Author: Tim Montrief (@EMinMiami, EM Resident Physician, Jackson Memorial Health System) // Edited by: Alex Koyfman, MD (@EMHighAK) and Brit Long, MD (@long_brit)
Case: A 56-year-old man with no past medical history presents to the emergency department with severe abdominal pain, nausea, vomiting, diarrhea, after eating two wild mushrooms he found the day prior while hiking. His labs include an AST and ALT >11,000U/L, INR of 6.5, serum pH of 7.1, and lactic acid level of 17 after fluid resuscitation. What is your next step?
Background
Although acute liver failure (ALF) is a rare clinical presentation in the ED, it carries a high mortality, morbidity, and resource cost. Across the developing world, the underlying etiology is primarily viral, with hepatitis B and E infections recognized as leading causes in many countries. Conversely, in the USA and much of Europe, the incidence of virally associated liver failure has declined substantially, with most cases now arising from drug-induced liver injury, frequently from acetaminophen or idiosyncratic drug reactions. While data from developing countries is sparse, surveillance reports from the developed world suggest an overall incidence between one and six cases per million people every year, or around a total of 2,000 cases per year.1-4
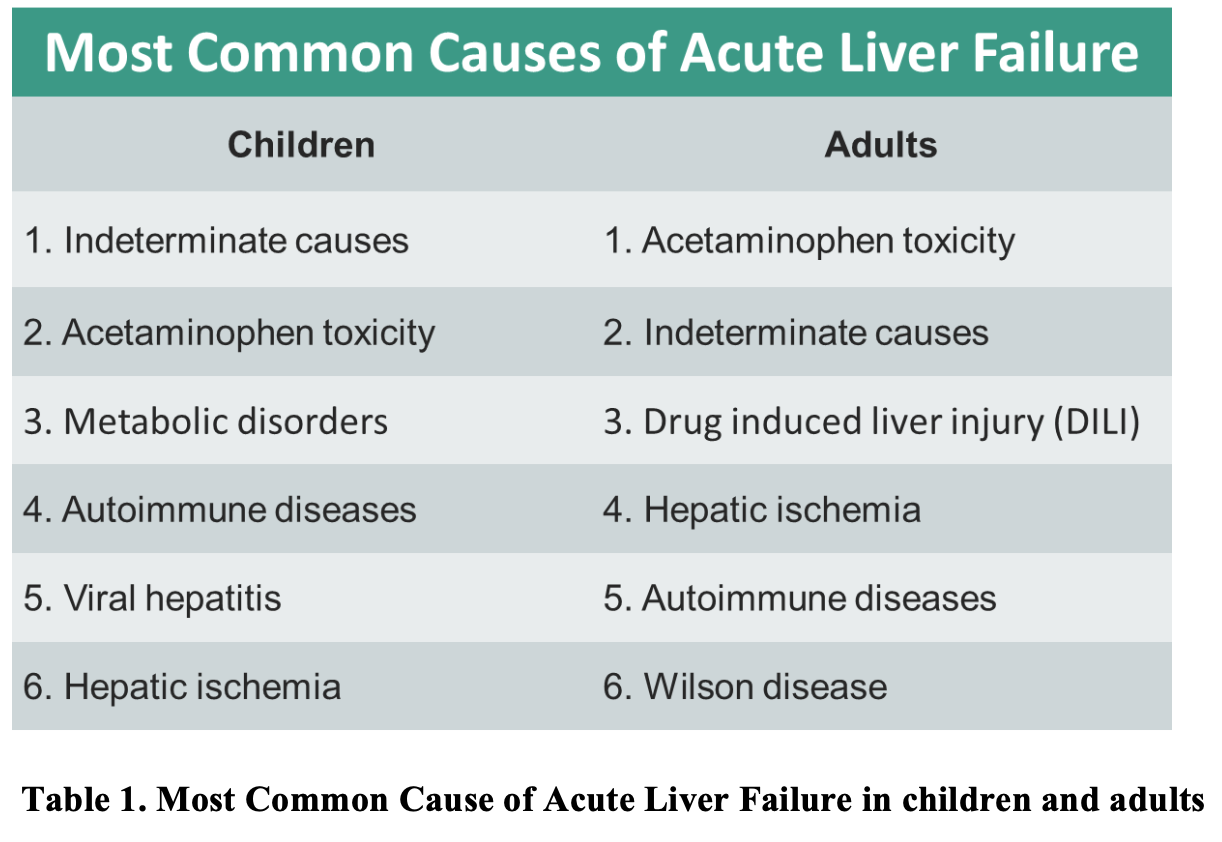
Acute liver failure is the clinical manifestation of sudden and severe hepatic injury, and has a variety of underlying etiologies, including drug toxicity, viral infections, autoimmune and genetic disorders, thrombosis, malignancy, heat injury, and ischemia. Additionally, metabolic disorders like Wilson disease (WD), HELLP (hemolysis, elevated liver enzymes, low platelets) syndrome, acute fatty liver of pregnancy, Reye’s syndrome, galactosemia, hereditary fructose intolerance, hemochromatosis, α1-antitrypsin deficiency and tyrosinemia may also cause ALF.5 The most common etiologies in adults and children, from most to least common, are provided in Table 1. While there are more than 40 definitions of acute liver failure in use, many of themodern definitions recognize the distinct disease phenotypes and seek to quantify the interval between the onset of symptoms and the development of encephalopathy.6-8 Presently, the definition of ALF includes the following features: international normalized ratio (INR) at least 1.5, neurologic dysfunction with any degree of hepatic encephalopathy, no preexisting cirrhosis, and disease course of 26 weeks or less (Table 2).9-11

Clinical features of acute liver failure
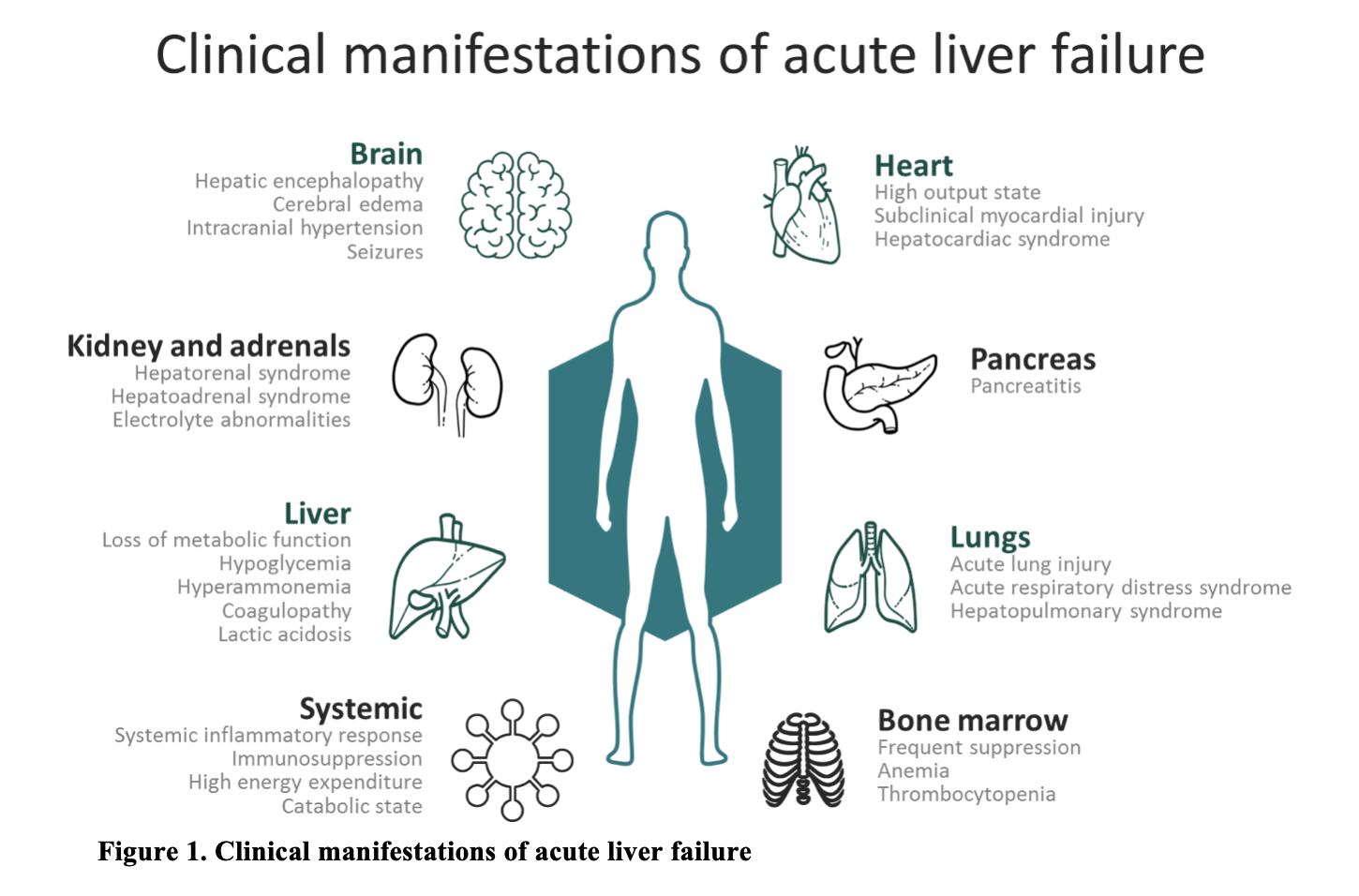
The manifestation, timing, and severity of ALF’s clinical features vary according to its underlying etiology, and a high index of suspicion is required to make an early diagnosis. The initial manifestation of ALF may range from non-specific constitutional symptoms including malaise, anorexia, fatigue, nausea, vomiting, and abdominal pain to severe hypotension, sepsis, seizures, and hepatic encephalopathy (Fig. 1).12,13 The clinical course of ALF follows that of multiple organ failure. The loss of hepatocyte function results in liver necrosis, as well as a release of toxins and cytokines leading to severe systemic inflammation and secondary bacterial infections from decreased immunity.14-16 Direct hepatic necrosis causes severe and rapid loss of metabolic function, resulting in decreased gluconeogenesis, lactate clearance, ammonia clearance, and synthetic capacity, which present clinically as hypoglycemia, lactic acidemia, hyperammonemia, and coagulopathy, respectively.17-22 Following liver necrosis and release of cytokines, a systemic inflammatory state begins, characterized by vasodilation resembling septic shock.23-25 Likewise, acute renal failure and hepatorenal syndrome are important complications of ALF and are primarily a result of the hemodynamic alterations in ALF.26-28 Initially renal injury is prerenal in etiology secondary to hypovolemia, but acute tubular necrosis rapidly develops due to ongoing ischemia of renal tubules.29,30 Likewise, direct renal toxicity may be seen, as is in the case of acetaminophen toxicity or amanita poisoning.31, 32
Encephalopathy in ALF remains a key neurological manifestation of ALF, and comprises a number of clinical manifestations of differing severity, ranging from drowsiness, slowed mentation, cognitive impairment, confusion, and euphoria to deep coma.33 Hepatic encephalopathy is classified based on severity, ranging from grade 1 to grade 4.34 Grade 1 is defined as altered behavior with euphoria, anxiety, and decreased attention span, while grade 2 is distinguished by disorientation, lethargy or asterixis. Grade 3 presents with marked disorientation, incoherent speech, and somnolence; and grade 4 is defined as a patient becoming comatose or unresponsive to verbal or pain stimuli. While the pathogenesis of hepatic encephalopathy is not fully understood, it is believed that inflammatory mediators and circulatory neurotoxins, such as ammonia, alter cerebral blood flow and blood-brain barrier permeability.35, 36 Cerebral edema is seen in 75% to 80% of patients with ALF and grade 4 hepatic encephalopathy.37 This edema can be life threatening, as it can progress to intracranial hypertension, accounting for 20% to 25% of deaths in ALF.12,38
History and physical pearls
The diagnosis of ALF is established through a thorough history, including chronology of events prior to presentation, physical exam, and supportive laboratory studies. It is imperative to obtain specific historical information to help better decide the potential cause of the patients’ presentation to the emergency department, and necessary investigations. Assessment of risk factors and potential exposures, including current medications, drug ingestions, alcohol or substance abuse, pregnancy status, family history, and recent travel is helpful in discovering the underlying etiology. Further qualifying any exposure to specific hepatotoxins, such as the Amanita phalloides mushroom, is imperative, as this information may change management strategies and therapy. Likewise, key historical risk factors for ALF include age > 40 years, female gender, poor nutritional status, pregnancy, chronic hepatitis, the use of multiple acetaminophen-containing medications for chronic pain.39-42 Clarification of the time course of the illness, specifically the interval from the onset of jaundice to the development of encephalopathy, provides the clinician with diagnostic and prognostic value.
The underlying etiology of ALF is not only established through history and laboratory testing, but also by the exclusion of alternative causes, including acute presentations of chronic liver diseases. Chronic liver diseases, including hepatitis B and autoimmune hepatitis, routinely present as an acute exacerbation with clinical features indistinguishable from ALF, a well-described phenomena known as “acute-on- chronic” hepatic failure.43-47 All components of the physical exam are important when presented with a patient with ALF. For instance, jaundice, scleral icterus, fetor hepaticus, and Kayser-Fleischer rings may be subtle signs prompting further evaluation of ALF. A detailed abdominal exam may reveal hepatomegaly due to acute viral hepatitis, CHF with secondary hepatic congestion, Budd-Chiari syndrome, or infiltrative malignancies. Physical exam findings may provide evidence suggesting the presence of an underlying chronic liver disease, including splenomegaly, caput medusa, gynecomastia, spider angiomata, palmar erythema, and ascites. Likewise, a detailed neurologic exam, especially in the patient presenting with hepatic encephalopathy, is necessary to evaluate for signs of cerebral edema and increased intracranial hypertension. Evaluation for any signs and symptoms of a concomitant infectious process may provide significant clinical value.
Laboratory and Imaging considerations
Laboratory and imaging studies should be pursued in order to confirm the diagnosis of ALF, evaluate for end organ dysfunction, and provide accurate prognostication of the patient’s clinical condition. It is beneficial to evaluate for electrolyte and metabolic abnormalities with a basic metabolic panel, venous blood gas, and lactate level. Additionally, hepatic abnormalities should be investigated by obtaining liver enzymes, alkaline phosphatase, direct/indirect bilirubin, ammonia, albumin, and coagulation studies. A pregnancy test should be pursued in any woman of child bearing age at risk for HEELP syndrome, and a blood type and screen sent in cases of suspected gastrointestinal bleed from severe coagulopathy. A complete blood count will reveal any leukocytosis, anemia, and thrombocytopenia, while blood cultures may be useful in the febrile patient. Likewise, the clinician should have a low threshold for obtaining toxicologic studies, including acetaminophen level and any possible co-ingestants. The Association for the Study of Liver Diseases recommends obtaining acetaminophen levels in all patients with ALF, regardless of any history of acetaminophen ingestion.9 Acetaminophen levels in the blood vary depending on the time of consumption, and therefore, a low acetaminophen level does not exclude acetaminophen-induced hepatotoxicity.48 Other tests should be guided by the physical exam, including serum ceruloplasmin and copper in suspected presentations of Wilson disease, viral hepatology serologies, autoimmune hepatitis markers, serum ammonia, and viral hepatitis PCR studies.
Imaging is not necessary to establish a diagnosis of ALF but may be useful in the correct clinical context. For instance, an abdominal ultrasound may provide information regarding any hepatic venous disease or underlying Budd-Chiari syndrome, while a chest x-ray is useful to evaluate for aspiration pneumonia in the vomiting patient. Likewise, in the patient with encephalopathy, a CT scan of the brain or bedside ultrasound may provide information regarding cerebral edema and intracranial hypertension.
Management of acute liver failure
General Approach
The most important aspect of management involves the timely diagnosis of ALF. Making a timely diagnosis of ALF in a patient who presents with liver dysfunction and an altered mental state remains the single most important management step for the clinician, as a delay can lead to substantial morbidity and mortality. While there is no proven therapy for ALF, understanding the progression of ALF, from loss of hepatocytes to the development of multiorgan failure, helps the clinician in disease-specific complication management. Generally speaking, the management of ALF should involve:49
- Identification of the etiology of ALF whenever possible and initiation of specific treatment.
- Supportive and symptomatic management of ALF, with timely transfer to the critical care unit.
- Early consultation with liver transplant specialists and transfer of patients to a liver transplant center when necessary.
Identification and treatment of underlying etiology
Identification of the underlying cause of ALF is necessary, as treatment varies. For instance, the most prevalent cause of ALF in the U.S. is acetaminophen toxicity, and there is an effective antidote available, N-acetylcysteine (NAC). Hepatotoxicity is not usually seen in the immediate period following acetaminophen ingestion and the treatment of patients with acetaminophen toxicity differs from the treatment of patients with ALF. The Rumack-Mathew nomogram helps predict the development of hepatotoxicity in patients with acetaminophen toxicity.50 In confirmed cases of acetaminophen toxicity, acetaminophen levels should be plotted on the nomogram in order to determine the risk of development of hepatotoxicity, and NAC should be immediately started, as it is most efficacious when given within 8 hours of ingestion.51 However, it may still be effective up to 48 hours post-ingestion. NAC has a favorable side-effect profile.52 Thus, NAC should be administered in all patients with suspected or confirmed acetaminophen toxicity even if they present beyond 8 hours of presentation.9 Dosing information is provided below, for both intravenous and oral preparations. Studies have shown that the oral NAC is as effective as intravenous NAC.53

Drug-induced hepatotoxicity is a diagnosis of exclusion, any drug identified as the possible cause of ALF should be immediately stopped. While the efficacy of NAC in drug-induced liver failure has not been clearly demonstrated,NAC administration is recommend, with equivalent dosing as in acetaminophen toxicity.9 Additionally, three drugs have been proposed for the treatment of ALF secondary to mushroom poisoning: penicillin G, silibinin (also known as silymarin or milk thistle), and NAC.9 The dose of intravenous penicillin G for mushroom induced ALF in the U.S is 300,000 units to 1 million units/kg/day.54,55
Finally, an underlying etiology of viral hepatitis may affect management of the patient with ALF. Unfortunately, Hepatitis A- and hepatitis E-induced ALF have no specific treatment and should receive supportive care.56 However, Acute hepatitis B-induced ALF patients benefit from antiviral agents, including Lamivudine, and their use is recommended by the AASLD.57,58 Likewise, patients with documented or suspected herpes virus or varicella zoster virus infection should be given intravenous acyclovir, 5 to 10 mg/kg every 8 hours for at least 7 days.9
Supportive and symptomatic management
Since there is no proven therapy for ALF, supportive and symptomatic management remain the cornerstone of treatment. One of the most prevalent complications of ALF is hepatic encephalopathy. Treatment goals include preventing the onset of encephalopathy if possible, slowing the progression to severe encephalopathy, and minimizing the development of cerebral edema and ICH. Lactulose has been an accepted treatment for acute hepatic encephalopathy, with at an initial dose of 45 ml, followed by a repeated dose every hour until the patient has a bowel movement. For patients at significant risk for aspiration, lactulose may be given as an enema (dosing is 300ml in 700 ml of water, retained for one hour) every two hours as needed until mental function improves.59 Likewise, any intracranial hypertension requires aggressive management, aiming for a goal intracranial pressure (ICP) of less than 20 to 25 mm Hg.
Coagulopathy remains an important complication of ALF. In the absence of bleeding, routine correction of thrombocytopenia or elevated INR by infusion of plasma is not indicated in ALF.9 However, patients with ALF have been shown to be deficient in vitamin K and the AASLD recommends administration of vitamin K (5 to10 mg subcutaneously).9 If the patient has clinically significant bleeding, or needs to have an invasive procedure with a high risk of bleeding, such as placement of an ICP monitor, then any coagulopathy should be corrected.9
Infection complicates many cases of ALF, leading to substantial morbidity and mortality, as they can worsen encephalopathy and may preclude liver transplantation. Therefore, in critically ill patients with ALF, particularly those presenting with severe hepatic encephalopathy, broad spectrum antibiotics, most commonly a third generation cephalosporin and vancomycin, should be given.9,63 Gram-positive cocci, including staphylococci and streptococci, as well as enteric gram-negative bacteria are the most common organisms isolated in ALF patients, along with candidiasis.63
Acute renal failure plays an important role in the prognostication of ALF. Management of renal dysfunction begins with identifying the underlying etiology, which is difficult given the multifactorial nature of renal failure in the ALF patient. Prerenal failure is managed by maintaining hemodynamic stability, correcting hypovolemia, and using vasopressors as needed. Additionally, the clinician should avoid all nephrotoxic medications, including antibiotics such as aminoglycosides and nonsteroidal anti-inflammatory agents. Acute renal failure secondary to hepatorenal syndrome usually only improves when liver function is recovered or in the case of liver transplantation. Early initiation of dialysis should be considered when clinically indicated. Likewise, any associated electrolyte abnormalities, as well as hypoglycemia, should be treated promptly. Frequent monitoring of blood glucose is critical as hepatic encephalopathy commonly masks symptoms of hypoglycemia.
Liver Transplantation
When presented with a patient in ALF, an early decision should be made about whether or not the patient is a candidate for liver transplantation. If the patient is a candidate, early transfer to a transplant center is recommended to initiate simultaneous evaluation for a liver transplant by the transplant team and advanced ALF management.9,64
Prognosis and disposition
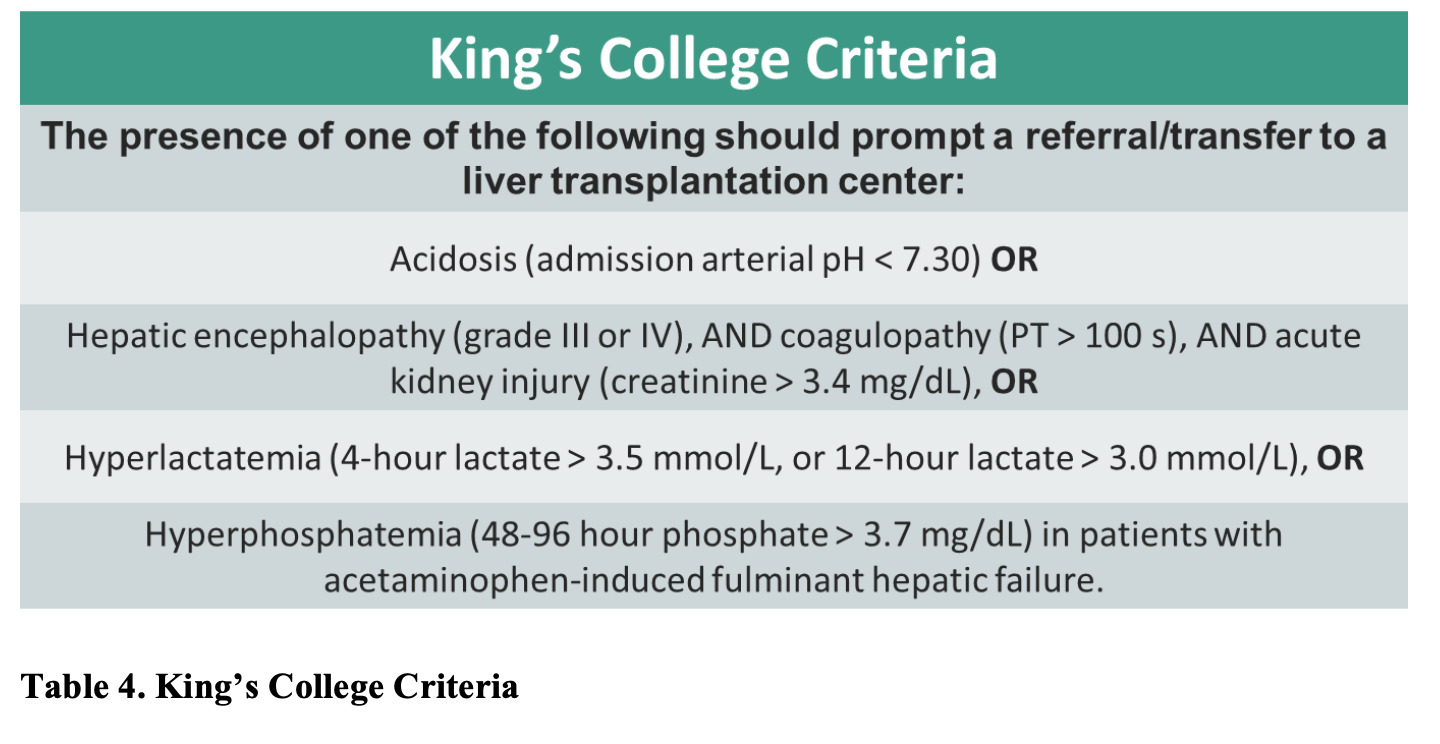
There are many prognostic tools for patients who present with ALF, but the two most common are the King’s College criteria,65 and the Model for End-Stage Liver Disease (MELD) score.66 While the King’s College Criteria remain the most widely accepted prognostic tool for patients presenting with ALF, due to its high specificity for mortality, the sensitivity and negative predictive value remain low.67,68 Thus, not fulfilling the criteria does not ensure patient survival. The King’s College Criteria has a sensitivity of 68-69% with a specificity of 82-92%,69 and may not reliably prognosticate ALF in pediatric populations.70 Conversely, the MELD score is a well-established and validated predictive model of short-term mortality in patients with liver failure. Adopted by the United Network for Organ Sharing (UNOS) and the Organ Procurement and Transplantation Network (OPTN) organization, it is currently the preferred method for the allocation of donor organs in patients awaiting liver transplantation in the US.71,72
Disposition is dependent on a variety of factors, including underlying etiology of ALF, clinical stability, and severity of disease, possibly necessitating emergent liver transplantation. It is vital to use prognostic criteria in the evaluation of ALF patients, and contact a liver transplant hospital early, as transplantation may be the only treatment which can provide long-term benefit. Many of these patients in fulminant liver failure will require intensive care unit admission at the very least, and most likely transfer to a transplant center.73
Pearls and Pitfalls
- Acute liver failure is defined when a patient meets all of the following:
- INR ≥ 1.5
- Neurologic dysfunction with any degree of hepatic encephalopathy
- No prior evidence of liver disease
- Disease course ≤ 26 weeks
- The most common cause of ALF in the U.S. is acetaminophen toxicity, treated by N-acetylcysteine.
- Patients with ALF are at increased risk for systemic complications, including infections, encephalopathy, cerebral edema, renal failure, and severe electrolyte abnormalities.
- The management of ALF involves:
- Identification of the etiology of ALF whenever possible and initiation of specific treatment.
- Supportive and symptomatic management of ALF, with timely transfer to the critical care unit.
- Early consultation with liver transplant specialists and transfer of patients to a liver transplant center when necessary. Use the King’s College criteria for guidance.
Case Resolution: The patient is correctly diagnosed with acute liver failure secondary to amanita phalloides toxicity, and given penicillin G, silibinin, and NAC. The patient meets King’s College Criteria due to his elevated post-resuscitation lactic acidosis and serum academia. He is stabilized and transferred to a tertiary care facility for further evaluation and management, successfully undergoing an orthotopic liver transplant.
From Dr. Katy Hanson at Hanson’s Anatomy:

References/Further Reading:
- Bower WA, Johns M, Margolis HS, Williams IT, Bell BP. Population-based surveillance for acute liver failure. Am J Gastroenterol. 2007;102(11):2459-63.
- Brandsaeter B, Höckerstedt K, Friman S, et al. Fulminant hepatic failure: outcome after listing for highly urgent liver transplantation-12 years experience in the nordic countries. Liver Transpl. 2002;8(11):1055-62.
- Escorsell A, Mas A, De la mata M. Acute liver failure in Spain: analysis of 267 cases. Liver Transpl. 2007;13(10):1389-95.
- Khashab M, Tector AJ, Kwo PY. Epidemiology of acute liver failure. Curr Gastroenterol Rep. 2007;9(1):66-73.
- Wang DW, Yin YM, Yao YM. Advances in the management of acute liver failure. World J Gastroenterol. 2013;19(41):7069-77.
- Wlodzimirow KA, Eslami S, Abu-Hanna A, Nieuwoudt M, Chamuleau RA. Systematic review: acute liver failure — one disease, more than 40 definitions. Aliment Pharmacol Ther2012;35:1245-1256
- Bernuau J, Rueff B, Benhamou JP. Fulminant and subfulminant liver failure: definitions and causes. Semin Liver Dis 1986;6:97-106
- Mochida S, Nakayama N, Matsui A, Nagoshi S, Fujiwara K. Re-evaluation of the Guideline published by the Acute Liver Failure Study Group of Japan in 1996 to determine the indications of liver transplantation in patients with fulminant hepatitis. Hepatol Res 2008;38:970-979
- Lee W, Larson AM, Stravitz RT. AASLD position paper: the management of acute liver failure: update 2011. Available at: http://www.aasld.org/sites/default/files/guideline_documents/141022_Position_ALF_4UFb.pdf
- O’Grady JG, Schalm SW, Williams R. Acute liver failure: redefi ning the syndromes. Lancet 1993; 342: 273–75.
- Marudanayagam R, Shanmugam V, Gunson B, et al. Aetiology and outcome of acute liver failure. HPB (Oxford). 2009;11(5):429-34.
- Bernal W, Auzinger G, Dhawan A, Wendon J. Acute liver failure. Lancet. 2010;376(9736):190-201.
- Schiødt FV, Lee WM. Fulminant liver disease. Clin Liver Dis. 2003;7(2):331-49, vi.
- Possamai LA, Thursz MR, Wendon JA, Antoniades CG. Modulation of monocyte/macrophage function: a therapeutic strategy in the treatment of acute liver failure. J Hepatol. 2014;61(2):439-45.
- Antoniades C, Berry P, Wendon J, Vergani D. The importance of immune dysfunction in determining outcome in acute liver failure. J Hepatol 2008;49:845–61.
- Antoniades C, Quaglia A, Taams L, Mitry R, Hussain M, Abeles R, et al. Source and characterization of hepatic macrophages in acetaminophen-induced acute liver failure in humans. Hepatology 2012;56:735–46.
- Shakil AO, Kramer D, Mazariegos GV, Fung JJ, Rakela J. Acute liver failure: clinical features, outcome analysis, and applicability of prognostic criteria. Liver Transpl. 2000;6(2):163-9.
- Audimoolam VK, McPhail MJ, Wendon JA, Willars C, Bernal W, Desal SR, et al. Lung injury and its prognostic significance in acute liver failure. Crit Care Med 2014; 42(3):592–600.
- Tung J, Hadzic N, Layton M, et al. Bone marrow failure in children with acute liver failure. J Pediatr Gastroenterol Nutr. 2000;31(5):557-61.
- Bernal monterde V, Campillo arregui A, Sostres homedes C, García gil A, Simón marco MA, Serrano aulló MT. Acute necrotizing pancreatitis and severe hepatic failure: description of three cases. Transplant Proc. 2008;40(9):3117-20.
- Kuo PC, Plotkin JS, Johnson LB. Acute pancreatitis and fulminant hepatic failure. J Am Coll Surg. 1998;187(5):522-8.
- Møller S, Bernardi M. Interactions of the heart and the liver. Eur Heart J. 2013;34(36):2804-11.
- Fede G, Privitera G, Tomaselli T, Spadaro L, Purrello F. Cardiovascular dysfunction in patients with liver cirrhosis. Ann Gastroenterol. 2015;28(1):31-40.
- Moller S, Henriksen JH. Cardiovascular complications of cirrhosis. Gut. 2008;57:268–278.
- Fouad YM, Yehia R. Hepato-cardiac disorders. World J Hepatol. 2014;6(1):41-54.
- Tujios SR, Hynan LS, Vazquez MA, et al. Risk factors and outcomes of acute kidney injury in patients with acute liver failure. Clin Gastroenterol Hepatol. 2015;13(2):352-9.
- Wilkinson SP, Blendis LM, Williams R. Frequency and type of renal and electrolyte disorders in fulminant hepatic failure. Br Med J. 1974;1:186–189.
- Karvellas CJ, Durand F, Nadim MK. Acute Kidney Injury in Cirrhosis. Crit Care Clin. 2015;31(4):737-50.
- Moore K. Renal failure in acute liver failure. Eur J Gastroenterol Hepatol. 1999;11(9):967-75.
- Betrosian AP, Agarwal B, Douzinas EE. Acute renal dysfunction in liver diseases. World J Gastroenterol. 2007;13(42):5552-9.
- Kirchmair M, Carrilho P, Pfab R, et al. Amanita poisonings resulting in acute, reversible renal failure: new cases, new toxic Amanita mushrooms. Nephrol Dial Transplant. 2012;27(4):1380-6.
- Pazhayattil GS, Shirali AC. Drug-induced impairment of renal function. Int J Nephrol Renovasc Dis. 2014;7:457-68.
- Bleibel W, Al-osaimi AM. Hepatic encephalopathy. Saudi J Gastroenterol. 2012;18(5):301-9.
- Montagnese S, Biancardi A, Schiff S, et al. Different biochemical correlates for different neuropsychiatric abnormalities in patients with cirrhosis. Hepatology. 2011;53(2):558-66.
- Rama rao KV, Reddy PV, Tong X, Norenberg MD. Brain edema in acute liver failure: inhibition by L-histidine. Am J Pathol. 2010;176(3):1400-8.
- Scott TR, Kronsten VT, Hughes RD, Shawcross DL. Pathophysiology of cerebral oedema in acute liver failure. World J Gastroenterol. 2013;19(48):9240-55.
- Blei AT. Medical therapy of brain edema in fulminant hepatic failure. Hepatology. 2000;32(3):666-9.
- Jalan R. Intracranial hypertension in acute liver failure: pathophysiological basis of rational management. Semin Liver Dis. 2003;23(3):271-82.
- Larson AM, Polson J, Fontana RJ, et al. Acetaminophen-induced acute liver failure: results of a United States multicenter, prospective study. Hepatology. 2005;42(6):1364-72.
- Poddar U, Thapa BR, Prasad A, Sharma AK, Singh K. Natural history and risk factors in fulminant hepatic failure. Arch Dis Child. 2002;87(1):54-6.
- Patton H, Misel M, Gish RG. Acute liver failure in adults: an evidence-based management protocol for clinicians. Gastroenterol Hepatol (N Y). 2012;8(3):161-212.
- Canbay A, Tacke F, Hadem J, Trautwein C, Gerken G, Manns MP. Acute liver failure: a life-threatening disease. Dtsch Arztebl Int. 2011;108(42):714-20.
- Arroyo V, Jalan R. Acute-on-Chronic Liver Failure: Definition, Diagnosis, and Clinical Characteristics. Semin Liver Dis. 2016;36(2):109-16.
- Hernaez R, Solà E, Moreau R, Ginès P. Acute-on-chronic liver failure: an update. Gut. 2017;66(3):541-553.
- Arroyo V, Moreau R, Kamath PS, et al. Acute-on-chronic liver failure in cirrhosis. Nat Rev Dis Primers. 2016;2:16041.
- Arroyo V, Moreau R, Jalan R, Ginès P. Acute-on-chronic liver failure: A new syndrome that will re-classify cirrhosis. J Hepatol. 2015;62(1 Suppl):S131-43.
- De mattos ÂZ, De mattos AA. Acute-on-chronic liver failure-old concepts made clearer. Transl Gastroenterol Hepatol. 2017;2:111.
- Fontana RJ. Acute liver failure including acetaminophen overdose. Med Clin North Am. 2008;92(4):761-94, viii.
- Murali A, Narayanan Menon KV. Acute Liver Failure. Cleveland Clinic Center for Continuing Education: Disease Management. Http://www.clevelandclinicmeded.com/medicalpubs/diseasemanagement/hepatology/acute-liver-failure/. Published September 2017.
- Rumack BH, Matthew H. Acetaminophen poisoning and toxicity. Pediatrics 1975; 55:871–876.
- Whyte IM, Francis B, Dawson AH. Safety and efficacy of intravenous N-acetylcysteine for acetaminophen overdose: analysis of the Hunter Area Toxicology Service (HATS) database. Curr Med Res Opin 2007; 23:2359–2368.
- Sansone RA, Sansone LA. Getting a Knack for NAC: N-Acetyl-Cysteine. Innov Clin Neurosci. 2011;8(1):10-4.
- Kanter MZ. Comparison of oral and i.v. acetylcysteine in the treatment of acetaminophen poisoning. Am J Health Syst Pharm. 2006;63(19):1821-7.
- Nordt SP, Manoguerra A, Clark RF. 5-Year analysis of mushroom exposures in California. West J Med. 2000;173(5):314-7.
- Enjalbert F, Rapior S, Nouguier-soulé J, Guillon S, Amouroux N, Cabot C. Treatment of amatoxin poisoning: 20-year retrospective analysis. J Toxicol Clin Toxicol. 2002;40(6):715-57.
- Manka P, Verheyen J, Gerken G, Canbay A. Liver Failure due to Acute Viral Hepatitis (A-E). Visc Med. 2016;32(2):80-5.
- Tillmann HL, Patel K. Therapy of acute and fulminant hepatitis B. Intervirology. 2014;57(3-4):181-8.
- Shiffman ML. Management of acute hepatitis B. Clin Liver Dis. 2010;14(1):75-91.
- Al sibae MR, Mcguire BM. Current trends in the treatment of hepatic encephalopathy. Ther Clin Risk Manag. 2009;5(3):617-26.
- Murphy N, Auzinger G, Bernel W, Wendon J. The effect of hypertonic sodium chloride on intracranial pressure in patients with acute liver failure. Hepatology 2004; 39:464–470
- Huang WT, Cang WC, Derry KL, Lane JR, Von drygalski A. Four-Factor Prothrombin Complex Concentrate for Coagulopathy Reversal in Patients With Liver Disease. Clin Appl Thromb Hemost. 2017;23(8):1028-1035.
- Stravitz RT, Ellerbe C, Durkalski V, et al. Bleeding complications in acute liver failure. Hepatology. 2018;67(5):1931-1942.
- Stravitz RT, Kramer AH, Davern T, et al. Intensive care of patients with acute liver failure: recommendations of the U.S. Acute Liver Failure Study Group. Crit Care Med 2007;35: 2498–2508.
- O’Grady J. Timing and benefit of liver transplantation in acute liver failure. J Hepatol. 2014;60(3):663-70.
- O’Grady JG, Alexander GJ, Hayllar KM, et al. Early indicators of prognosis in fulminant hepatic failure. Gastroenterology. 1989;97:339-345.
- Wiesner R, Edwards E, Freeman R, et al. Model for end-stage liver disease (MELD) and allocation of donor livers. Gastroenterology. 2003;124:91-96.
- Bailey B, Amre DK, Gaudreault P. Fulminant hepatic failure secondary to acetaminophen poisoning: a systematic review and meta-analysis of prognostic criteria determining the need for liver transplantation. Crit Care Med. 2003;31:299-305.
- McPhail MJ, Wendon JA, Bernal W. Meta-analysis of performance of Kings’s College Hospital Criteria in prediction of outcome in non-paracetamol-induced acute liver failure. J Hepatol. 2010;53:492-499.
- American Association for the Study of Liver Diseases. AASLD position paper: the management of acute liver failure: update 2011. November 2011. http://www.aasld.org/ Accessed July 28, 2018.
- Sundaram V, Shneider BL, Dhawan A, et al. King’s College Hospital Criteria for non-acetaminophen induced acute liver failure in an international cohort of children. J Pediatr. 2013;162(2):319-23.e1.
- Kamath PS, Wiesner RH, Malinchoc M, et al. A model to predict survival in patients with end-stage liver disease. Hepatology. 2001;33:464-470.
- Wiesner R, Edwards E, Freeman R, et al. Model for end-stage liver disease (MELD) and allocation of donor livers. Gastroenterology. 2003;124:91-96.
- Castaldo ET, Chari RS. Liver transplantation for acute hepatic failure. HPB (Oxford). 2006;8(1):29-34.





