Authors: Nicholas Mancuso, MD (EM Resident Physician, University of Kentucky) and Michael Sweeney, MD (Assistant Professor, University of Kentucky, Dept. of Emergency Medicine) // Edited by: Alex Koyfman, MD (@EMHighAK, EM Attending Physician, UTSW / Parkland Memorial Hospital) and Brit Long, MD (@long_brit)
Case
A 68-year-old female presents to the ED with abdominal pain, bloating, and nausea which she states began this morning upon wakening about 3 hours prior to arrival. She appears moderately uncomfortable and pale. She is urgently brought to an acute care room and evaluated by the resident physician. Her initial vital signs reveal tachycardia of 120 beats/minute, blood pressure of 103/52, oxygen saturation of 90% room air, respiratory rate of 24 breaths/minute, and an oral temperature of 99.0° F. Prior medical history includes hypertension and hypercholesterolemia, and she has a surgical history of two caesarean sections, bilateral tubal ligation, and open cholecystectomy.
She says it is worse “right in the middle,” and she “feels like vomiting but hasn’t yet”. She does report belching several times with a “foul smell”. Her daughter and granddaughter are sick with a stomach virus, and she thinks she has the same. Her last bowel movement was the evening prior and was formed.
She is rolling on the bed. She is tender over her upper quadrants diffusely with guarding but no rebound. Her exam otherwise is unremarkable. Analgesic and anti-emetic medicine is administered, as well as a bolus of intravenous fluid. Labs results reveal a leukocytosis of 15, lactate of 5.3, and normal lipase and CMP. Urinalysis is unremarkable. KUB for her 7/10 pain shows slightly dilated small bowel concerning for “possible obstruction, please base on history”. CT with IV and PO contrast is obtained which shows complete obstruction with transition point in the RUQ consistent with a small bowel obstruction (SBO), trace contrast distal to this point, and mild free fluid around the dilated bowel.
Presentation
Bowel obstructions are a relatively common presentation in Emergency Departments (EDs) across the country and are most often due to mechanical obstruction. The majority (~75%) of bowel obstructions occur in the small bowel (1), with the remainder afflicting the large bowel (LBO).
Bowel obstructions may present along a spectrum and can pose a diagnostic challenge, with early or low grade obstruction manifesting with non-specific symptoms and a non-focal exam in a relatively well appearing patient. Others are more obvious with classic symptoms of abdominal distention, feculent belching or emesis, significant pain, and decreased to no bowel movements/flatus. Presentation will also vary depending on the location of the obstruction (proximal versus distal).
Recognition of risk factors can aid in the timely workup and diagnosis of an ill patient with bowel obstruction. It is well known that previous abdominal surgeries are a risk factor due to development of adhesions, which complicate up to 90-93% intra-abdominal surgeries (2,3). 75% of small bowel obstructions are caused by adhesions in adults in one review (4). In another meta-analysis that looked at 196 studies, those with a known cause of SBO had adhesions as the etiology in 56% (5). Other common causes include a history or current abdominal or groin hernia, prior radiation treatment, neoplasm, inflammation, abscess, or ingested foreign body.
Notably, those with a previous obstruction due to any cause have higher rates of re-obstruction and tend to have them occur sooner (6, 7).
A common misconception among some is that ongoing passage of stools is inconsistent with small bowel obstruction. However, flatus and feces may pass for 12 to 24 hours after obstruction as the distal bowel decompresses.
Large bowel obstruction typically occurs in older patients. The most common cause is a colorectal malignancy in up to 60%. Other etiologies include volvulus, hernia with incarceration, repetitive diverticular disease, and less frequently, ischemia, adhesions, or intussusception (8).
Diagnosis
According to Eastern Association for the Surgery of Trauma (EAST) guidelines, Level III Evidence recommends obtaining plain abdominal films in a patient with a concern for a bowel obstruction (9). CT can delay surgical management and may be unnecessary in the unstable patient with plain film evidence of bowel obstruction. On plain film, signs of bowel obstruction include dilated loops of bowel (>3 cm), air-fluid levels, and paucity of gas in the distal bowel. However, many providers will go straight to CT, which is often the required test for diagnosis.
Per EAST guidelines, if plain films are inconclusive (and the patient is stable enough), CT with IV and oral contrast is indicated (Level I), which will reliably identify the degree and location of obstruction, and often the cause. In 2015 the American College of Radiology (ACR) Appropriateness criteria actually recommended against PO contrast as it increased time to CT, patient discomfort, and symptoms, and it was not found to increase accuracy (10). IV contrast helps to distinguish ischemia. A study did find up to 70% of institutions still use PO contrast, and this is highly institution and surgeon specific. Findings include a “transition point” between distended and decompressed bowel, bowel wall and mesenteric edema, and transudative fluid or ascites. These findings do not require PO contrast (10).
A meta-analysis in 2013 pooled data from 5 large studies and demonstrated x-ray to have a sensitivity of 75% and specificity of 66%. CT with IV and PO had a sensitivity of 87% and specificity of 81%. Modern scanners demonstrate sensitivity as high as 96% to 100% (11). CT without PO contrast demonstrates similar statistics with modern scanning technology (10,11).
Finally, ultrasound can assist at the bedside. In one prospective study with 123 patients, a sensitivity of 95% and specificity of 82% were reported (12), with a second study demonstrating sensitivity and specificity of 91% and 84%, respectively (13). This can be learned reportedly in a 10-15 minute session, with different diagnosis requirements all focusing on non-compressible bowel next to compressible segments. A high-frequency probe is most commonly used to evaluate for bowel greater than 2.5 cm in diameter next to compressible bowel, while edema of bowel wall and free fluid are secondary signs (14). Please see this post for more information on US in bowel obstruction: http://www.emdocs.net/ultrasound-small-bowel-obstruction/.
From: http://www.em.emory.edu/ultrasound/ImageWeek/Abdominal/small_bowel_obstruction1.html.
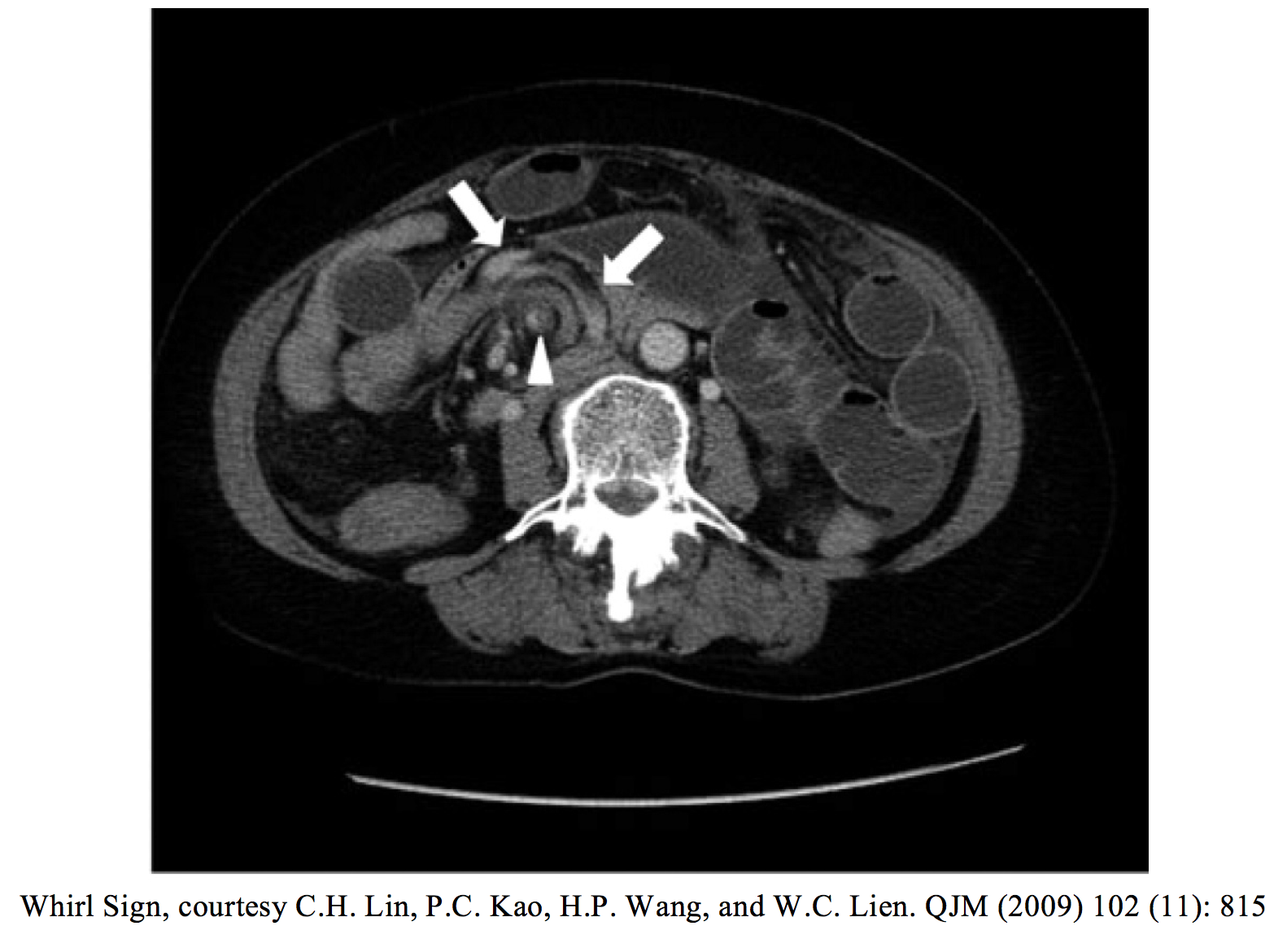
An important subset of bowel obstruction which can be missed on exam and imaging studies is a closed loop obstruction. This is a true emergency and occurs when the bowel is obstructed in two places, creating a “closed loop” with no anterograde or retrograde flow possible. Closed loop obstructions have higher complication rates for ischemia and perforation, and for a septic presentation. Many are diagnosed in the OR. Signs to look for on CT are a distended, fluid-filled bowel loop in C or U shape, the “whirl sign” (abdomen bowel rotated around its mesentery resembling a whirlpool), the “beak sign” (tapering bowel loops at the point of obstruction), or two adjacent collapsed loops of bowel at the site of obstruction (15).
For LBOs, radiographs can be an appropriate starting point, though again CT displays better sensitivity and specificity. Two classic findings are useful for distinguishing sigmoid versus cecal volvulus. The sigmoid colon is mobile on the mesentery, and the classic “coffee-bean” sign is seen on x-ray in sigmoid volvulus, which is more common in elderly (16,17). Cecal volvulus is typically seen in younger patients (20-60 years old), who present with a dilated loop in the mid abdomen pointing to the epigastrium or left upper quadrant (16,17).
Management
Early surgical intervention is the most important step in the management of the unstable patient with bowel obstruction. With high grade obstructions, bowel wall ischemia can lead to perforation and pneumoperitoneum. If leukocytosis, fever, peritonitis, metabolic acidosis, or a high lactic acidosis are present, exploratory laparotomy is Level 1 recommendation according to EAST guidelines.
Serum lactate is a sensitive marker of bowel ischemia or severe volume depletion. In addition to routine labwork such as CBC and CMP, pre-operative labs such as type and screen and coagulation studies should be added to the unstable patient. Confirmation of code status and goals of care in selected patients with significant comorbidities and increase peri-operative mortality is advised.
Bowel obstruction, especially proximal SBO, can present with significant vomiting and fluid losses. IV fluid resuscitation is indicated in those with abnormal vitals or signs of shock. Some surgeons favor early surgical management, as fluid resuscitation carries the risk of significant bowel edema, which can complicate operative intervention.
Early administration of antibiotics after blood and urine cultures are obtained is indicated in unstable or febrile patient. Regimens include Piperacillin/Tazobactam (3.375 grams IV q 6hr) or the combination of Cefepime (2 grams IV q8 hr) and Metronidazole (500 mg IV q8hr).
Nasogastric tube placement and low intermittent suction can help decompress the bowel proximal to the obstruction and relieve bowel wall ischemia due to high intraluminal pressures. In actively vomiting patients, this may also reduce the incidence of aspiration.
For a partial or chronic SBO, or a hemodynamically stable complete obstruction, NG placement, NPO status, appropriate analgesics and anti-emetics, and IV fluid are indicated. Non-operative management is often successful for up to 3-5 days (Level III). Call a surgical service for admission.
LBOs from volvulus are treated typically with a rectal tube or flexible sigmoidoscopy for sigmoid volvulus, with admission for observation given high rate of recurrence. (17,18). However, cecal volvulus more commonly requires surgical management (17,18). Patients often need fluid resuscitation. For perforation or cases due to malignancy, surgery is needed.
In terms of morbidity and mortality, numbers vary greatly by institution and surgeon. In one retrospective study from 1991 to 2002 using the VA database, the 30 day mortality for SBOs was 7.7%, with an odds of death reported higher in sicker patients and older patients (over 80) (19,20). In another, all postop mortality were due to patients 75 and older, long term affected by ASA class III disease, number of “obstructed structures”, and number of surgical complications such as intestinal injury (21).
Pearls
-For the unstable patient with bowel obstruction early surgical consultation and laparotomy are crucial. Manage ABCs, administer resuscitative IV fluids, and place a nasogastric tube to decrease chance of aspiration and for pain control. Abdominal upright x-ray is not as sensitive as CT; if x-ray unremarkable and clinical concern still exists, obtain CT. CT first may be warranted.
– Ultrasound is a viable newer alternative that appears to take place between X-ray and CT in sensitivity and specificity, is faster, but will be difficult to convince surgeons for now.
–If peritonitic or septic, cover with antibiotics (institution-specific, not a lot of evidence).
– Be wary of nonspecific findings in early presentations of bowel obstruction. Labwork directed to detect signs of ischemia (lactate, blood gas). Ongoing passage of stools or flatus does not rule out an obstruction.
– Closed loop obstruction represents both a diagnostic challenge and a complication with high morbidity and mortality. These patients may present with very little distention but can be very ill. Look for C or U signs, whirl sign, or beak sign on CT.
– For LBOs, most common cause is by far malignancy (~60%). Sigmoid volvulus shows up as the “coffee-bean” sign, more common in elderly and nursing home patients. Cecal volvulus points to the left upper quadrant and is typically seen in a younger patient (20-60 years old).
References / Further Reading
- DrożdżW, Budzyński P. Change in mechanical bowel obstruction demographic and etiological patterns during the past century: observations from one health care institution. Arch Surg. 2012 Feb;147(2):175-80.
- Parker MC, Ellis H, Moran BJ, Thompson JN, Wilson MS, Menzies D, et al. Postoperative adhesions: ten–year follow–up of 12,584 patients undergoing lower abdominal surgery.Dis Colon Rectum. 2001 Jun;44(6):822–829. discussion 9–30.
- Menzies D, Ellis H. Intestinal obstruction from adhesions––how big is the problem? Ann R Coll Surg Engl. 1990 Jan;72(1):60–63.
- Bizer LS, Liebling RW, Delany HM, Gliedman ML. Small bowel obstruction: the role of nonoperative treatment in simple intestinal obstruction and predictive criteria for strangulation obstruction. 1981 Apr;89(4):407–413
- Ten Broek, Richard P G et al. “Burden of Adhesions in Abdominal and Pelvic Surgery: Systematic Review and Met-Analysis.”The BMJ 347 (2013): f5588. PMC. Web. 16 Jan. 2017.
- Miller G, Boman J, Shrier I, Gordon PH. Natural history of patients with adhesive small bowel obstruction. Br J Surg. 2000 Sep;87(9):1240-7. PMID 10971435
- Barkan H, Webster S, Ozeran S. Factors predicting the recurrence of adhesive small-bowel obstruction. Am J Surg. 1995 Oct;170(4):361-5 PMID 7573729
- Kahi CJ, Rex DK. Bowel obstruction and pseudo-obstruction Gastroenterol Clin North Am. 2003 Dec;32(4):1229-47
- Maglinte DD, Reyes BL, Harmon BH, et al. Reliability and role of plain film radiography and CT in the diagnosis of small-bowel obstruction. AJR Am J Roentgenol. 1996;167:1451–1455
- Katz DS, Baker ME, Rosen MP, Lalani T, Carucci LR, Cash BD, Kim DH, Piorkowski RJ, Small WC, Smith MP, Yaghmai V, Yee J, Expert Panel on Gastrointestinal Imaging. ACR Appropriateness Criteria® suspected small-bowel obstruction. [online publication]. Reston (VA): American College of Radiology (ACR); 2013.
- Mark R. Taylor, MD, and Nadim Lalani, MD, FRCPC. Adult Small Bowel Obstruction EVIDENCE-BASED DIAGNOSTICS, SAEM publication. 2013
- Schmutz, G.R. et al. Small bowel obstruction: role and contribution of sonography. Eur Radiol 7, 1054-1058
- Jang, Timothy B. Schindler, Danielle. Kaji, Amy H. Bedside ultrasonography for the detection of small bowel obstruction in the emergency department. Emerg Med J 2011 28:676-678
- Dawson, Matthew. Mallin, Mike. Introduction to Bedside Ultrasound: Volumes 1 and 2. Chapter 29: Small Bowel Obstruction
- Balthazar EJ, Birnbaum BA, Megibow AJ, Gordon RB, Whelan CA, Hulnick DH. Closed-loop and strangulating intestinal obstruction: CT signs. Radiology. 1992 Dec;185(3):769-75.
- Brant WE, Helms CA. Fundamentals of Diagnostic Radiology. Lippincott Williams & Wilkins. (2007) ISBN:0781761352.
- Salati U, Mcneill G, Torreggiani WC. The coffee bean sign in sigmoid volvulus. Radiology. 2011;258 (2): 651-2
- Swenson BR, Kwaan MR, Burkart NE, Wang Y, Madoff RD, Rothenberger DA, Melton GB. Colonic volvulus: presentation and management in metropolitan Minnesota, United States. Dis Colon Rectum. 2012 Apr;55(4):444-9.
- Jose J. Diaz, Jr., MD, Faran Bokhari, MD, Nathan T. Mowery, MD, Jose A. Acosta, MD, Ernest F. J. Block, MD, William J. Bromberg, MD, Bryan R. Collier, DO, Daniel C. Cullinane, MD, Kevin M. Dwyer, MD, Margaret M. Griffen, MD, John C. Mayberry, MD, and Rebecca Jerome, MLIS, MPH. Guidelines for Management of Small Bowel Obstruction. J Trauma. 2008;64:1651–1664
- Margenthaler JA, Longo WE, Virgo KS, Johnson FE, Grossmann EM, Schifftner TL, Henderson WG, Khuri SF. Risk factors for adverse outcomes following surgery for small bowel obstruction Ann Surg. 2006 Apr;243(4):456-64.
- Duron JJ, du Montcel ST, Berger A, Muscari F, Hennet H, Veyrieres M, Hay JM; French Federation for Surgical Research. Prevalence and risk factors of mortality and morbidity after operation for adhesive postoperative small bowel obstruction. Am J Surg. 2008 Jun;195(6):726-34. doi: 10.1016/j.amjsurg.2