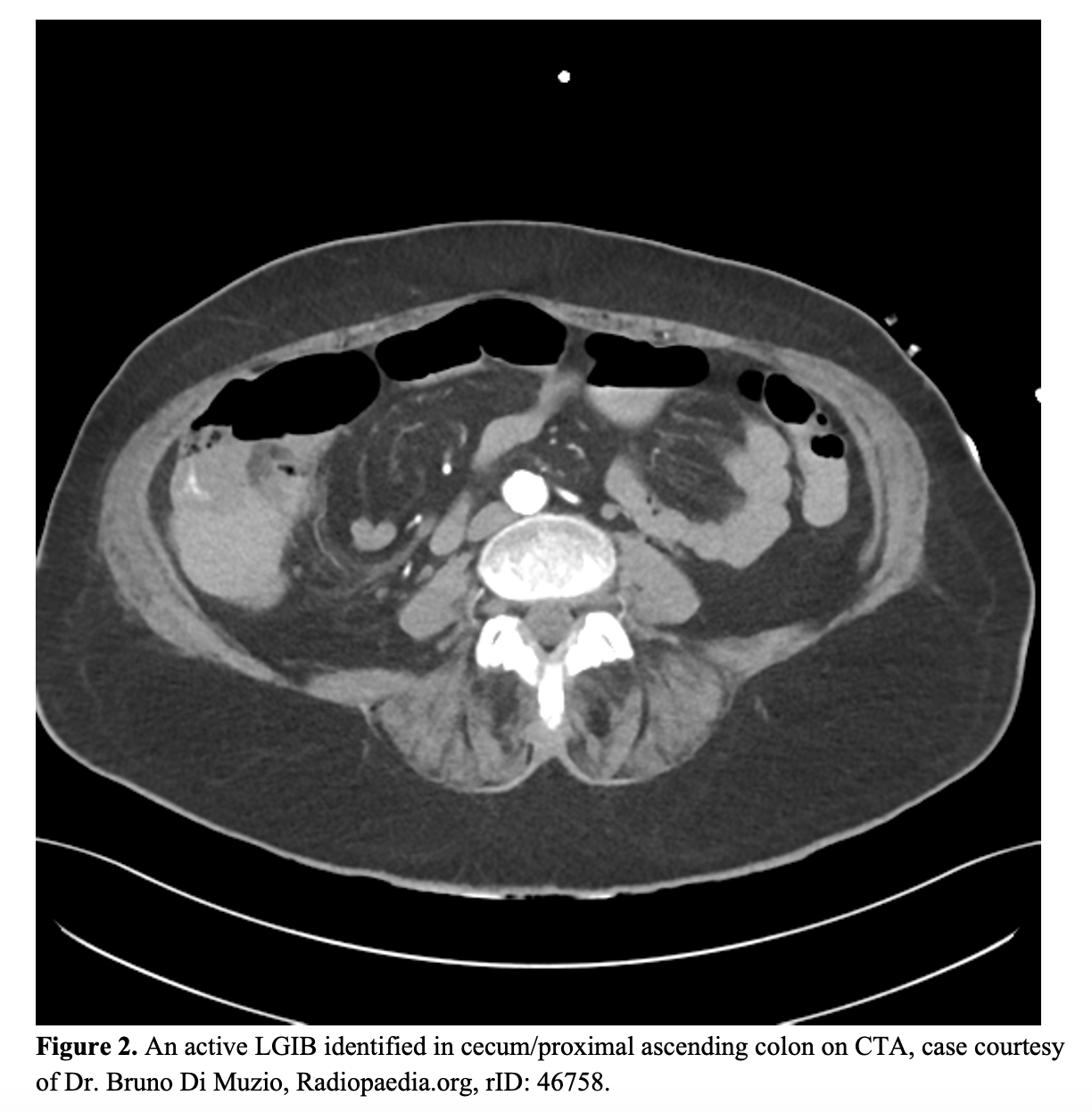
- CT angiography (CTA) is emerging as the imaging tool of choice for GI bleeds, but recommendations for its use differ between organizations4,5,24,26 CTA relies on an active bleed for visualization and carries a sensitivity of 85% and specificity of 92% for moderate to severe acute bleeds (Figure 2).24 The most recent American College of Gastroenterology (ACG) guidelines give preference to direct visualization with endoscopy and/or colonoscopy, with CTA reserved for patients with active bleeding who have negative esophagogastroduodenoscopies (EGDs) and are “unable to tolerate bowel prep;” i.e., it most likely would not be performed in the ED.5 The 2019 guidelines from the British Society of Gastroenterology stress the importance of CTA for unstable patients in finding the source of bleeds and providing targets for interventional radiology (IR) embolization procedures. If CTA is negative and the patient continues to bleed, then an EGD is recommended.4 An unstable patient in these guidelines is defined as one with a shock index > 1. (For more on the 2019 British guidelines see the emdocs.net update)
- Nasogastric tube (NG tube) lavage is occasionally used to evaluate for an UGIB source in unstable patients with hematochezia, but its use is controversial. The ACG guidelines state that patients with a high enough suspicion for an upper GI source should proceed to EGD, but an NG tube can be considered if there is “moderate” suspicion for an upper GI source.5 The British guidelines do not recommend routine NG tube use as it does not reliably affect outcomes and is not a benign procedure.4 NG lavage is 91% specific for identifying UGIB but not sensitive (42%) and has not been shown to reliably affect clinical outcomes.27-29
Treatment:
- Bottom line: if there is a high index of suspicion for an UGIB, treat as such.
- Blood transfusion
- Threshold of hemoglobin <7 g/dL, target 7-9 g/dL4,5,30,31
- In patients with cardiovascular disease, the threshold is commonly 8 g/dL with goal 10.4,5,31,32
- Consider transfusion regardless of hemoglobin level in unstable patients with massive bleeds.
- For non-anorectal sources, stop anticoagulants (warfarin, direct/novel oral anticoagulants) at presentation, and consider reversal if the patient is unstable.4,5 Aspirin should not be discontinued if being used for secondary prevention in patients with CAD, though briefly pausing aspirin may be considered if its indication is for primary prevention.4,5,33,34 Resuming anticoagulants may be safe at 7 days, though this decision will likely be made by a multidisciplinary group of specialists and is out of the scope for the ED physician.4,5,35,36
- Tranexamic acid is no longer recommended following the results of the HALT-IT trial.37
- Patients with self-limiting, minor bleeds without risk factors can be discharged with outpatient followup.4,5 These patients can be identified by an Oakland score ≤8-10, depending on the individual clinician’s risk threshold.4,13
- Active bleeding or bleeding with risk factors warrants GI consultation +/- surgical consultation. These patients should be admitted for observation and likely inpatient endoscopy and/or colonoscopy.4,5,13
- Unstable patients (shock index >1) should be resuscitated and have emergent GI and IR consultation. Effective colonoscopy requires adequate bowel preparation, which is not possible in the acutely unstable patient.4,5Therefore, IR may be required to perform an angiogram with embolization to resolve bleeding. CTA is recommended prior to angiogram to help solidify the diagnosis and identify embolization targets for IR.4
- Previously, GI specialists aimed to perform colonoscopies for hospitalized patients with LGIB within 24 hours, as recommenced in the ACG 2016 guidelines.5 However, newer literature comparing “early” colonoscopy (within 24 hours of presentation) versus delayed or elective scoping (within 1-3 days) shows that clinical outcomes are not different.38-40 The British guidelines subsequently state that hospitalized patients should be placed on a next-available list for colonoscopy, but the scope can be delayed to ensure clinical stabilization and adequate bowel preparation.4
Take Home Points/Pearls/Pitfalls:
- Stability is key. Most lower GI bleeds will self-resolve and tend to be less severe than upper GI bleeds. Consider UGIB with rapid transit if the patient is unstable and/or has upper GI risk factors (e.g., cirrhosis).
- CTA is the preferred imaging modality for active lower GI bleeds, with a high sensitivity and specificity for identifying bleeding sources in active If the bleeding has stopped, the sensitivity and specificity is lower.
- Patients on anticoagulation with active bleeds should pause their anticoagulation. Unstable patients should be reversed, if possible.
- Always consider aortoenteric fistula in your differential. Ask the patient about their surgical history (specifically regarding abdominal aortic aneurysm repair), as this is a life-threatening diagnosis.
References/Further Reading:
- Walls RM, Hockberger RS, Gausche-Hill M, Meguerdichian DA, Goralnick E. Gastrointestinal Bleeding. In: Rosen’s Emergency Medicine: Concepts and Clinical Practice. Vol 1. 9th ed. Philadelphia, PA, PA: Elsevier; 2018:242-248.
- Longstreth GF. Epidemiology and outcome of patients hospitalized with acute lower gastrointestinal hemorrhage: a population-based study. Am J Gastroenterol. 1997;92(3):419-424.
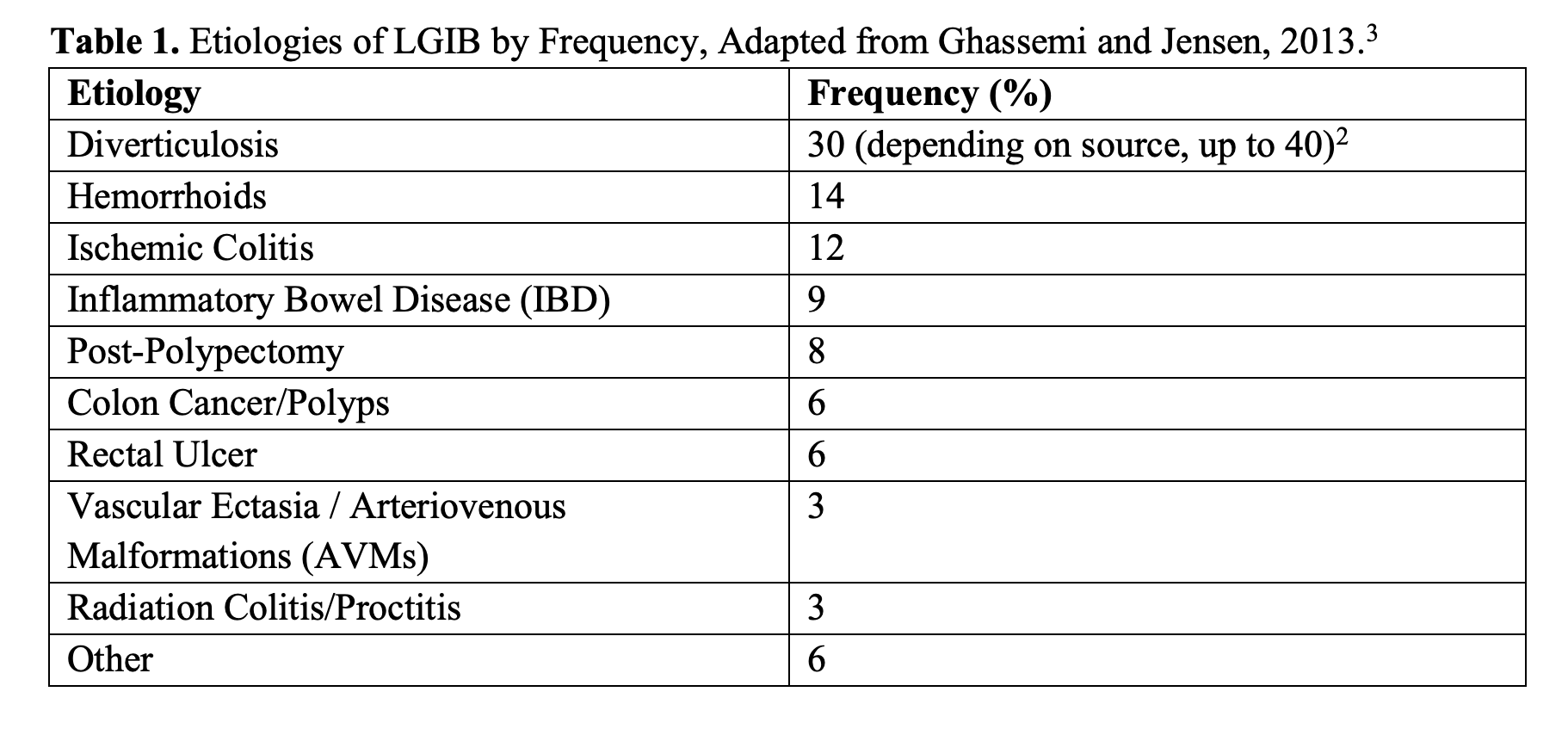
- Ghassemi KA, Jensen DM. Lower GI bleeding: epidemiology and management. Curr Gastroenterol Rep. 2013;15(7):333.
- Oakland K, Chadwick G, East JE, et al. Diagnosis and management of acute lower gastrointestinal bleeding: guidelines from the British Society of Gastroenterology. Gut 2019;68:776-789.
- Strate LL, Gralnek IM. ACG Clinical Guideline: Management of patients with acute lower gastrointestinal bleeding. Am J Gastroenterol. 2016;111(4):459-474.
- Vajravelu RK, Mamtani R, Scott FI, Waxman A, Lewis JD. Incidence, risk factors, and clinical effects of recurrent diverticular hemorrhage: a large cohort study. Gastroenterology. 2018;155(5):1416-1427.
- Mott T, Latimer K, Edwards C. Hemorrhoids: diagnosis and treatment options. AFP. 2018;97(3):172-179.
- Hawks M, Svarverud JE. Acute lower gastrointestinal bleeding: evaluation and management. AFP. 2020;101(4):206-212.
- Srygley FD, Gerardo CJ, Tran T, Fisher DA. Does this patient have a severe upper gastrointestinal bleed? JAMA. 2012;307(10):1072.
- Becq A, Rahmi G, Perrod G, Cellier C. Hemorrhagic angiodysplasia of the digestive tract: pathogenesis, diagnosis, and management. Gastrointestinal Endoscopy. 2017;86(5):792-806.
- Abraham NS, Hartman C, Richardson P, Castillo D, Street RL, Naik AD. Risk of lower and upper gastrointestinal bleeding, transfusions, and hospitalizations with complex antithrombotic therapy in elderly patients. Circulation. 2013;128(17):1869-1877.
- Strate LL, Liu YL, Huang ES, Giovannucci EL, Chan AT. Use of aspirin or nonsteroidal anti-inflammatory drugs increases risk for diverticulitis and diverticular bleeding. Gastroenterology. 2011;140(5):1427-1433.
- Oakland K, Kothiwale S, Forehand T, et al. External validation of the Oakland Score to assess safe hospital discharge among adult patients with acute lower gastrointestinal bleeding in the us. JAMA Netw Open. 2020;3(7):e209630.
- Nagata N, Niikura R, Aoki T, et al. Lower GI bleeding risk of nonsteroidal anti-inflammatory drugs and antiplatelet drug use alone and the effect of combined therapy. Gastrointestinal Endoscopy. 2014;80(6):1124-1131.
- Miller CS, Dorreen A, Martel M, Huynh T, Barkun AN. Risk of gastrointestinal bleeding in patients taking non–vitamin k antagonist oral anticoagulants: a systematic review and meta-analysis. Clinical Gastroenterology and Hepatology. 2017;15(11):1674-1683.e3.
- Abraham NS, Noseworthy PA, Yao X, Sangaralingham LR, Shah ND. Gastrointestinal safety of direct oral anticoagulants: a large population-based study. Gastroenterology. 2017;152(5):1014-1022.e1.
- FitzGerald J, Hernandez III L. Ischemic colitis. Clinics in Colon and Rectal Surgery. 2015;28(02):093-098.
- Brandt LJ, Feuerstadt P, Blaszka MC. Anatomic patterns, patient characteristics, and clinical outcomes in ischemic colitis: a study of 313 cases supported by histology. American Journal of Gastroenterology. 2010;105(10):2245-2252.
- Drescher MJ, Stapleton S, Britstone Z, Fried J, Smally AJ. A call for a reconsideration of the use of fecal occult blood testing in emergency medicine. The Journal of Emergency Medicine. 2020;58(1):54-58.
- Kaur K, Adamski JJ. Fecal occult blood test. In: StatPearls. StatPearls Publishing; 2021.
- Chiang T-H, Lee Y-C, Tu C-H, Chiu H-M, Wu M-S. Performance of the immunochemical fecal occult blood test in predicting lesions in the lower gastrointestinal tract. Canadian Medical Association Journal. 2011;183(13):1474-1481.
- Davis BR, Lee-Kong SA, Migaly J, Feingold DL, Steele SR. The American Society of colon and rectal surgeons clinical practice guidelines for the management of hemorrhoids. Diseases of the Colon & Rectum. 2018;61(3):284-292.
- Cruz C, Abujudeh HH, Nazarian RM, Thrall JH. Ischemic colitis: spectrum of CT findings, sites of involvement and severity. Emerg Radiol. 2015;22(4):357-365.
- Wells ML, Hansel SL, Bruining DH, et al. Ct for evaluation of acute gastrointestinal bleeding. RadioGraphics. 2018;38(4):1089-1107.
- Umezawa S, Nagata N, Arimoto J, et al. Contrast-enhanced CT for colonic diverticular bleeding before colonoscopy: a prospective multicenter study. Radiology. 2018;288(3):755-761.
- Jacovides CL, Nadolski G, Allen SR, et al. Arteriography for lower gastrointestinal hemorrhage: role of preceding abdominal computed tomographic angiogram in diagnosis and localization. JAMA Surg. 2015;150(7):650.
- Huang ES, Karsan S, Kanwal F, Singh I, Makhani M, Spiegel BM. Impact of nasogastric lavage on outcomes in acute GI bleeding. Gastrointestinal Endoscopy. 2011;74(5):971-980.
- Witting MD, Magder L, Heins AE, Mattu A, Granja CA, Baumgarten M. Usefulness and validity of diagnostic nasogastric aspiration in patients without hematemesis. Ann Emerg Med. 2004 Apr;43(4):525-32.
- Dakik HK, Srygley FD, Chiu S-T, Chow S-C, Fisher DA. Clinical performance of prediction rules and nasogastric lavage for the evaluation of upper gastrointestinal bleeding: a retrospective observational study. Gastroenterology Research and Practice. 2017;2017:1-8.
- Villanueva C, Colomo A, Bosch A, et al. Transfusion strategies for acute upper gastrointestinal bleeding. N Engl J Med. 2013;368(1):11-21.
- Carson JL, Grossman BJ, Kleinman S, Tinmouth AT, Marques MB, Fung MK, Holcomb JB, Illoh O, Kaplan LJ, Katz LM, Rao SV, Roback JD, Shander A, Tobian AA, Weinstein R, Swinton McLaughlin LG, Djulbegovic B; Clinical Transfusion Medicine Committee of the AABB. Red blood cell transfusion: a clinical practice guideline from the AABB. Ann Intern Med. 2012 Jul 3;157(1):49-58.
- Docherty AB, O’Donnell R, Brunskill S, et al. Effect of restrictive versus liberal transfusion strategies on outcomes in patients with cardiovascular disease in a non-cardiac surgery setting: systematic review and meta-analysis. BMJ. Published online March 29, 2016:i1351.
- Derogar M, Sandblom G, Lundell L, et al. Discontinuation of low-dose aspirin therapy after peptic ulcer bleeding increases risk of death and acute cardiovascular events. Clinical Gastroenterology and Hepatology. 2013;11(1):38-42.
- Chan FKL, Leung Ki E-L, Wong GLH, et al. Risks of bleeding recurrence and cardiovascular events with continued aspirin use after lower gastrointestinal hemorrhage. Gastroenterology. 2016;151(2):271-277.
- Staerk L, Lip GYH, Olesen JB, et al. Stroke and recurrent haemorrhage associated with antithrombotic treatment after gastrointestinal bleeding in patients with atrial fibrillation: nationwide cohort study. BMJ. Published online November 16, 2015:h5876.
- Qureshi W, Mittal C, Patsias I, et al. Restarting anticoagulation and outcomes after major gastrointestinal bleeding in atrial fibrillation. The American Journal of Cardiology. 2014;113(4):662-668.
- HALT-IT Trial Collaborators. Effects of a high-dose 24-h infusion of tranexamic acid on death and thromboembolic events in patients with acute gastrointestinal bleeding (HALT-IT): an international randomised, double-blind, placebo-controlled trial. Lancet. 2020 Jun 20;395(10241):1927-1936.
- Niikura R, Nagata N, Yamada A, et al. Efficacy and safety of early vs elective colonoscopy for acute lower gastrointestinal bleeding. Gastroenterology. 2020;158(1):168-175.e6.
- Rongen I, Thomassen BJW, Perk LE. Early versus standard colonoscopy: a randomized controlled trial in patients with acute lower gastrointestinal bleeding: results of the BLEED study. Journal of Clinical Gastroenterology. 2019;53(8):591-598.
- Tsay C, Shung D, Stemmer Frumento K, Laine L. Early colonoscopy does not improve outcomes of patients with lower gastrointestinal bleeding: systematic review of randomized trials. Clinical Gastroenterology and Hepatology. 2020;18(8):1696-1703.e2.