Authors: Ashley and Brooke Barlow, PharmDs (@theABofPharmaC) // Edited by: Craig Cocchio, PharmD, BCPS (@iempharmd); Tim Montrief, MD (@EMinMiami); Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit)
Case
It’s a busy day in your emergency department (ED) when you walk into see a 44 year old man with a past medical history of hypertension, asthma, cirrhosis 2/2 hepatitis C who arrived at the ED complaining of an abrupt onset of severe, sharp chest pain that radiates to his back, pain score of 10/10. On physical exam, the patient is hunched over clenching his chest, diaphoretic, and wheezing. His vital signs include blood pressure (BP) 192/120 mmHg, heart rate (HR) 102 beats/minute, and respiratory rate (RR) 32 breaths/minute. Laboratory results reveal serum creatinine 6.4 mg/dL (baseline 0.7 mg/dL), AST 110 U/L, ALT 95 mg/dL. He is rushed for a chest computed tomography with angiography, which reveals an acute type A aortic dissection. What is the etiology of this patient’s hypertension? Is this classified as uncontrolled hypertension or hypertensive emergency? Finally, of all the anti-hypertensives in your institution, which would be the best agent for this patient?
Introduction
Hypertensive emergency is characterized by severe hypertension, defined as systolic blood pressure (SBP) >180mmHg or diastolic blood pressure (DBP) >120mmHg, with evidence of end-organ dysfunction. Hypertensive emergencies require prompt recognition and treatment with IV antihypertensives to prevent from potentially fatal sequalae. In contrast, hypertensive urgencies present with severe hypertension, SBP >180mmHg or DBP >120mmHg, but lack evidence of end-organ failure and therefore can often be managed with optimization of oral antihypertensives.As ED providers, you arebound to be faced with the critical patient presenting in a hypertensive emergency, where quick treatment decisions must be made in order to reverse or minimize the ensuing end-organ dysfunction. With a multitude of intravenous (IV) antihypertensives available, it canbequite painstaking to acutely navigate treatment options. Further complicating matters, the varying etiologies of hypertensive crises requires a tailored treatment approach, as there is no one size fits all medication for the varying presentations. Therefore, clinicians must assess patient-specific factors (organ dysfunction, age, vital signs) in conjunction with medication-related factors (pharmacokinetics and pharmacodynamics, hemodynamic and adverse effect profile) to guide medication selection.
If choosing the agent wasn’t enough to think about, the next series of questions is what dose is best to use? When do I titrate when there is no response, and by how much? In a perfect world, the ideal agent for the management of hypertensive emergency would be easy to administer, have a rapid onset, titratable with a predictable hemodynamic response, minimal adverse effects and easily accessible from a medication dispensing system. Unfortunately, there is no agent that fits all of these ideal characteristics (that is not otherwise cost-prohibitive.) With this in mind, this post will highlight some key clinical pearls and pitfalls with the available therapeutic armamentarium for hypertensive emergency, with the addition of informative infographics that can serve as handy “pocket guides” to aid in the decision making process for the next time you are faced with these acute cardiovascular emergencies.
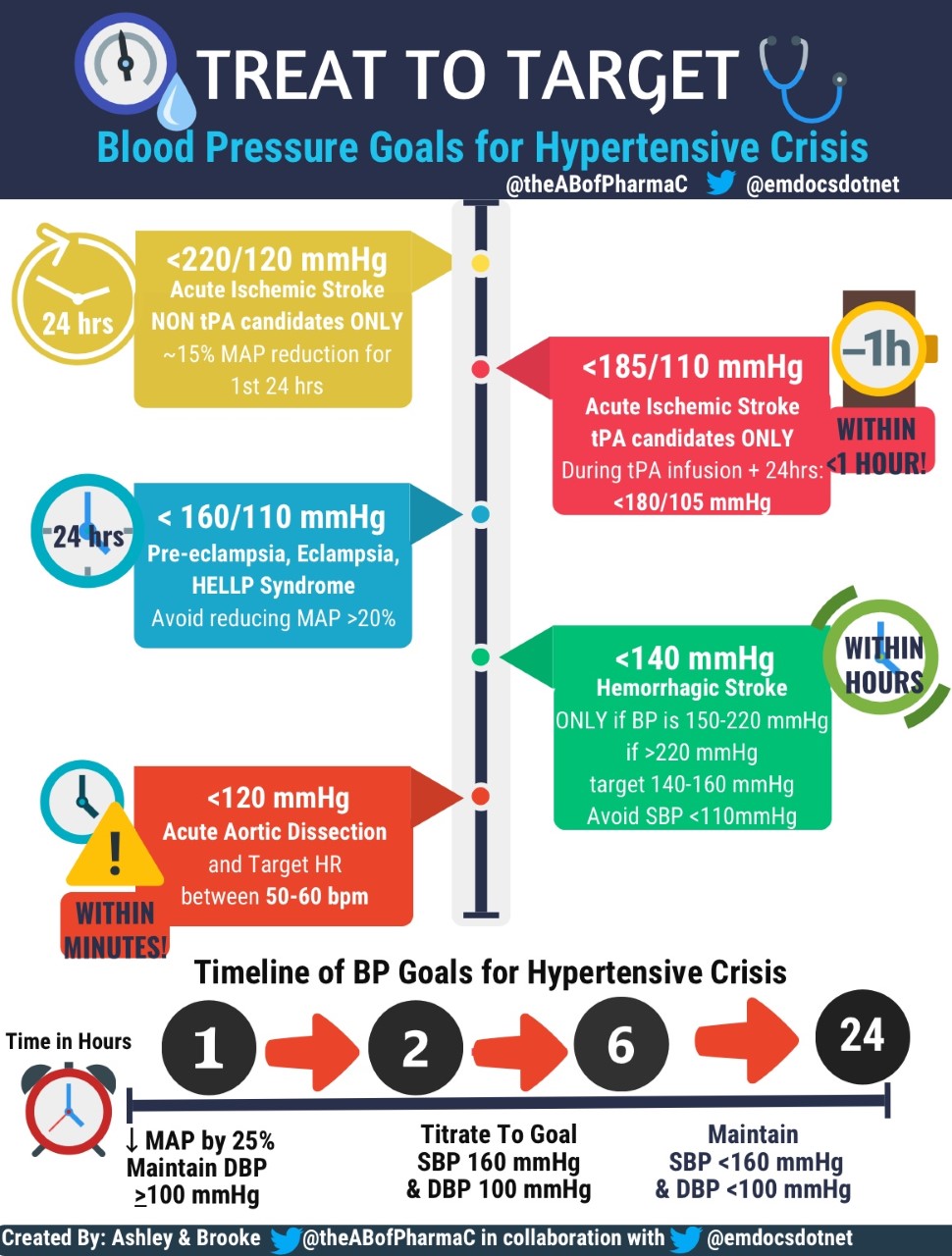


Calcium Channel Blockers
Calcium channel blockers (CCBs) remain the workhorses for management of hypertensive emergency due to their ideal pharmacokinetics, minimal adverse effects, and (relatively) predictable hemodynamic response.
Dihydropyridine calcium channel blockers inhibit L-type calcium channels on the vascular endothelium resulting in relaxation and vasodilation of the coronary, cerebral, and peripheral smooth muscle without affecting cardiac conduction.1 Now it’s important to specify, dihydropyridine CCBs are preferred over non-dihydropyridines (verapamil or diltiazem) as their selective afterload reduction is more effective in blood pressure (BP) control without compromising chronotropic activity (DI–think DI-LATOR).1 Nicardipine and clevidipine are the only dihydropyridine CCBs available IV, with a major benefit is their fixed dosing, continuous IV infusion (CIVI) administration, and wide applicability to the etiology of hypertensive emergency.
Indications
CCBs have minimal effects on intracranial pressure (ICP) and are endorsed by recent guidelines as the drug of choice for acute ischemic stroke, or hemorrhagic stroke.2,3,4 CCBs are also highly effective for afterload reduction and improve the myocardial oxygen supply in patients with an acute myocardial infarction (MI) or aortic dissection. However, the risk of reflex tachycardia, albeit small, should not be overlooked, as this may increase myocardial oxygen consumption and precipitate ischemia during acute coronary syndrome (ACS) in patients without adequate rate control.5
Contraindications
Reduction in afterload in advanced aortic stenosis can result in profound hypotension, therefore non-dihydropyridine CCBs should be avoided in this setting.6,7 Clevidipine is formulated as a lipid emulsion, which poses an inherent risk of hypertriglyceridemia, pancreatitis, bacterial contamination, and excess caloric intake (2kcal/mL), especially if used concurrently with propofol.8 Patients with soy or egg allergies or with disorders of defective lipid metabolism should not use clevidipine.1,7
Pearls
Nicardipine is the most commonly used agent, often due to familiarity and lower acquisition cost, however, clevidipine has several advantages that may favor its use in this setting. Clevidipine has an ultra-rapid onset of1-4 minutes vs nicardipines onset of 5-10 minutes, allowing for more frequent dose titrations to achieve the BP goal.4,5 Nicardipine’s pharmacokinetics are not as desirable as clevidipine, because the peak effect can be unpredictable (range 5-120 minutes)and an extended half-life of 2-6 hours.6 Too frequent dose titrations can lead to “dose stacking” and predispose patients to prolonged hypotensive episodes. To prevent accumulation, itis prudent to promptlyreduce the infusion rate of nicardipine once BPtarget is achieved.1 Clevidipine is devoid of these concerns as its ultra-rapid onset allows for more finite BP adjustments and short duration (5-15 minutes) makes adverse effects short lived and minimizes the risk of delayed complications.1,7 Clevidipine is a favorable agent for patients with organ dysfunction, as plasma esterases are responsible for its degradation.1,7 On the contrary, nicardipine undergoes hepatic metabolism via CYP3A4, CYP2C8, and CYP2D6, which places patients with cirrhosis at risk for prolonged hypotension due to delayed elimination.6,9
Pitfall
A frequently overlooked caveat to nicardipine infusions is the risk of volume overload. Nicardipine is commercially available as a premixed bag or vial prepared at a concentration of25 mg/250 mL.6 Assuming an infusion rate of 15 mg/hr, this would equal 150 mL/hr or up to 3.6 liters of fluid per day! Awareness of the excess volume is essential as volume overload can be detrimental in the setting of acutely decompensated heart failure (HF), pulmonary edema, or acute kidney injury. In these cases, one could consider clevidipine where the maximum volume limit is 1000 mL/24 hours.7,9
Take home point: Clevidipine has several advantages over nicardipine, however cost remains the limiting factor to its wide-spread use.
Beta-blockers
Pharmacology
Through blunting the catecholamine surge, IV β-blockers labetalol and esmolol rapidly reduce HR and cardiac output in states of increased sympathetic tone. Recall that labetalol is a non-selective blocker of β-1, β-2, and to a lesser extent the α1 receptor, resulting in a reduction in cardiac contractility and systemic vascular resistance (SVR).1 Labetalol has a higher affinity to β-receptors, though uniquely, this pharmacodynamic effect is formulation dependent, with a β:α ratio of 7:1 IV vs. 3:1 PO.1,9 In contrast, esmolol is an ultra-rapid acting dose-dependent, cardioselective to the β-1 receptor with minimal effect on SVR at doses <500mcg/kg/minute.10
Indications
β-blockers remain a cornerstone treatment option for acute aortic dissection, where urgent reduction in HR and SVR is warranted to prevent propagation of the dissection. The dual receptor blockade of labetalol permits its use as monotherapy for acute dissections, in contrast to esmolol which must be used WITH an arterial dilator for afterload reduction.11 Important to note, if sodium nitroprusside is used as a vasodilator, esmolol has a delayed onset of action compared to nitroprusside and therefore must be administered BEFORE to prevent reflex tachycardia and propagation of the dissection.12
Due to its negligible lipophilicity, labetalol has minimal placental transfer and is considered the drug of choice for pregnancy-induced hypertensive emergency.13 Similar to CCBs, labetalol maintains cerebral perfusion which makes it another “go-to” option for acute ischemic stroke. In addition, it is a convenient option in the ED because it is often easily accessible from pyxis machines as a premade syringe that is ready for quick administration as an IV bolus.
Contraindications
β-blockers should be avoided in acutely decompensated HF, flash pulmonary edema, and bronchospasm in uncontrolled asthma or chronic obstructive pulmonary disease (non-selective β-blockers) due to the risk of acute or worsening exacerbations.14 Esmolol is also preferred in asthma or chronic obstructive pulmonary disease patients due to its cardioselectivity and minimal effect on bronchodilation.8,14
Pearl
The higher cost acquisition of esmolol does not allow it to be as readily available in the ED. However, this agent is highly favored amongst many clinicians for unstable patients presenting with hepatic or renal dysfunction due to its organ independent elimination via erythrocyte plasma esterases.15 Of note patients with anemia may have decreased esterase activity and may have a prolonged half-life and delayed clearance.1
Pitfall
Labetalol can be administered as as low infusion of 1-2mg/minute after the initial bolus, however prolonged infusions are attended to acutely load patient prior to transitioning to PO labetalol and should be AVOIDED in hypertensive emergency. The long half-life of labetalol can lead to accumulation and has been associated with dangerous, potentially fatal cases of delayed hemodynamic compromise.16 Logically, we know the max cumulative dose of IV labetalol is 300 mg, therefore in an average 70 kg patient can be reached in as little as ~3-5 hours via an infusion at the recommended rate. The infusion is further flawed for acute BP management because it cannot be titrated more frequently than every two hours. Don’t let this dosing deceive you when transitioning to maintenance anti-hypertensive, switch to alternative agents such as nicardipine or esmolol infusions in this setting.17
For more information on this topic, refer to the phenomenal blog post on Atrium Cardiology Collaborative for pitfalls of IV labetalol infusions.
Take-home point: Esmolol is ultra-short acting beta-1 selective blocker that is preferred for critically ill patients with organ dysfunction. In contrast labetalol is longer acting, and serves as a preferred agent for pregnancy-related hypertensive emergency or acute ischemic stroke.
Nitroglycerin
Pharmacology
Nitroglycerin is a rapid acting selective VENO-dilator that primarily reduces preload, though at higher doses, mild afterload reduction can be seen.4,14
Indications
Nitroglycerin restores oxygen demand and coronary perfusion, making it an important preload reducer for acutely decompensated HF, acute pulmonary edema, sympathetic crashing acute pulmonary edema or acute coronary ischemia.4,9 In these settings, reductions in preload and pulmonary wedge pressure aids in rapid resolution of ischemic chest pain and venous congestion.2
Contraindications
Remember that nitroglycerin works predominantly through reduction in preload, therefore if administered to a volume-depleted or preload dependent patient, the potent venodilation can plummet the cardiac output, leading to profound hypotension, reflex tachycardia, and compromised in organ perfusion.2 If treatment is warranted, small fluid boluses should be considered to mitigate this effect.18 In the setting of ischemic chest pain, ruling out a right inferior wall MI is imperative prior to administration, because the reduction in venous return can decrease right atrial filling pressure, reductions in cardiac output, and potentiation of ischemia.18 Prior to nitroglycerin initiation, a comprehensive medication history should be completed to screen for vasodilators such as phosphodiesterase-5 inhibitors or riociguat due to the risk of fatal prolonged hypotension when used in combination.19
Pitfall
The labeled starting dose of nitroglycerin is 5-10 mcg/minute,however,using this standard dosing recommended is less than ideal for hypertensive emergencies. Keep in mind that patients who use sublingual nitroglycerin prior to ED arrival for their chest pain are receiving up to 400mcg in just one tablet.19 Once the patient arrives with recurrent chest pain and a hypertensive emergency secondary to ACS, starting at 5 mcg/minute is diminishing the dose response effect and will not provide nearly enough coronary dilatory effects to achieve symptom resolution. Dosing of the sublingual tablet is equal to ~100 mcg/minute when administered IV, and therefore clinicans should consider higher initial starting doses (~50mcg/minute) with rapid dose escalation to achieve improvements in chest pain.20
Pearl
Multiple studies have used boluses with high doses of IV nitrates (2 mg IV push)every 5 minutes to allow for a more rapid preload reduction and prevent further respiratory decompensation. The use of higher initial doses has been demonstrated to reduce the need for endotracheal intubation, bilevel positive airway pressure (BiPAP), and ICU admission and minimal increased risk of hypotension.21,22
Headaches are the most common, dose-limiting adverse effect of nitroglycerin which can be mitigated by slowing the infusion and titrating at longer intervals. In cases where headaches continue despite these measures, acetaminophen can be used for symptom relief, as agents such as nonsteroidal anti-inflammatory drugs or triptans can adversely affect BP and potentiate coronary ischemia.23 A well-known phenomenon with nitroglycerin is the development of tachyphylaxis. Though not an immediate issue in the ED, it is important to consider if a patient needs prolonged treatment because without a nitrate-free interval the onset of tolerance can be seen as early as 24-48 hours.8 Titration to high doses will not overcome, but worsen tachyphylaxis, so a change in therapy should be warranted.1,4
Take home point: Nitroglycerin is best utilized as an adjunct therapy for preload reduction, however note higher than recommended starting doses of IV nitroglycerin are often required to achieve the same veno-dilatory properties as sublingual nitroglycerin.
Nitroprusside
Historically, nitroprusside was extensively used due to its potent venous and arterial dilating properties that allowed for a wide applicability to various hypertenisve emergencies. However, given safety and administration concerns, nitroprusside has largely fallen out of favor. The rapid, profound, and precipitous drop in BP seen with nitroprusside has resulted in hypotension related fatalities, which warranted the FDA to add a black box warning to its package labeling.23 The unpredictable, yet potentially severe drop in BP warrants patients to receive ICU level care with continuous monitoring through a dedicated arterial line.24 Unfortunately this labor intensive monitoring is not feasible in the ED.
Contraindications
Nitroprusside dilates the large capacitance vessels in the central nervous system which can result in detrimental spikes in ICP. Therefore, nitroprusside should be avoided in hypertensive encephalopathy, acute ischemic, or hemorrhagic strokes.1,2 Nitroprusside should be avoided in the setting of an acute MI or patients with ischemic heart disease.24 This theory hypothesizes that nitroprusside can exacerbate coronary ischemia secondary to its preferential vasodilation of NON-ischemic arteries, which favors redistribution of oxygenated blood away from the ischemic tissue and towards non-ischemic tissues.24 The shunting of oxygenated blood away from the acutely ischemic areas can exacerbate myocardial necrosis and increase overall mortality in this population.25
Cyanide and thiocyanate toxicity are hallmark, yet potentially dangerous complications of nitroprusside administration.1,9 Nitroprusside is ~44% cyanide by weight and when liberated into the plasma, cyanide is metabolized in the liver to thiocyanate. Under normal circumstances, the body can detoxify up to 50 mg of nitroprusside; however, patients with cirrhosis or who are malnourished have a reduced metabolic reserve to detoxify cyanide and are at high risk for toxicity.9 Thiocyanate is excreted by the kidneys and can accumulate in patients with acute or chronic renal failure, resulting in toxicities such as muscle spasms, seizures, or even coma. Although these toxicities may be less relevant in the acute setting, it is prudent to recognize the dose limit is 10mcg/minute for NO GREATER than 10 minutes, to reduce toxicity, especially in patients at high risk of accumulation.1,2
Take home point: Given the advent of newer, safer therapies, nitroprusside should be reserved as a last-linetreatment for hypertensive emergency when alternatives agents cannot be employed.
Drugs NOT to use for hypertensive emergencies:
Hydralazine and diuretics have limited value, and in-fact a potential for harm if used for hypertensive emergency. Hydralazine remains extensively used despite its suboptimal pharmacokinetics and the substantial, unpredictable antihypertensive response.2 After IV administration, there is an initial latency period of up to 15 minutes, followed by sporadic, progressive and often precipitous decline in BP.26 Furthermore, don’t let the half-life of 3 hours deceive you, as the pharmacologic response to hydralazine can persist for up to ~8-12 hours.27,28 Taken together, the unpredictable response and difficulty in titration, its use is best avoided in the management of hypertensive crises. Outside of scleroderma renal crisis for those unable to take an oral angiotensin converting enzyme inhibitor (ACEi), enalaprilat is best avoided due to its delayed, unpredictable onset, long duration of action (~12-24 hours), and associated risk of acute kidney injury and hyperkalemia.2 There is also a high degree of interpatient variability to the hemodynamic response, especially in African Americans due to the low renin concentrations observed in this population that minimizes the therapeutic benefit from ACE inhibition.29 In fact, they may be at greater risk of harm with ACEi due to a heightened incidence of angioedema.30 All ACEi are contraindicated during all trimesters of pregnancy due to oligohydramnios (decreased production of amniotic fluid).31 Finally, diuretics have virtually no role in hypertensive emergencies with the exception of use as adjuncts in pulmonary edema.2 Most patients with hypertensive emergency are actually volume depleted and the administration of diuretics can cause a synergistic decline in BP and can compromise organ perfusion.2
Finally, we will leave you with some pearls related to medication-induced hypertension. Medications are an important modifiable, though often unrecognized precipitant of hypertensive crises. A thorough medication history, addressing both present and past medication use, over-the-counter, and dietary supplements should be part of the initial evaluation. Some common offenders that should be on the top of every ED clinicians’ radar are medications that activate the renin angiotensin aldosterone system (nonsteroidal anti-inflammatory drugs, glucocorticoids, licorice) direct vasoconstrictors (cocaine, amphetamines, monoamine oxidase inhibitors), vascular endothelial growth factor inhibitors and erythropoietin stimulating agents.32 Hypertensive emergency can also be precipitated by abrupt WITHDRAWAL of clonidine or β-blockers, due to the rebound catecholamine surge. Resumption of these therapies will blunt the catecholamine response, and if therapy must be discontinued, a slow tapering schedule should be used or if treatment is continued, switch to an extended release product will improve patient compliance to hopefully withdrawal and rebound hypertension.
Case Conclusion:
Our first case of our patient with acute aortic dissection warrants urgent medical therapy to alleviate the aortic wall stress and prevent propagation. Initial therapy should be targeted towards ventricular rate control, preferably with beta-blockade. Our patients liver disease may pose a bit of a challenge, however, the organ-independent elimination of esmolol would make it the optimal agent in this setting. If additional BP control is warranted, afterload reduction with arterial vasodilators such as CCBs can be used, with consideration to lower doses of nicardipine due to the risk of accumulation in hepatic impairment.
Take Home Points:
- Hypertensive emergency is a medical emergency that requires prompt treatment with IV antihypertensives in attempts to reverse the ensuing multi-organ system failure.
- Calcium channel blockers and/or beta blockers remain the preferred treatment option for most cases of hypertensive emergency, with vasodilator serving as adjunctive agents when preload reduction is warranted.
- Although nicardipine is most commonly employed CCB, clevidipine has been increasingly in popularity due to its improved pharmacokinetics, ease of titratability, and lower risk of volume accumulation (max 1000mL/24hrs).
- Esmolol is ultra-short acting beta-1 selective blocker that is preferred for critically ill patients with organ dysfunction. In contrast labetalol is longer acting, and serves as a preferred agent for pregnancy-related hypertensive emergency or acute ischemic stroke.
- Labetalol should only be administered as an IV push for acute BP control NOT an IV infusion due to the risk of accumulation and prolonged toxicity.
- Nitroglycerin and nitroprusside are vasodilators that assist with preload reduction in states of congestion such as acute heart failure or pulmonary edema.
- Consider higher doses of nitroglycerin than the labeled dosing for patients with ACS, acute heart failure with pulmonary edema, or sudden cardiovascular acute pulmonary edema, to provide the necessary preload reduction for symptom control.
- Nitroprusside has significant toxicities (cyanide and thiocyanate poisoning) and requires ICU level monitoring due to the profound hemodynamic response, therefore should be reserved as salvage therapy for acute BP control in the ED.
- Hydralazine, enalaprilat, diuretics, and nifedipine have limited utility and potentially harmful consequences if used in the setting of hypertensive emergency.
- Don’t forget to have drug-induced hypertension on your differential, including drug withdrawal from b-blockers or clonidine.
- When in doubt, don’t be afraid to consult your friendly pharmacist!
More FOAM Resources
- EM in 5: Hypertensive Emergency Treatment
- EM@3AM: Hypertensive Emergency
- Pulmonary Hypertensive Crisis
- Hypertensive Crisis in Kids
- Scleroderma Renal Crisis
References:
- Rhoney D, Peacock WF. Intravenous therapy for hypertensive emergencies, part 1. Am J Health Syst Pharm. 2009;66(15):1343-52.
- Varon J, Marik PE. Clinical review: the management of hypertensive crises. Crit Care. 2003;7(5):374-84.
- Hemphill JC III, Greenberg SM, Anderson CS, et al. Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2015;46:2032-60.
- Watson K, Broscious R, Devabhakthuni S, Noel ZR. Focused Update on Pharmacologic Management of Hypertensive Emergencies. Curr Hypertens Rep. 2018;20(7):56.
- Ozawa Y, Hayashi K, Kobori H. New Generation Calcium Channel Blockers in Hypertensive Treatment. Curr Hypertens Rev. 2006;2(2):103–111.
- Cardene IV (0.1 mg/mL) (nicardipine) [prescribing information]. Bedminister, NJ: EKR Therapeutics; January 2019.
- Cleviprex (clevidipine) [prescribing information]. Cary, NC: Chiesi USA, Inc; October 2017.
- Kaur H, Nattanamai P, Qualls KE. Propofol and Clevidipine-induced Hypertriglyceridemia. Cureus. 2018;10(8):e3165.
- Benkin S. Hypertensive emergencies. Medical Issues in the ICU, Critical Care Self Assessment Program. American College of Clinical Pharmacy. Book 1:2018.
- Lowenthal DT, Porter RS, Saris SD, Bies CM, SlegowskiMB, Staudacher A: Clinical pharmacology, pharmacodynamics and interactions with esmolol. Am J Cardiol 1985, 56:14F-18F.
- Gupta PK, Gupta H, Khoynezhad A. Hypertensive Emergency in Aortic Dissection and Thoracic Aortic Aneurysm-A Review of Management. Pharmaceuticals (Basel). 2009;2(3):66–76. Published 2009 Sep 28.
- Suzuki T, Eagle KA, Bossone E, Ballotta A, Froehlich JB, Isselbacher EM. Medical management in type B aortic dissection. Ann Cardiothorac Surg. 2014;3(4):413-7.
- Webster LM, Webb AJ, Chappell LCWhat is the evidence for using labetalol as a first-line treatment option for hypertension in pregnancy?Drug and Therapeutics Bulletin 2018;56:107-111.
- Papadopoulos DP, Sanidas EA, Viniou NA, et al. Cardiovascular hypertensive emergencies. Curr Hypertens Rep. 2015;17(2):5.
- Gray RJ, “Managing Critically Ill Patients With Esmolol: An Ultra-Short Acting Beta-Adrenergic Blocker,” Chest, 1988, 93(2):398-403.
- Fahed S, Grum DF, Papadimos TJ.Labetalol infusion for refractory hypertension causing severe hypotension and bradycardia: an issue of patient safety. Patient Saf Surg. 2008;2:13.
- Reed B. No Love Lost for Intravenous Labetalol Infusions: Risks of Prolonged Use. Atrium Cardiology Collaborative Blog. 17 October 2017.
- Martinez JA. Complications of acute myocardial infarction. Emergency Medicine, July 2003.
- Nitroglycerin in 5% Dextrose Injection (nitroglycerin) [prescribing information]. Deerfield, IL: Baxter; August 2016.
- Bussman WD, et al. Effect of sublingual nitroglycerin in emergency treatment of severe pulmonary edema. Am J Cardiol 1978;41(5):931-936.
- Levy P, Compton S, Welch R, et al. Treatment of severe decompensated heart failure with high-dose intravenous nitroglycerin: a feasibility and outcome analysis. Ann Emerg Med. 2007;50(2):144-55.
- Hsieh YT, Lee TY, Kao JS, Hsu HL, Chong CF. Treating acute hypertensive cardiogenic pulmonary edema with high-dose nitroglycerin. Turk J Emerg Med. 2018;18(1):34-36.
- Demaagd G. The pharmacological management of migraine, part 1: overview and abortive therapy. P T. 2008;33(7):404-16.
- Nipride RTU (sodium nitroprusside) [prescribing information]. Lenoir, NC: Exela Pharma Sciences LLC; July 2018.
- Hottinger, D. G., Beebe, D. S., Kozhimannil, T., Prielipp, R. C., & Belani, K. G. (2014). Sodium nitroprusside in 2014: A clinical concepts review. Journal of anaesthesiology, clinical pharmacology, 30(4), 462–471.
- Mann T, Cohn PF, Holman LB, et al. Effect of nitroprusside on regional myocardial blood flow in coronary artery disease. Results in 25 patients and comparison with nitroglycerin. Circulation 1978;57:732-8.
- Ludden TM, Shepherd AM, McNay JL, Lin MS: Hydralazine kinetics in hypertensive patients after intravenous administration. Clin Pharmacol Ther 1980, 28:736-742.
- O’malley K, Segal JL, Israili ZH, et al. Duration of hydralazine action in hypertension. Clin Pharmacol Ther. 1975;18(5 Pt 1):581-6.
- Hirschl MM, Binder M, Bur A, et al. Impact of the renin-angiotensin-aldosterone system on blood pressure response to intravenous enalaprilat in patients with hypertensive crises. J Hum Hypertens. 1997;11(3):177-83.
- Brown NJ, Ray WA, Snowden M, et al. Black Americans have an increased rate of angiotensin converting enzyme inhibitor-associated angioedema. Clin Pharmacol Ther. 1996;60:8-13.
- Vasotec (enalapril maleate) i.v. package insert. Bridgewater, NJ: Biovail Pharma- ceuticals; 2007.
- Aronow WS. Drug-induced causes of secondary hypertension. Ann Transl Med. 2017;5(17):349.
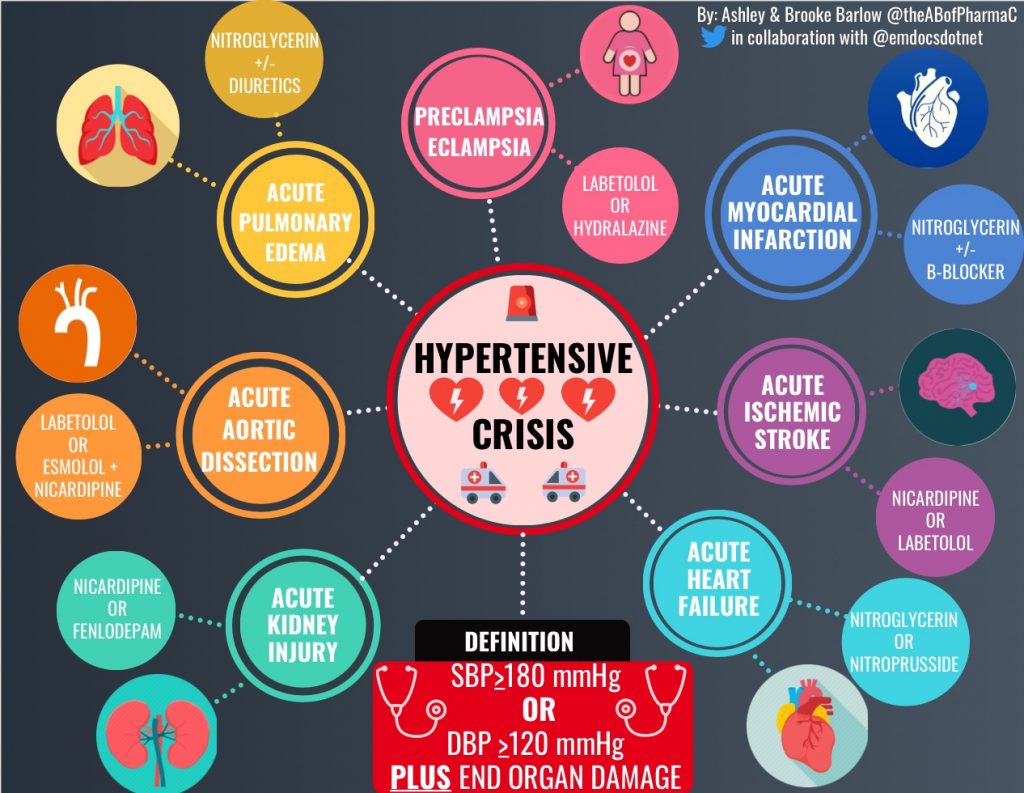
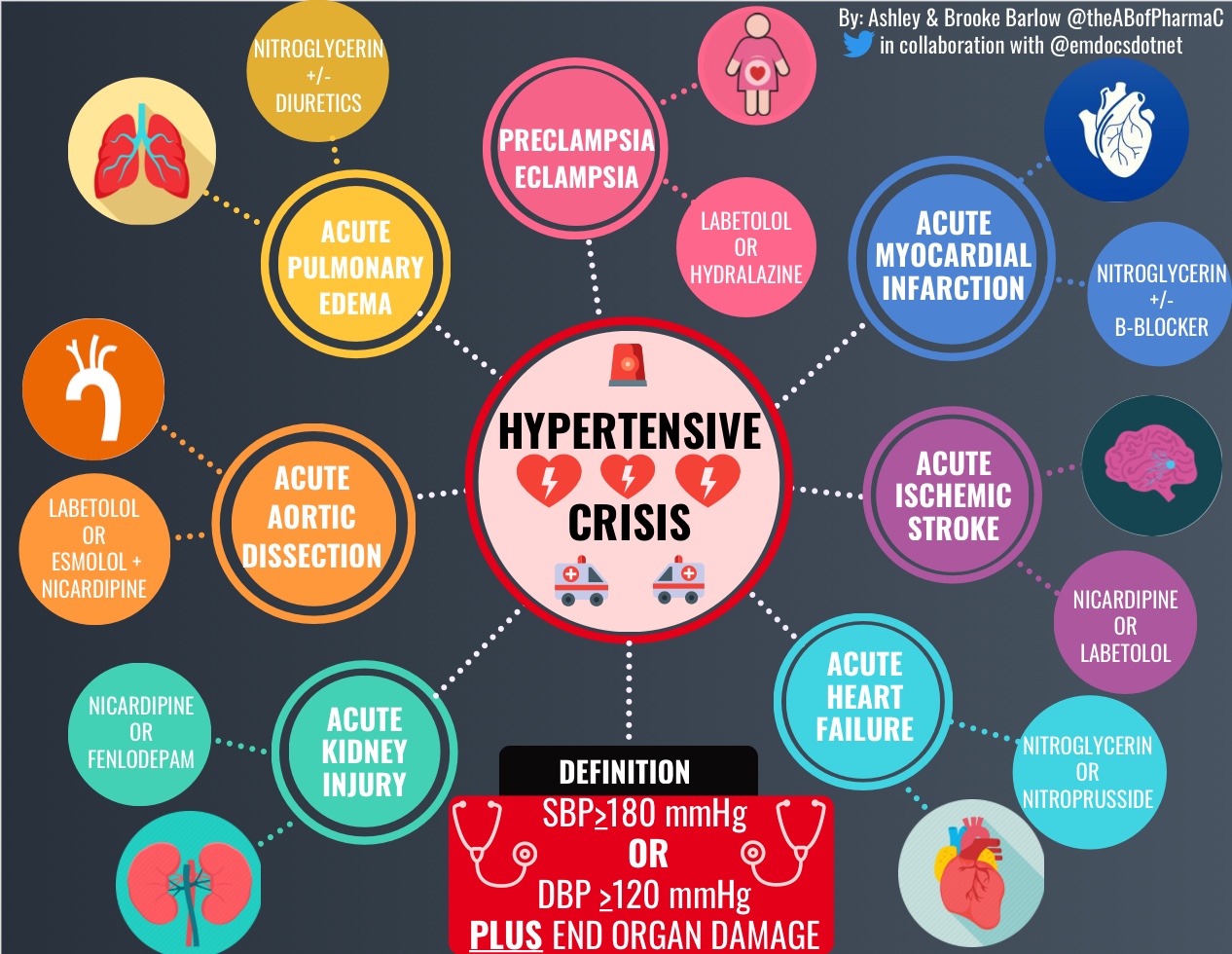






2 thoughts on “Hypertensive Emergency: Pearls and Pitfalls for the ED Physician”
Pingback: Episode 8: Transition from Pharmacy School to Residency with the Pharmacy Twins Ashley & Brooke Barlow - The Pharm So Hard Podcast
Pingback: Deep Dive – Round 23 – EM Clerkship, LLC