Authors: Zack Brady, MD (EM Attending Physician, San Antonio, TX); Maggie Moran, MD (EM Resident Physician, SAUSHEC / San Antonio, TX); Brit Long, MD (@long_brit) // Reviewed by: Alex Koyfman, MD (@EMHighAK)
Case
A 54-year-old male with a past medical history of hypertension, type II diabetes mellitus and hyperlipidemia who presents to the ED with slurred speech and left arm and leg weakness that started abruptly on waking this morning and has completely resolved upon arrival to the ED.
Triage vital signs (VS) include BP 156/92, HR 85, RR 16, SpO2 97% on room air, temperature 37.2 F (oral). Blood glucose is 198. While awaiting imaging, nursing staff alerts you that the patient is experiencing a recurrence of symptoms.
On evaluation, he has 1/5 strength and minimal sensation to the left lower extremity and 3/5 strength and decreased sensation to the left upper extremity. Marked left sided facial droop is noted in addition to slurred but intelligible speech. Patient is taken emergently to radiology for non-contrast CT of the head and CT angiography of the head and neck where he is found to have a large vessel occlusion in the right middle cerebral artery.
What treatments should you consider?
Epidemiology
There are approximately 795,000 strokes in the United States every year, the vast majority (87%) of which are ischemic in nature.1-2 Ischemic stroke generally portends a better outcome than hemorrhage, which is associated with both greater morbidity and mortality. A Swedish long term, longitudinal cohort revealed 30 days after index ischemic stroke, 88.9% of patients are living, compared to 49.4% at five years, which is statistically significantly less than a reference cohort of the general population. Over 2 out of 3 patients who experienced an ischemic stroke were dead or functionally dependent five years after index event.3 Large vessel occlusions (LVOs), blockages of the proximal intracranial anterior and posterior circulation, account for approximately 24% to 46% of acute ischemic strokes.4 These large vessel occlusions have been shown to be amenable to neurointerventional procedures with better clinical outcomes than thrombolytics alone.4
Background
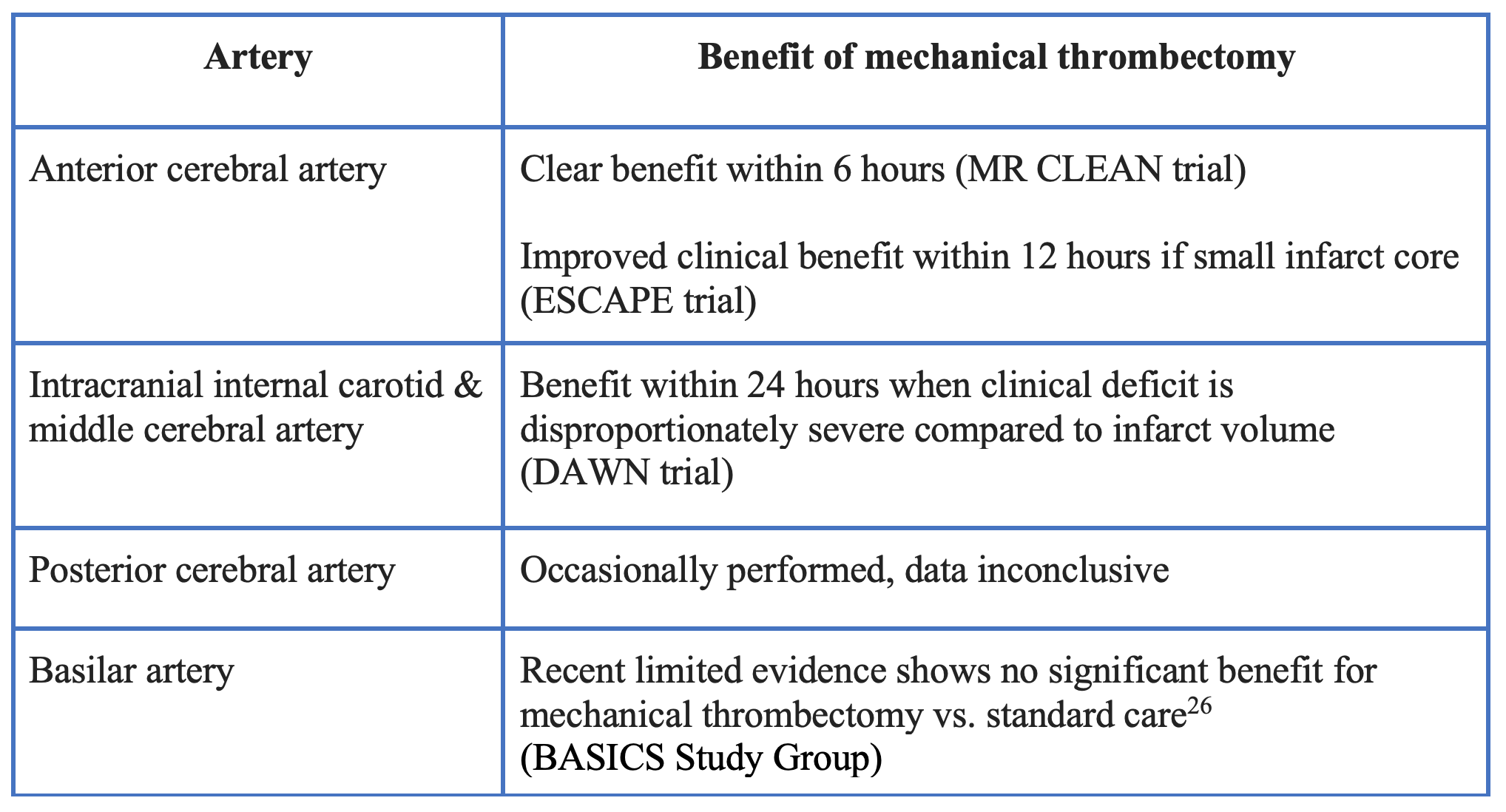
The first important discovery in stroke management was the demonstration of improved outcomes after intravenous (IV) administration of the thrombolytic, rt-PA (recombinant tissue plasminogen activator) within a 6-hour time window.5However, it was not until ten years later that a consensus time window of 4.5 hours was agreed upon, in order to mitigate the risk of conversion to hemorrhagic stroke.6-7 Although early research for mechanical thrombectomy was largely flawed and did not show significant benefit8, MR CLEAN, a multicenter randomized control trial in 2015 showed a 13.5% difference in the rate of functional independence in patients with anterior cerebral artery LVOs in the treated group (rt-PA plus mechanical thrombectomy within six hours) vs. those receiving rt-PA alone.9
The ESCAPE trial, a randomized control trial enrolling over 300 participants, showed evidence of improved clinical outcomes within 12 hours of stroke symptom onset in patients with an anterior circulation LVO with a small infarct core. The ESCAPE trial was stopped early due to efficacy, with the treatment group demonstrating both improved functional outcomes and decreased mortality.10
The DAWN trial, expanded the time window to 24 hours in patients with intracranial internal carotid artery or proximal middle cerebral artery occlusions. where the clinical deficit was disproportionately severe relative to the infarct volume found on MRI or perfusion CT. Recanalization at 24 hours was achieved in 77% of patients who underwent thrombectomy (compared to 36% treated medically).11
Based on this multicenter, prospective, randomized control trial, the AHA/ASA now recommends (COR IIa, LOE B-R) consideration of mechanical thrombectomy within 24 hours of stroke onset based on this evidence.12
When to consider large vessel occlusion (LVO)?
A 2018 retrospective consecutive cohort study evaluated prehospital clinical indicators for the presence of LVO and identified the “VAN” scoring technique as a reliable indicator for likely presence of large vessel occlusion. This study looked for the presence of hemiparesis of any extremity as indicated by drift of any severity, visual neglect noted as deviation of gaze or head positioning, and aphasia through spontaneous speech, following of verbal command, and object naming. This study found the presence of these cortical symptoms were 91% sensitive and 70% specific for the presence of LVO and 90% sensitive and 60% specific for the need for mechanical thrombectomy and, therefore, propose the presence of cortical symptoms should raise clinical suspicion for LVO.13
Treatment Options
Stent retrievers
o The thrombus is traversed using a microcatheter which houses a retrievable stent
o The stent is deployed, provided immediate reperfusion, and allowed to integrate into the clot for a few minutes
o The stent and microcatheter are retrieved, pulling out the clot

Aspiration
o A guidewire and microcatheter are navigated to or through the thrombus
o The aspiration catheter is then delivered to the proximal end of the thrombus
o The guidewire/microcatheter are removed and suction is initiated
o The clot is either aspirated through the catheter or becomes stuck at the catheter tip
o The catheter is withdrawn into the guide catheter
Benefits of contact aspiration thrombectomy include decreased procedure time14 and cost savings.15 Contact aspiration may also be amenable to more distal M2/M3 branch occlusion due to less torque placed on the vessel from a stent.8
Mechanical Thrombectomy plus Thrombolytics vs. Mechanical Thrombectomy Alone
Until recently, there has been minimal research exploring functional outcomes in patients who received mechanical thrombectomy plus thrombolytics versus those who received thrombectomy alone. A 2021 study by Suzuki et. al, the SKIP trial, was a non-inferiority study which included 204 patients with acute ischemic stroke due to a large vessel occlusion. Patient were randomly assigned to mechanical thrombectomy alone (n=101) or combined IV thrombolysis followed by thrombectomy (n=103). The primary endpoint was defined as a modified Rankin score of 0-2 at 90 days. Favorable outcome occurred in 60 patients (59.4%) in the mechanical thrombectomy alone group and 59 patients (57.3%) in the combined IV thrombolysis plus mechanical thrombectomy group, with no significant between-group difference (difference, 2.1% [1-sided 97.5% CI, -11.4% to ∞]; odds ratio, 1.09 [1-sided 97.5% CI, 0.63 to ∞] for noninferiority). This suggests that among patients with acute large vessel occlusion stroke, mechanical thrombectomy alone, compared with combined IV thrombolysis plus mechanical thrombectomy, failed to demonstrate noninferiority regarding favorable functional outcome.16 Another 2021 prospective multicenter cohort study of 226 patients with anterior circulation LVOs compared the efficacy of direct endovascular thrombectomy (dEVT) compared to IV tissue plasminogen activator (tPA) plus endovascular therapy (bridging therapy). Bridging therapy (BT) was associated with higher odds of 90-day functional independence (57% vs 44%, adjusted odds ratio [aOR] 2.02, 95% confidence interval [CI] 1.01-4.03) and functional improvement (adjusted common OR 2.06, 95% CI 1.18-3.60) and lower likelihood of 90-day mortality (11% vs 23%, aOR 0.20, 95% CI 0.07-0.58). In subgroup analyses, patients with BT with baseline NIHSS scores <15 had higher functional independence likelihood compared to those with dEVT (aOR 4.87, 95% CI 1.56-15.18); this association was not evident for patients with NIHSS scores ≥15 (aOR 1.05, 95% CI 0.40-2.74). Similarly, functional outcomes improvements with BT were detected in patients with core volume strata (ischemic core <50 cm3: aOR 2.10, 95% CI 1.02-4.33 vs ischemic core ≥50 cm3: aOR 0.41, 95% CI 0.01-16.02). This study showed better functional outcomes, especially with milder NIHSS score and smaller infarct core volumes in patients who received mechanical thrombectomy plus thrombolytics (BT).17 The MR CLEAN-NO trial consisting of 539 patients sought to assess the superiority of endovascular therapy (EVT) alone vs. EVT plus alteplase as well a non-inferiority. The primary outcome was the functional outcome on the modified Rankin scale at 90 days. The median score on the modified Rankin scale at 90 days was 3 (interquartile range, 2 to 5) with EVT alone and 2 (interquartile range, 2 to 5) with alteplase plus EVT. The adjusted common odds ratio was 0.84 (95% confidence interval [CI], 0.62 to 1.15), which showed neither superiority nor noninferiority of EVT alone. This data suggests that EVT alone was not superior or non-inferior to IV alteplase followed by EVT based on disability at 90 days.18 In a non-inferiority study, DIRECT-MT, 656 Chinese patients with anterior circulation LVOs were enrolled. 327 patients were assigned to the thrombectomy-alone group and 329 were assigned to a group receiving alteplase within 4.5 hours of stroke onset and prior to thrombectomy. Disability using the Rankin score was the primary outcome. Endovascular thrombectomy alone was noninferior to combined IV alteplase and endovascular thrombectomy with regard to the primary outcome (adjusted common odds ratio, 1.07; 95% confidence interval, 0.81 to 1.40; P=0.04 for noninferiority) but was associated with lower percentages of patients with successful reperfusion before thrombectomy (2.4% vs. 7.0%) and overall successful reperfusion (79.4% vs. 84.5%).19 This suggests that in Chinese patients with acute ischemic stroke from anterior circulation LVO, endovascular thrombectomy alone was non-inferior to combination therapy with regard to functional outcome. Another study from China, DEVT, enrolled 234 patients with proximal anterior circulation LVOs who presented within 4.5 hours of symptom onset and were eligible for IV thrombolysis. 116 patients were randomized to the endovascular thrombectomy-alone group and 118 received combination therapy with IV thrombolysis and endovascular thrombectomy. The primary end point was the proportion of patients achieving functional independence at 90 days. At the 90-day follow-up, 63 patients (54.3%) in the endovascular thrombectomy alone group vs 55 (46.6%) in the combined treatment group achieved functional independence at the 90-day follow-up (difference, 7.7%, 1-sided 97.5% CI, -5.1% to ∞)P for noninferiority = .003).20 Among patients with ischemic stroke due to proximal anterior circulation occlusion within 4.5 hours from onset, endovascular treatment alone, compared with IV alteplase plus endovascular treatment, met the prespecified statistical threshold for noninferiority for the outcome of 90-day functional independence.
If concern for LVO is present based on history and exam, the patient should undergo rapid neuroimaging and consultation with the neurologist and neurointerventionalist, who can assist in the decision to administer thrombolytics if the patient presents within the time window for thrombolysis. If the patient is not at a stroke center and requires transfer, consultation with the specialist prior to thrombolysis should be considered.
Investigational Therapies
Ultrasound-enhanced Thrombolysis
A metanalysis of seven RCTs including a total of 272 patients evaluating ultrasound-enhanced thrombolysis showed a nearly a two-fold increase in the odds of complete recanalization compared with IV thrombolysis alone in patients with large vessel occlusions. Patients randomized to sonothrombolysis had a 40.3% increased rate of recanalization vs. 22.4% for those receiving IV thrombolysis alone (OR, 2.17 [95% CI, 1.03–4.54]; adjusted OR, 2.33 [95% CI, 1.02–5.34]). The likelihood of symptomatic intracranial hemorrhage was not significantly different between the two groups (7.3% versus 3.7%; OR, 2.03 [95% CI, 0.68–6.11]; adjusted OR, 2.55 [95% CI, 0.76–8.52]).21
Intravenous & Intra-arterial tPA
A small pilot study using combined IV and local intra-arterial thrombolysis (IV/IA) vs. intraarterial thrombolysis alone (placebo/IA) within three hours of stroke symptom onset showed increased rates of recanalization but no improvement in clinical outcomes in thirty-five patients. Recanalization was better (p=0.03) in the in IV/IA group with TIMI 3 flow in 6 /11 IV/IA patients versus 1/10 placebo/IA patients and correlated to the total dose of r-TPA (p=0.05). There was no difference in the treatment groups in the primary clinical outcome as measured by the proportion of patients with a 7-point or greater improvement in the NIHSSS or a score of 0 or 1 at 7 days (24% for both groups). Additionally, there was no difference in the 90-day outcomes.22
Sphenopalatine Ganglion Stimulation
Sphenopalatine ganglion stimulation has been shown in preclinical models to increase collateral cerebral blood flow, stabilize the blood-brain barrier and reduce infarct size.23 Studies indicate that sphenopalatine ganglion stimulation is safe for patients with acute ischemic stroke 8-24 hours after onset, who are ineligible for thrombolytic therapy. In subjects with confirmed cortical involvement, 50% (121/244) in the intervention group had a 3-month disability level that was better than expected versus 40% (110/276) in the sham-control group (OR 1.48, 95% CI 1.05-2.10). These results did not reach significance but did demonstrate that patients with evidence of cortical involvement would likely have improved functional outcomes with sphenopalatine stimulation.24-25
Pearls
- Patients who present with symptoms of large vessel occlusion stroke should receive both a non-contrast CT scan of the head and a CT angiogram of the head and neck. CT perfusion imaging, if available, can assist.
- Mechanical thrombectomy has clear benefit for patients with acute ischemic stroke secondary to a large vessel occlusion in the proximal anterior cerebral artery if treated within 6 hours.
- Benefit of mechanical thrombectomy is likely in patients with LVOs who present in the 6-24 hour time frame, but should be based on clinical deficits and imaging.
- Thrombectomy should be pursued if indicated in patients, regardless of tPA status.
Case Conclusion
In consultation with neurology and neurointerventional radiology, you decide to administer tPA. Your patient is taken for mechanical thrombectomy by microcatheter and retrievable stent. Reperfusion is achieved and your patient is discharged to an acute rehabilitation facility for strength and stability before ultimately returning home with minimal residual deficits.
Digital subtraction angiography (DSA) demonstrating reperfusion can be seen below.27

References
- Benjamin EJ, Blaha MJ, Chiuve SE, et. al. Heart disease and stroke statistics-2017 update: a report from the American Heart Association. Circulation 2017; 135(10):e146-e603.
- Mozaffarian D, Benjamin EJ, Go AS, et al. Heart disease and stroke statistics-2015 update: a report from the American Heart Association. Circulation 2015; 131(4):e29-e322.
- Sennfält S, Norrving B, Petersson J et al. Long-Term Survival and Function after Stroke. Stroke 2018; Advance online publication.
- Rennert RC, Wali AR, Steinberg JA, et al. Epidemiology, Natural History, and Clinical Presentation of Large Vessel Ischemic Stroke. Neurosurgery 2019; 85(suppl_1):S4-S8.
- Hacke W, Kaste M, Fieschi C, et. al. Intravenous thrombolysis with recombinant tissue plasminogen activator for acute hemispheric stroke. JAMA 1995; 274(13):1017-1025.
- Steiner T, Bluhmki E, Kaste M, et. al. The ECASS 3-hour cohort. Secondary analysis of ECASS data by time stratification. ECASS Study Group. European Cooperative Acute Stroke Study. Cerebrovascular Diseases 1998; 8(4):198-203.
- Hacke W, Kaste M, Bluhmki E, et. al. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med 2008; 359(13):1317-1329.
- Munich SA, Vakharia K & Levy EI, Overview of Mechanical Thrombectomy Techniques, Neurosurgery 2019; 85(suppl_1): S60–S67.
- Berkhemer OA, Fransen PS, Beumer D et al. A randomized trial of intraarterial treatment for acute ischemic stroke. N Engl J Med 2015; 372(1):11-20. (MR CLEAN)
- Goyal M, Demchuk AM, Menon BK et al. Randomized assessment of rapid endovascular treatment of ischemic stroke. N Engl J Med 2015; 372(11):1019-1030. (ESCAPE)
- Nogueira RG, Jadhav AP, Haussen DC et al. Thrombectomy 6 to 24 hours after stroke with a mismatch between deficit and infarct. N Engl J Med 2018; 378(1):11-21. (DAWN)
- Powers WJ, Rabinstein AA, Ackerson T et al. 2018 Guidelines for the Early Management of Patients with Acute Ischemic Stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2018; 49(3):e46-e110.
- Beume LA, Hieber M, Kaller CP et al. Large Vessel Occlusion in Acute Stroke. Stroke 2018; 49(10):2323-2329.
- Turk AS, Frei D, Fiorella D et al. ADAPT FAST study: a direct aspiration first pass technique for acute stroke thrombectomy. J NeuroIntervent Surg 2014; 6(4):260-264.
- Spiotta A, Fargen KM, Chaudry I, Turner RD, Turk AS. ADAPT: a direct aspiration first pass technique. Endovasc Today 2016; 15(2):68-70.
- Suzuki K, Matsumaru Y, et. al. Effect of Mechanical Thrombectomy Without vs With Intravenous Thrombolysis on Functional Outcome Among Patients With Acute Ischemic Stroke: The SKIP Randomized Clinical Trial. JAMA 2021; 325(3):244-253.
- Sarraj A, Grotta J, Albers GW, et al. Clinical and Neuroimaging Outcomes of Direct Thrombectomy vs Bridging Therapy in Large Vessel Occlusion: Analysis of the SELECT Cohort Study. Neurology 2021; 96(23):e2839-e2853.
- LeCouffe NE, Kappelhof M, Treurniet KM, et al. A Randomized Trial of Intravenous Alteplase before Endovascular Treatment for Stroke. N Engl J Med 2021;385(20):1833-1844.
- Yang P, Zhang Y, Zhang L, et al. Endovascular Thrombectomy with or without Intravenous Alteplase in Acute Stroke. N Engl J Med 2020; 382(21):1981-1993.
- Zi W, Qiu Z, Li F, et al. Effect of Endovascular Treatment Alone vs Intravenous Alteplase Plus Endovascular Treatment on Functional Independence in Patients With Acute Ischemic Stroke: The DEVT Randomized Clinical Trial. JAMA 2021; 325(3):234-243.
- Tsivgoulis G, Katsanos AH, Eggers J, et al. Sonothrombolysis in Patients With Acute Ischemic Stroke With Large Vessel Occlusion: An Individual Patient Data Meta-Analysis. Stroke 2021; 52(12):3786-3795.
- Lewandowski LA, Frankel M, Tomsick TA, et. al. Combined Intravenous and Intra-Arterial r-TPA Versus Intra-Arterial Therapy of Acute Ischemic Stroke. Stroke 1999; 30(12):2598-2605
- Haviv L, Schnoknecht K, Prager O, et. al. Stimulation of the sphenopalatine ganglion induces reperfusion and blood-brain barrier protection in the photothrombotic stroke model. PLoS One. 2012; 7:e39636.
- Bornstein NM, Saver JL, Diener HC, et al. An injectable implant to stimulate the sphenopalatine ganglion for treatment of acute ischaemic stroke up to 24 h from onset (ImpACT-24B): an international, randomised, double-blind, sham-controlled, pivotal trial. Lancet 2019; 394(10194):219-229.
- Bornstein NM, Saver JL, Diener HC, et al. Sphenopalatine Ganglion Stimulation to Augment Cerebral Blood Flow: A Randomized, Sham-Controlled Trial. Stroke 2019; 50(8):2108-21117.
- Langezaal L, Van der Hoeven, E, Mont’Alverne, F, et. al. Endovascular Therapy for Stroke Due to Basilar-Artery Occlusion. The New England Journal of Medicine 2021; 384(20):1910–1920.
- Case courtesy of Dr Matt A. Morgan, <ahref=”https://radiopaedia.org/?lang=us”>Radiopaedia.org</a>. From the case <a href=”https://radiopaedia.org/cases/53899?lang=us”>rID: 53899</a>







1 thought on “Diagnosis and Neurointerventional Management of Large Vessel Occlusion Acute Ischemic Stroke”
Great read, Zach!