Author: Brit Long, MD (@long_brit, EM Attending Physician at SAUSHEC, USAF) // Edited by: Alex Koyfman, MD (@EMHighAK, EM Attending Physician, UTSW / Parkland Memorial Hospital)
A 24-year-old male arrives by EMS with a GCS of 5 after a severe MVC. He was ejected from the vehicle, and EMS placed a C-collar, followed by positioning on a backboard for transport. Bilateral IV’s were placed, and 1 L NS was started. They did not intubate him in the field, as they were 4 minutes from your hospital. What are your priorities in evaluation and management of this patient? What are the pearls and pitfalls in resuscitating the sick neurotrauma patient?
Neurotrauma, particularly traumatic brain injury (TBI), is a significant cause of death around the world and the leading cause of death in patients age 1 to 45 years.1-5 Approximately 78% of patients are managed in the emergency department (ED), with males and young adults the two primary populations affected.1.2 Traumatic neurologic injury not only causes an initial primary injury, but it is associated with several secondary insults.1-6
Some pathophysiology…
Cerebral perfusion pressure (CPP) is defined by the mean arterial pressure (MAP) minus the intracranial pressure (ICP). ICP is a function of the brain parenchyma, blood, and cerebrospinal fluid.2,7-12 The key is that an increase in one requires a decrease in another.2,7-12 Once compensatory methods are exhausted, further volume leads to drastic increases in ICP. Cerebral perfusion pressure is related to ICP, and increase in ICP may decrease cerebral perfusion. The ultimate goal of resuscitation and management of the neurotrauma patient is to ensure normal ICP, while preserving cerebral blood flow and perfusion.2,8,11,12
Are there physiologic goals?
Hypotension is associated with increased morbidity and mortality. A CPP goal of 50 to 70 mm Hg should be used, with SBP of at least 100 mm Hg (ages 50-69) and 110 mm Hg for 15-49 years and > 70 years, per the Brain Trauma Foundation.2,8,11-13 Otherwise, a MAP of 70-80 mm Hg is advised.5 Avoiding cerebral hypotension is recommended, though aggressive CPP targeting is not associated with improved outcomes.13-15 Hypoxemia also results in a significant increase in mortality. Key targets of resuscitation are shown below.
Goal Physiologic Parameters:5,11,12
-Pulse oximetry > 94%, less than 100% (avoid hypoxemia and hyperoxemia)
-PaCO2 35-45 mm Hg
-SBP > 100 mm Hg (ages 50-69), and > 110 mm Hg (ages 15-49, > 70 years)
-pH 7.35-7.45
-ICP < 20 mm Hg
-Glucose 80-180 mg/dL
-CPP > 60 mm Hg
-Serum Na 135-145, (Hypertonic saline goal is 145-160)
-INR < 1.4
-Platelets > 75 x 103/mm3
-Hgb > 8 mg/dL
What are several dangerous secondary injuries?
Neurotrauma begins a cascade that may cause further cell death.8,11,12 The following are significant secondary injuries, all which increase adverse outcomes. Awareness of these injuries is necessary for prevention.5,8,11-15
– Hypotension: 30% of patients, resulting in higher likelihood of poor outcome (OR 2.67).2,8,11-15
– Hypoxia: 50% of patients, resulting in higher likelihood of poor outcome (OR 2.14).2,8,11-15
– Hyperoxia: PaO2 levels above 300-470 mm Hg are associated with worse outcome.2,8,11-15
– Fever: Elevated temperature worsens morbidity by secondary brain injury aggravation.2,13
– Coagulopathy: Associated with the traumatic event and may cause worsening of the neurologic injury and death. Acute TBI may cause coagulopathy itself through tissue factor and phospholipid release.16
– Glucose: Hyper- and hypoglycemia are predictors of poor neurologic status.2,8,11-15
Pearls in Evaluation and Management
Focus on airway, breathing, circulation, disability, and exposure in the primary survey, with spinal precautions.2,11,17-20 Avoid secondary complications, and other markers of worse outcome include poor GCS motor score, pupillary dysfunction, and increased ICP. Abnormal pupillary response and altered motor function are markers for severe brain trauma, as is posturing. Decorticate posturing (arm flexion and leg extension) is due to injury above the midbrain, and decerebrate posturing (arm extension and internal rotation, wrist and finger flexion, leg internal rotation and extension) is a sign of more caudal injury involving the midbrain.11,12,17 Abnormal pupils, decreased mental status, abnormal GCS, penetrating injury, abnormal motor status, and severe injury require neuroimaging. Severe neurotrauma requires consultation with neurosurgery.2,11,12,17 This post will focus on the hypotensive patient with neurologic injury.
ED Considerations:
-Maintain spinal precautions
-Conduct primary and secondary surveys; address life-threatening injuries
-Advanced airway management may be needed for airway protection, hypoxia, and control of ventilation
-Obtain rapid IV access
-Optimize oxygenation, blood pressure, and ventilation
-Target oxygen saturation > 94%, with systolic blood pressure > 100-110 mm Hg
– Focused neuro exam: GCS, motor function, and pupillary function
-Obtain head CT noncontrast
-Any sign of worsening neurologic status warrants hyperosmolar therapy
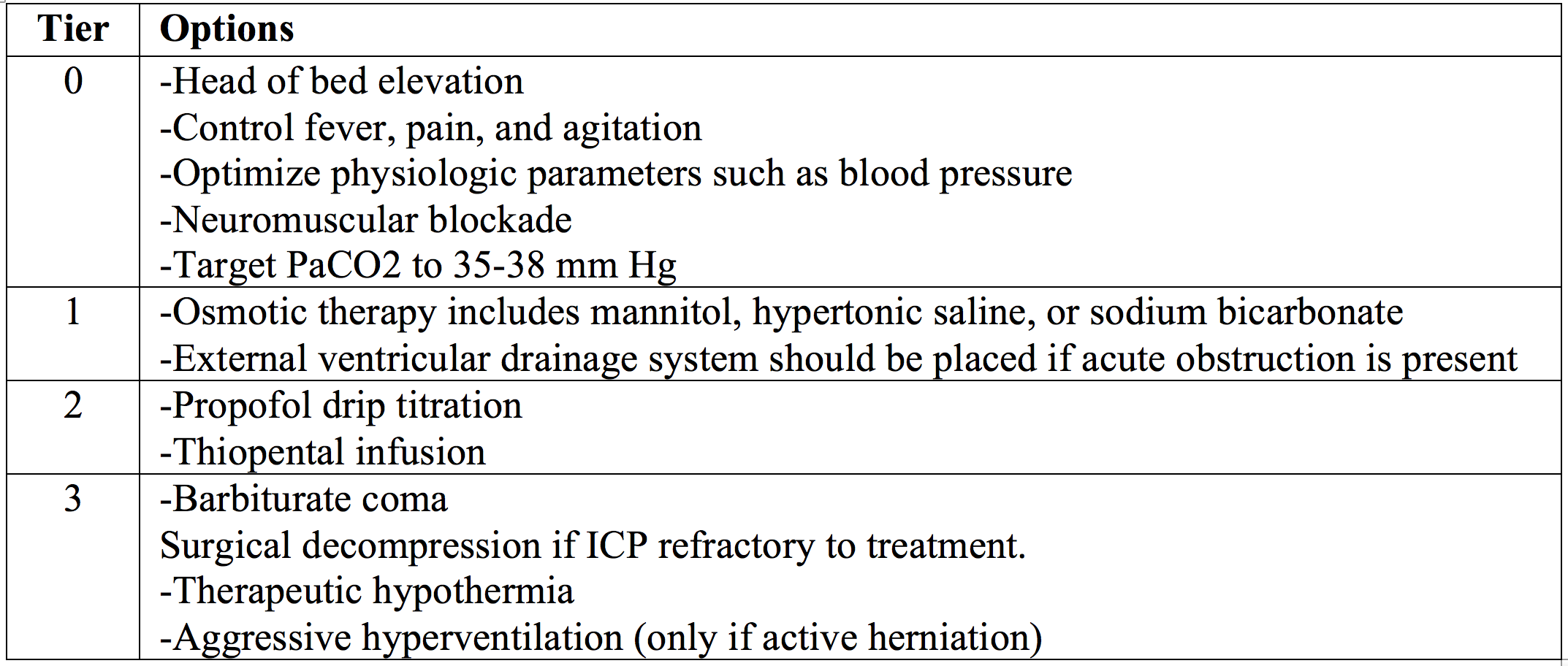
Tiers of ICP management, per the Brain Trauma Foundation, are shown below.11,12,17 We will discuss pearls and subtleties for these therapies, but first, let’s discuss airway.
How should the airway be managed?
Airway protection and blood pressure support for severe neurotrauma are priorities, and first pass success is vital. Rapid sequence intubation with in-line stabilization of the cervical spine may be necessary.2,11,18-20 Preoxygenation is important to avoid desaturation, so start your NO DESAT interventions early.21,22
Intubation Considerations:
-Preparation: Proper positioning, preoxygenate, and use apneic oxygenation with nasal cannula, facemask, or noninvasive positive pressure ventilation
-Elevate head of bed to improve CPP and decrease aspiration
-Premedication regimens are controversial. Fentanyl at 2-5 micrograms/kg IV or esmolol 1.5mg/kg IV may decrease catecholamine surge and control the hemodynamic response to intubation
-Induction agent may include ketamine (does not adversely affect patients with neurotrauma) and etomidate – these agents have less hemodynamics effects
-Propofol has neuroprotective effects, but hypotension may occur
-Post intubation analgesia and sedation are essential – have your drips ready to go at the time of intubation
Pretreatment
Up to 80% of patients may experience a hypertensive response to laryngoscopy or suctioning, and lidocaine was initially thought to blunt this. However, lidocaine has not demonstrated ability to reduce ICP or improve neurologic outcome.18-20,23 Fentanyl at doses of 2-5 micrograms/kg IV prior to intubation can reduce the hyperdynamic response to intubation, as can esmolol at 1.5 mg/kg IV. However, esmolol should be avoided in patients with hypotension, hemorrhagic shock, or signs of multiple trauma.18-20,24
Induction
Traditionally, ketamine was contraindicated for induction in intubation in TBI, but literature suggests that not only is it safe, but it may be beneficial. Ketamine improves cerebral blood flow, and evidence suggests it does not raise ICP.19,25 Propofol has high lipid solubility and rapid onset of action that can reduce ICP and oxidative stress. However, it may cause hypotension.19,20,26 Etomidate may result in less hypotension and cardiac dysfunction. It can reduce ICP and maintain CPP, but it also may lower the seizure threshold and increase the risk of vomiting and myoclonic movements.19,27 The key for an induction agent is utilizing lower doses in patients with hypotension, as any agent at full dose will worsen hypotension.
Paralysis
Paralysis is essential to improve first pass success including succinylcholine or rocuronium. Succinylcholine allows faster time to recovery and assessment of neurologic status, as rocuronium will last longer. Ultimately, the choice of paralytic rests on the physician, as both are safe and efficacious.19,20 However, defasciculating doses of paralytics such as succinylcholine or pancuronium are not beneficial and do not reduce ICP.19,20
Post Intubation
Post intubation sedation and analgesia are an absolute must.18-20 An intubated patient with inadequate analgesia and sedation may experience increased ICP due to sympathomimetic response.24 The key is to order your post intubation meds at the same time as your paralytic and induction agent.18-20 Analgesics including fentanyl and remifentanil are fast and predictable. Morphine and hydromorphone may accumulate with prolonged infusion, though they do have longer duration of action. Sedative medications include propofol, which possesses fast onset and offset, allowing for repeat neurologic assessment. If used keep close tabs on blood pressure. Infusion, rather than bolus doses, will mitigate the risk of decreasing BP. Benzodiazepines can be used, though they can reduce BP and respiratory status. However, they do reduce cerebral blood flow and ICP.28 Unfortunately, tolerance may develop, and reassessment is difficult due to metabolite accumulation. The new kid on the block, dexmedetomidine, is a selective alpha-2 receptor agonist with anxiolytic and sedative effects.19,29 Hypotension and bradycardia are the most common side effects, most commonly seen with bolus dosing. It may reduce ICP, though further study is needed.
Avoid hypoxia post intubation, targeting O2 saturation 94%-98%. Hyperoxia with PaO2 greater than 300-470 mm Hg is discouraged due to worse outcomes.13,18-20 Unless actively herniating, hyperventilation is not recommended. PaCO2 levels of 35 to 45 mm Hg, or end-tidal CO2 30 to 40 mm Hg, are your goals.2,11,18
What about hypotension?
Hypotension increases mortality two-fold, specifically any SBP drop less than 90 mm Hg.2,11-15,19 The polytrauma patient with head trauma can be difficult. These patients are often hypotensive, and permissive hypotension is not recommended. Target a SBP of at least greater than 100 mm Hg (for patients 50-69 years of age) or 110 mm Hg (for those 15-49 years and > 70 years).11 Keep in mind that ATLS states a MAP > 80 mm Hg in patients with severe TBI is reasonable.5 Start first with fluids, specifically normal saline or blood.2,11,18 Avoid hypo-osmotic fluids, which can increase cerebral edema and ICP.30 Albumin is associated with higher mortality, which was demonstrated in the SAFE trial in TBI patients.31 Literature suggests no difference between normal saline and HTS for patients with no signs of herniation.30
Wait, what’s this about neurogenic shock?
Neurogenic shock occurs with injury above T6 and is a form of distributive shock. It is caused by loss of sympathetic tone.32-35 Patients may show low HR and BP; however, hypovolemic shock must first be ruled out in the trauma patient. Spinal shock is different, with loss of sensation and motor function below the spinal cord injury. Reflexes are often depressed below the injury level.32-35
Are vasopressors needed?
A SBP of at least 100-110 mm Hg is recommended by the Brain Trauma Foundation, as hypotension increases morbidity and mortality.11 Specifically in neurogenic shock with hypotension and bradycardia, fluids and vasopressors will be needed.32,33 Loss of sympathetic tone is common within the first week of injury.6,32-35 A goal MAP of 85 mm Hg is recommended by the AANS and CNS in neurogenic shock.35 Dopamine, norepinephrine, or phenylephrine may be used. Norepinephrine will increase afterload and inotropy, needed with the loss of sympathetic tone. Phenylephrine will improve vascular tone and can be used in patients who are not bradycardic.6,33-35
What hyperosmolar therapies are available?
Hyperosmolar therapy is a foundation of management. These measures can reduce ICP and improve cerebral blood flow.2,12,17,18 Hyperosmolar agents should be used with signs of increased ICP, pupillary change, decrease in GCS > 2 points, or posturing.2,12,17,18 Agents include 20% mannitol 0.25-1 g/kg IV as a rapid infusion over 5 minutes or 3% NaCl 150 ml IV over 10 minutes (HTS 23.4% 30mL can be used through central line).2,11,17,18,30,36-38 We will discuss these agents further, but keep in mind that if hypotension is a concern, HTS may be a better resuscitation fluid.5,17,18 For ICP reduction, a 2015 meta-analysis finds no difference in neurologic outcome or mortality between mannitol and HTS.30
1. Mannitol
Mannitol is administered as a 20% solution.2,30,36-38 This solution deforms RBCs and decreases blood viscosity, improving cerebral blood flow.36-38 A major consideration is autoregulation and the presence of intact Blood-Brain-Barrier, as if these are intact, ICP will decrease. If not intact, mannitol may worsen outcomes.11,18,30 Rebound increases in ICP can occur, and the solution may cause renal injury through excess diuresis. IV fluids are necessary when providing mannitol, along with Foley catheter.11,17,18
2. HTS
HTS concentrations range from 2% to 23.4%, which also improves cerebral blood flow and reduces parenchymal water content.2,11,12,17 HTS can improve blood pressure as a volume expander. The risk of rebound ICP is less than that of mannitol.2,11,17,18,30 The most common side effect of HTS is hyperchloremic metabolic acidosis.2,18,30
What about ocular US for increased ICP?
Ultrasound can measure optic nerve sheath diameter (ONSD), which correlates closely with ICP.18,39,40 The normal optic nerve sheath is up to 5 mm in diameter, and ONSD will increase with elevated ICP. ONSD should be measured 3 mm posterior to the globe for both eyes, with an average between measurements. Values greater than 5 mm predict increased ICP, with sensitivity and specificity greater than 90%.39,40
When is surgery required?
Surgical management of TBI may be needed to repair depressed skull fracture or evacuate intracranial mass (such as blood).2,11,17,18 Decompressive craniectomy is indicated for refractory intracranial hypertension; however, decompressive craniectomy may not improve functional outcome while decreasing ICP, as shown in the DECRA trial.41-45 Ultimately, your neurosurgeon will make this decision, but they must be on board early in the patient’s management.11,45
Is hypothermia effective in neurotrauma?
Studies have not found an improvement in mortality or neurologic status with hypothermia in neurotrauma.2,11,18,46,47 However, hypothermia can reduce intracranial hypertension.46,47 Targeted management of temperature reduces cerebral metabolic rate and release of excitatory neurotransmitters, but more study is required.11,46,47
Should you reverse coagulopathy with traumatic intracerebral hemorrhage?
Coagulopathy is common, as close to 1/3 of patients with TBI display a coagulopathy, due to patient medication or release of tissue factor causing consumptive coagulopathy.11,16 Coagulation panel and TEG can be helpful, with reversal dependent on results and patient medication. Vitamin K, PCC/FFP, and novel antidotes can be utilized.11,48-50 We will not go into detail here on reversal. However, keep in mind that platelet transfusion in patients on antiplatelet medication with ICH may be harmful per the recent PATCH trial.51
What is the role of pharmacologically-induced coma?
Barbiturates can be used to reduce ICP, if refractory to other treatment, through suppression of cerebral metabolism, modification of vascular resistance, and decrease of neuronal excitotoxicity.2,18,52,53 However, barbiturates have significant side effects, as one in four experiences hypotension.2,11,53 A 2012 Cochrane Review found no change in outcomes for severe TBI with barbiturate coma.53
Corticosteroids used to be recommended, but what about today?
The CRASH trial suggests worse outcomes and increased mortality for patients with TBI given steroids.54-57 In the setting of spinal cord injury, the American Association of Neurologic Surgeons and Congress of Neurologic Surgeons do not recommend steroids.2,6,11
Should seizures be treated in neurotrauma?
Early posttraumatic seizures occur within 7 days of injury, with late seizures beyond 7 days.2,11,58,59 Seizures occur in up to 30% of TBI patients (50% of patients with penetrating injury).2,58,59 With seizures increasing ICP, any active seizure requires immediate treatment, with benzodiazepines first line. Prophylaxis on the other hand is more controversial. The presence of any risk factor including include GCS < 10, cortical contusion, any intracranial hematoma, depressed skull fracture, penetrating head injury, or seizure within 24 hours of injury requires prophuulaxis.11,58,59 Otherwise, prophylaxis is not recommended. Levetiracetam is equivalent to phenytoin for seizure reduction, and it is associated with less risk of side effect.58,59
Does tranexamic acid have a place in neurotrauma?
The CRASH-2 and MATTERs studies have suggested survival benefit within three hours of trauma for TXA.60,61 The CRASH-2 Intracranial Bleeding Study found a trend towards reduction in intracranial hemorrhage growth and lower mortality in patients with traumatic hemorrhage, with another study finding reduction in hemorrhage growth.60,62 CRASH-3 is underway, evaluating TXA in TBI specifically.63
Pitfalls in evaluation and management
Several medications have not demonstrated improvement in outcomes for TBI, including progesterone, magnesium, hyperbaric oxygen, and cyclosporine.2,11,64,65
Hyperventilation can reduce ICP for short periods.2,11,18,66,67 However, hyperventilation may result in secondary ischemia if used for prolonged periods and increases the risk of cerebral edema. Hyperventilation has demonstrated worse clinical outcomes in patients hyperventilated to PaCO2 less than 30 mm Hg for six hours up to five days.18,66,67 Mild hyperventilation can be used for acute worsening, but only for a short period, targeting PaCO2 30-35 mmHg.2,11,18
Key points
– Neurotrauma is common, as it is the leading cause of death in North America in those between ages 1 year to 45 years. Primary and secondary injuries result in severe morbidity and mortality.
– Neurotrauma includes head contusion, epidural hematoma, subdural hematoma, subarachnoid hemorrhage, diffuse axonal injury, skull fracture, and traumatic spinal cord injury.
– Cerebral perfusion pressure requires adequate cerebral blood flow.
– Evaluation and management in the emergency department entails initial stabilization and resuscitation while assessing neurologic status.
– Targeting mean arterial pressure, oxygen levels, and neurologic status are key components. ICP management should follow a tiered approach.
– Intubation of the patient with neurotrauma should be completed with several considerations.
– Hyperosmolar treatments include HTS and mannitol.
References/Further Reading
- Faul M, Xu L, Wald MM, et al. Traumatic brain injury in the United States: emergency department visits, hospitalizations and deaths 2002–2006. Atlanta (GA): Centers for Disease Control and Prevention, National Center for Injury Prevention and Control; 2010.
- Wan-Tsu WC, Badjatia N. Neurotrauma. Emerg Med Clin N Am 2014;32:889-905.
- Rutland-Brown W, Langlois JA, Thomas KE, Xi YL. Incidence of traumatic brain injury in the United States, 2003. J Head Trauma Rehabil 2006; 21:544.
- Stein DM, Roddy V, Mark J, Smith WS, Weingart SD. Emergency Neurological Life Support: Traumatic Spine Injury. Neurocrit Care 2012;17:S102-S111.
- ATLS Subcommittee; American College of Surgeons’ Committee on Trauma; International ATLS working group. Advanced trauma life support (ATLS®): the ninth edition. J Trauma Acute Care Surg. 2013 May;74(5):1363-6.
- Hadley MN, Walters BC, Aarabi B, et al. Guidelines for the management of acute cervical spine and spinal cord injuries. Neurosurgery 2013;72(Suppl 2): 1–259.
- Oddo M, Le Roux PD. What is the etiology, pathogenesis and pathophysiology of elevated intracranial pressure? In: Neligan P, Deutschman CS, editors. The evidenced based practice of critical care. Philadelphia: Elsevier Science; 2009.
- Swadron SP, LeRoux P, Smith WS, Weingart SD. Emergency Neurological Life Support: Traumatic Brain Injury. Neurocrit Care 2012;17:S112–S121.
- Bouma GJ, Muizelaar JP. Cerebral blood flow, cerebral blood volume, and cerebrovascular reactivity after severe head injury. J Neurotrauma 1992; 9 Suppl 1:S333.
- Bouma GJ, Muizelaar JP, Bandoh K, Marmarou A. Blood pressure and intracranial pressure-volume dynamics in severe head injury: relationship with cerebral blood flow. J Neurosurg 1992; 77:15.
- Carney N, Totten AM, O’Reilly C, Ullman JS, Hawryluk GWJ, bell MJ, et al. Guidelines for the Management of Severe Traumatic Brain Injury, Fourth Edition. Neurosurgery 2016;0(0):1-10.
- Stevens RD, Huff JS, Duckworth J, et al. Emergency neurological life support: intracranial hypertension and herniation. Neurocrit Care 2012;17(Suppl 1):S60–5.
- McHugh GS, Engel DC, Butcher I, et al. Prognostic value of secondary insults in traumatic brain injury: results from the IMPACT study. J Neurotrauma 2007; 24:287.
- Chestnut RM, Marshall LF, Klauber MR, et al. The role of secondary brain injury in determining outcome from severe head injury. J Trauma 1993;34(2):216-22.
- Marmarou A, Anderson RL, Ward JD, et al. Impact of ICP instability and hypotension on outcome in patients with severe head trauma. J Neurosurg 1991; 75(Suppl):S59–66.
- Harhangi BS, Kompanje EJ, Leebeek FW, Maas AI. Coagulation disorders after traumatic brain injury. Acta Neurochir (Wien) 2008; 150:165.]
- Emergency Neurological Life Support: Elevated ICP or Herniation. 2014. Available at http://enlsprotocols.org/files/ICP.pdf. Accessed 16 November 2016.
- Weingart S. EMCrit: Podcast 78 – Increased intra-cranial pressure (ICP) and herniation, aka brain code. Available at http://emcrit.org/podcasts/high-icp-herniation/. Accessed 16 November 2016.
- Bucher J, Koyfman A. Intubation of the Neurologically Injured Patient. JEM 2015;49(6):920-7.
- Seder DB, Riker RR, Jagoda A, Smith WS, Weingart SD. Emergency Neurological Life Support: Airway, Ventilation, and Sedation. Neurocrit Care 2010;17:S4-S20.
- Weingart SD, Levitan RM. Preoxygenation and prevention of desaturation during emergency airway management. Ann Emerg Med 2012;59:165–1751.
- Dixon BJ, Dixon JB, Carden JR, et al. Preoxygenation is more effective in the 25 degrees head-up position than in the supine position in severely obese patients: a randomized controlled study. Anesthesiology 2005;102:1110–5. discussion 1115A.
- Robinson N, Clancy M. In patients with head injury undergoing rapid sequence intubation, does pretreatment with intravenous lignocaine/lidocaine lead to an improved neurological outcome? A review of the literature. Emergency Medicine Journal : EMJ. 2001;18(6):453-457. doi:10.1136/emj.18.6.453
- Dahlgren N, Messeter K. Treatment of stress response to laryngoscopy and intubation with fentanyl. Anaesthesia 1981;36:1022–6.
- Cohen L, Athaide V, Wickham ME, Doyle-Waters MM, Rose NG, Hohl CM. The effect of ketamine on intracranial and cerebral perfusion pressure and health outcomes: a systematic review. Ann Emerg Med 2015;65:43–51.
- Rossaint J, Rossaint R, Weis J, et al. Propofol: neuroprotection in an in vitro model of traumatic brain injury. Crit Care 2009; 13:R61.
- Moss E, Powell D, Gibson RM, McDowall DG. Effect of etomidate on intracranial pressure and cerebral perfusion pressure. Br J Anaesth. 1979;51:347–52.
- Barrientos-Vega R, Mar Sanchez-Soria M, Morales-Garcia C, Robas-Gomez A, Cuena-Boy R, Ayensa-Rincon A. Prolonged sedation of critically ill patients with midazolam or propofol: impact on weaning and costs. Crit Care Med. 1997;25:33–40.
- Jakob SM, Ruokonen E, Grounds RM, et al. Dexmedetomidine vs. midazolam or propofol for sedation during prolonged mechanical ventilation: two randomized controlled trials. JAMA. 2012;307:1151–60.
- Boone MD, Oren-Grinberg A, Robinson TM, Chen CC, Kasper EM. Mannitol or hypertonic saline in the setting of traumatic brain injury: What have we learned? Surgical Neurology International. 2015;6:177.
- The SAFE Study Investigators. A Comparison of Albumin and Saline for Fluid Resuscitation in the Intensive Care Unit. N Engl J Med 2004; 350:2247-2256.
- Jia X, Kowalski RG, Sciubba DM, Geocadin RG. Critical care of traumatic spinal cord injury. J Intensive Care Med 2013; 28:12.
- Blood pressure management after acute spinal cord injury. Neurosurgery 2002; 50:S58.
- Ditunno JF, Little JW, Tessler A, Burns AS. Spinal shock revisited: a four-phase model. Spinal Cord 2004; 42:383.
- Guidelines for the management of Acute Cervical Spine and Spinal Cord Injuries: http://www.aans.org/en/Education%20and %20Meetings/*/media/Files/Education%20and%20Meetingf/ Clinical%20Guidelines/TraumaGuidelines.ashx (2007). Accessed May 2016.
- Vialet R, Albanèse J, Thomachot L, et al. Isovolume hypertonic solutes (sodium chloride or mannitol) in the treatment of refractory posttraumatic intracranial hypertension: 2 mL/kg 7.5% saline is more effective than 2 mL/kg 20% mannitol. Crit Care Med 2003; 31:1683.
- Kassell N, Baumann K, Hitchon P, et al. The effects of high dose mannitol on cerebral blood flow in dogs with normal intracranial pressure. Stroke 1982;13(1): 59–61.
- Mendelow AD, Teasdale GM, Russell T, et al. Effect of mannitol on cerebral blood flow and cerebral perfusion pressure in human head injury. J Neurosurg 1985; 63(1):43–8.
- Sekhon MS, McBeth P, Zou J, et al. Association between optic nerve sheath diameter and mortality in patients with severe traumatic brain injury. Neurocrit Care. 2014;21(2):245-252.
- Hassen GW, Bruck I, Donahue J, et al. Accuracy of optic nerve sheath diameter measurement by emergency physicians using bedside ultrasound. J Emerg Med. 2015;48(4):450-457.
- Bullock MR, Chestnut R, Ghajar J, et al. Guidelines for the surgical management of traumatic brain injury. Neurosurgery 2006;58(Suppl)S2-1-3.
- Cooper DJ, Rosenfeld JV, Murray L, et al. Decompressive craniectomy in diffuse traumatic brain injury. N Engl J Med 2011;364(16):1493–502.
- Honeybul S, Ho KM, Lind CR. What can be learned from the DECRA study. World Neurosurg 2013;79(1):159–61.
- Sahuquillo J, Martinez-Ricarte F, Poca MA. Decompressive craniectomy in traumatic brain injury after the DECRA trial. Where do we stand? Curr Opin Crit Care 2013;19(2):101–6.
- Hutchinson PJ, Kolias AG, Timofeev IS, Corteen EA, Czosnyka M, Timothy J, et al. Trial of Decompressive Craniectomy for Traumatic Intracranial Hypertension. N Engl J Med. 2016 Sep 7. [Epub ahead of print].
- Sydenham E, Roberts I, Alderson P. Hypothermia for traumatic head injury. Cochrane Database Syst Rev 2009;(2):CD00104.
- Clifton GL, Valadka A, Zygun D, et al. Very early hypothermia induction in patients with severe brain injury (the National Acute Brain Injury Study: Hypothermia II): a randomised trial. Lancet Neurol 2011;10(2):131–9.
- Morgenstern LB, Hemphill JC 3rd, Anderson C, et al. Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2010; 41:2108.
- Eller T, Busse J, Dittrich M, et al. Dabigatran, rivaroxaban, apixaban, argatroban and fondaparinux and their effects on coagulation POC and platelet function tests. Clin Chem Lab Med 2014; 52:835.
- Dickneite G, Hoffman M. Reversing the new oral anticoagulants with prothrombin complex concentrates (PCCs): what is the evidence? Thromb Haemost 2014; 111:189.
- Baharoglu MI, Cordonnier C, Al-Shahi Salman R, de Gans K, Koopman MM, Brand A, Majoie CB. Platelet transfusion versus standard care after acute stroke due to spontaneous cerebral haemorrhage associated with antiplatelet therapy (PATCH): a randomised, open-label, phase 3 trial. 2016 Jun 25;387(10038):2605-13.
- Eisenberg HM, Frankowski RF, Contant CF, et al. High-dose barbiturate control of elevated intracranial pressure in patients with severe head injury. J Neurosurg 1988;69(1):15–23.
- Roberts I, Sydenham E. Barbiturates for acute traumatic brain injury. Cochrane Database Syst Rev 2012;(12):CD000033.
- Roberts I, Yates D, Sandercock P, et al. Effect of intravenous corticosteroids on death within 14 days in 10008 adults with clinically significant head injury (MRC CRASH trial): randomised placebo-controlled trial. Lancet 2004;364(9442): 1321–8.
- Bracken M, Shepard M, Collins W, et al. A randomized, controlled trial of methylprednisolone or naloxone in the treatment of acute spinal-cord injury. Results of the Second National Acute Spinal Cord Injury Study. N Engl J Med 1990; 322(20):1405–11.
- Bracken MB, Shepard MJ, Holford TR, et al. Administration of methylprednisolone for 24 or 48 hours or tirilazad mesylate for 48 hours in the treatment of acute spinal cord injury. JAMA 1997;277(20):1597–604.
- Bracken M. Steroids for acute spinal cord injury. Cochrane Database Syst Rev 2012;(1):CD001046.
- Inaba K, Menaker J, Branco BC, et al. A prospective multicenter comparison of levetiracetam versus phenytoin for early posttraumatic seizure prophylaxis. J Trauma Acute Care Surg 2013;74(3):766-71 [discussion: 771-3].
- Torbic H, Forni A, Anger KE, et al. Use of antiepileptics for seizure prophylaxis after traumatic brain injury. Am J Heal Pharm 2013;70(9):759–66.
- Roberts I, Shakur H, Coats T, Hunt B, Balogun E, Barnetson L, Cook L, Kawahara T, et al. The CRASH-2 trial: a randomised controlled trial and economic evaluation of the effects of tranexamic acid on death, vascular occlusive events and transfusion requirement in bleeding trauma patients. Health Technol Assess. 2013 Mar;17(10):1-79.
- Morrison JJ, Dubose JJ, Rasmussen TE, Midwinter MJ. Military Application of Tranexamic Acid in Trauma Emergency Resuscitation (MATTERs) Study. 2012 Feb;147(2):113-9.
- Perel P, Al-Shahi Salman R, Kawahara T, Morris Z, Prieto-Merino D, Roberts I, Sandercock P, et al. CRASH-2 (Clinical Randomisation of an Antifibrinolytic in Significant Haemorrhage) intracranial bleeding study: the effect of tranexamic acid in traumatic brain injury–a nested randomised, placebo-controlled trial. Health Technol Assess. 2012;16(13):iii-xii, 1-54.
- Dewan Y, Komolafe EO, Mejía-Mantilla JH, Perel P, Roberts I, Shakur H. CRASH-3 – tranexamic acid for the treatment of significant traumatic brain injury: study protocol for an international randomized, double-blind, placebo-controlled trial. Trials. 2012;13:87.
- Wright DW, Yeatts SD, Silbergleit R, et al. Very Early Administration of Progesterone for Acute Traumatic Brain Injury. N Engl J Med 2014;371(26):2457-2466.
- Skolnick BE, Maas AI, Narayan RK, van der Hoop RG, et al; SYNAPSE Trial Investigators. A clinical trial of progesterone for severe traumatic brain injury. N Engl J Med 2014 Dec 25;371(26):2467-76.
- Coles JP, Minhas PS, Fryer TD, et al. Effect of hyperventilation on cerebral blood flow in traumatic head injury: clinical relevance and monitoring correlates. Crit Care Med 2002; 30:1950.
- Coles JP, Fryer TD, Coleman MR, et al. Hyperventilation following head injury: effect on ischemic burden and cerebral oxidative metabolism. Crit Care Med. 2007;35:568–78.






2 thoughts on “Neurotrauma Resuscitation: Pearls & Pitfalls”
Always tought that using a C-collar as spinal precaution gives an dramatic increase of ICP? Is MILS not a better way to go?
Great point! A C-collar can reduce venous drainage, potentially increasing ICP. MILS with foam blocks may be best to allow venous drainage, but more study is needed.