Welcome back to the “52 in 52” series. This collection of posts features recently published must-know articles. Our seventh post looks at the RECOVERY trial.

Author: Brannon Inman (Chief Resident, Emergency Medicine Physician, San Antonio, TX) // Reviewed by: Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit)
Clinical question:
Does dexamethasone in addition to usual care versus usual care alone decrease 28-day mortality in hospitalized patients with COVID-19?
Study design:
- Open label multicenter randomized controlled trial
- Prespecified analyses of the primary outcome were performed in six subgroups: age, sex, race, level of respiratory support, days since symptom onset, and predicted 28-day mortality risk.
- Randomized in a 2:1 ratio (usual care arm vs intervention arm)
- Side note: The RECOVERY (Randomised Evaluation of COVid-19 thERapY) trial describes an overarching trial design that looks at different treatment options simultaneously in patients with SARS-CoV-2 infection, in an adaptive design. The treatments under study include:
- Group A
- No additional treatment
- Steroid in the form of dexamethasone administered orally (liquid or tablets) or intravenously 6 mg once daily for 10 days. In pregnancy or breastfeeding women, prednisolone 40 mg was administered by mouth (or IV hydrocortisone 80 mg twice daily) administered instead of dexamethasone
- Lopinavir 400mg-Ritonavir 100mg by mouth (or nasogastric tube) every 12 hours for 10 days (The arm of this trial has ceased recruitment, as no benefit demonstrated)
- Hydroxychloroquine (this arm of the trial has ceased recruitment, as no benefit demonstrated)
- Azithromycin 500mg by mouth (or nasogastric tube) or intravenously once daily for 10 days
- Group B
- In addition to these treatments patients can be randomised to receive convalescent plasma or not
- Group C
- If there is evidence of a progressive inflammatory state patients can be randomised to receive Tocilizumab or not
- Group A
- This study looked specifically at the dexamethasone arm.
PICO:
Population:
- Inclusion criteria
- Clinically suspected or laboratory confirmed SARS-CoV-2
- Age > 18 years
- Admitted to the hospital
- Pregnant and breastfeeding patients were eligible
- Included 176 hospitals in the United Kingdom
- 6425 patients randomized, 2104 patients received dexamethasone and 4321 patients received usual care
- At randomization, 16% IMV/ECMO, 60% O2 therapy, 24% no O2
- Exclusion criteria
- Nothing explicitly provided in manuscript
Intervention:
- 6mg Dexamethasone (oral or IV) + usual care for 10 days
Comparator:
- Usual care
Outcome:
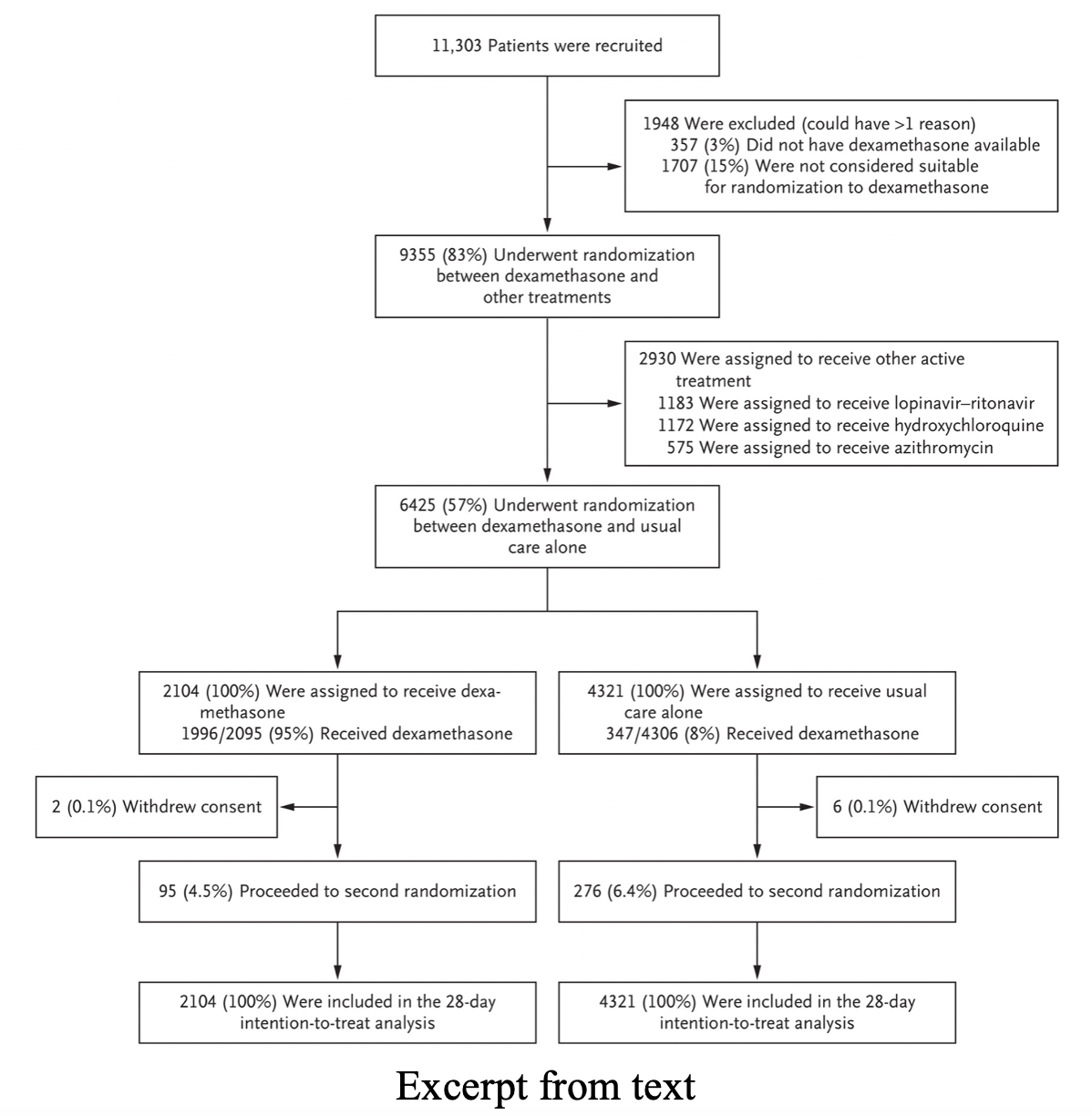
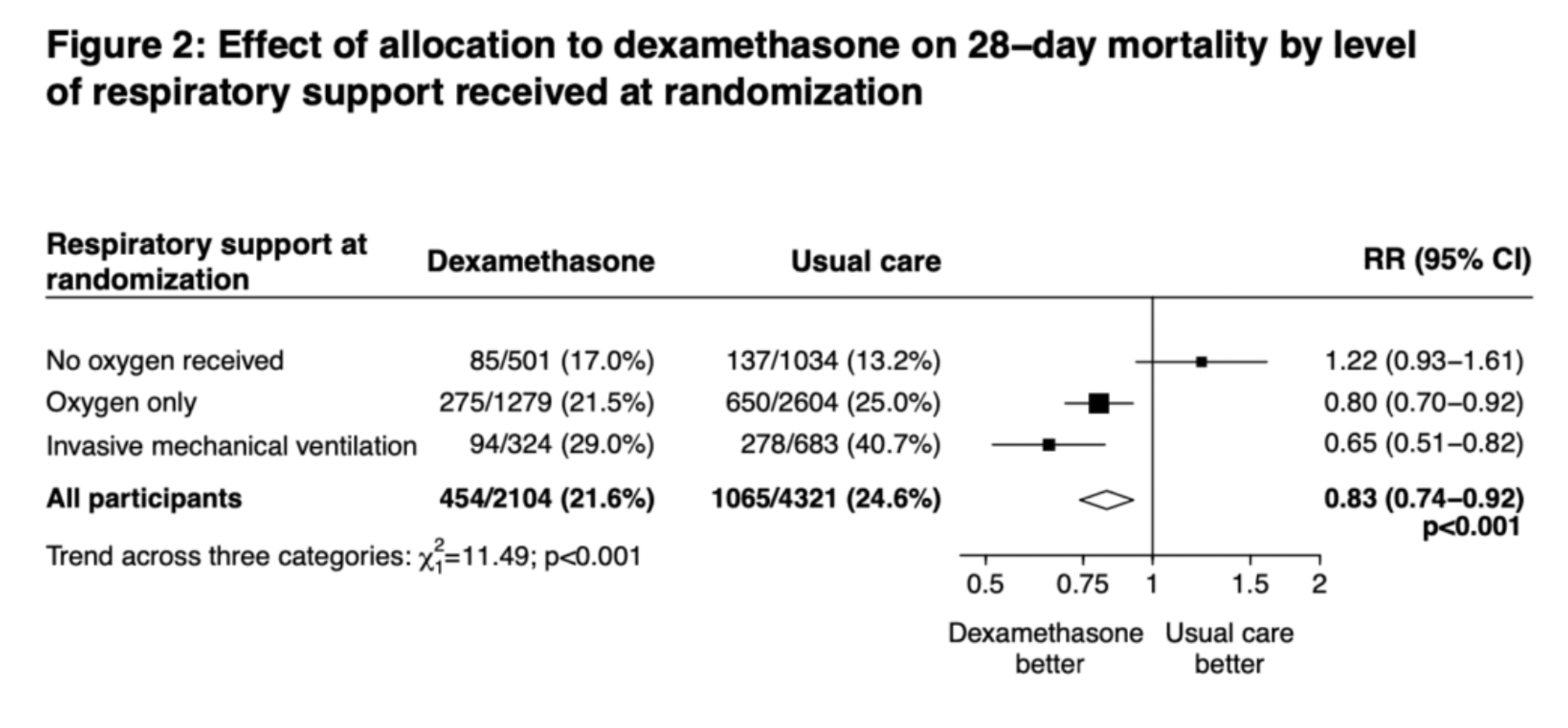
- Overall 28-day mortality
- Dexamethasone + usual care 21.6%
- Usual care 24.6%
- RR 0.65 (95% CI 0.51-0.82)
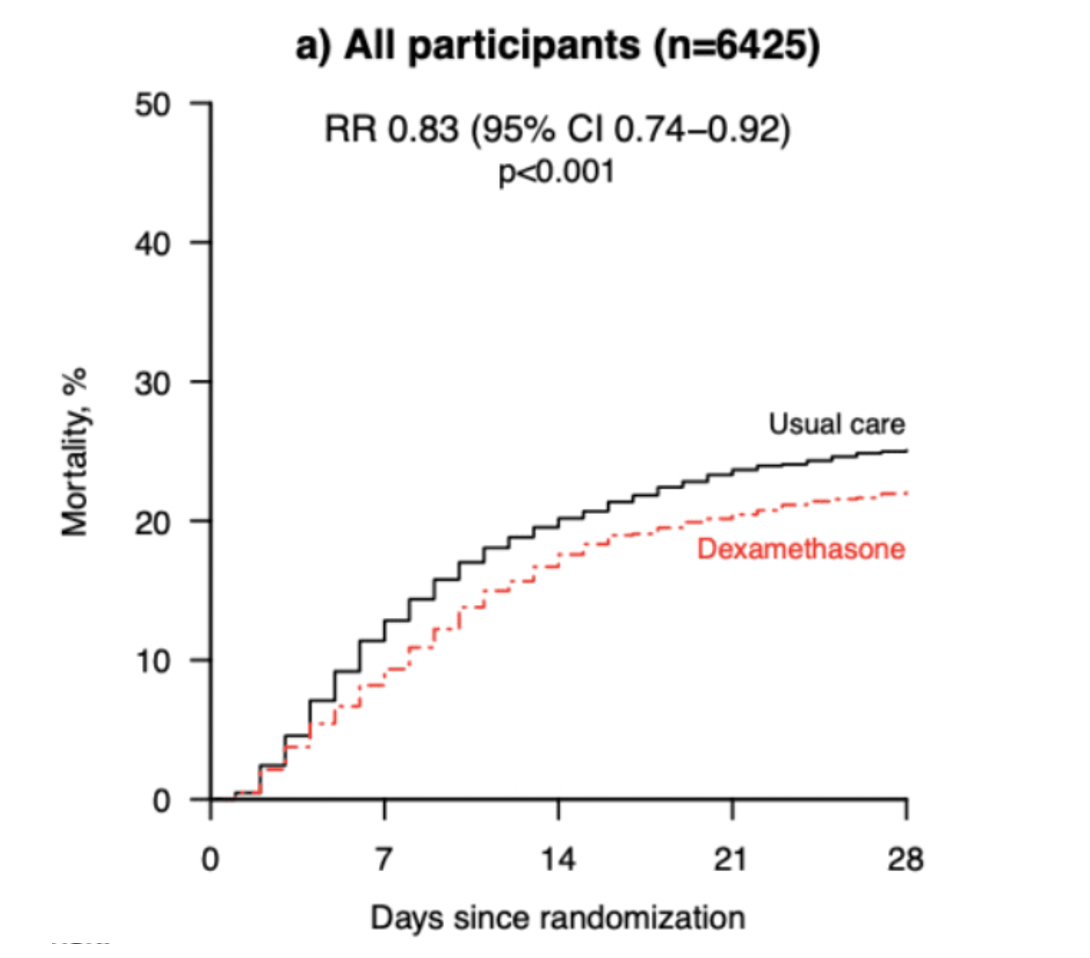
- 28-day mortality in patients on invasive mechanical ventilation [IMV]
- Dexamethasone + usual care 20%
- Usual care 40.7%%
- RR 0.65 (95% CI 0.51-0.82)
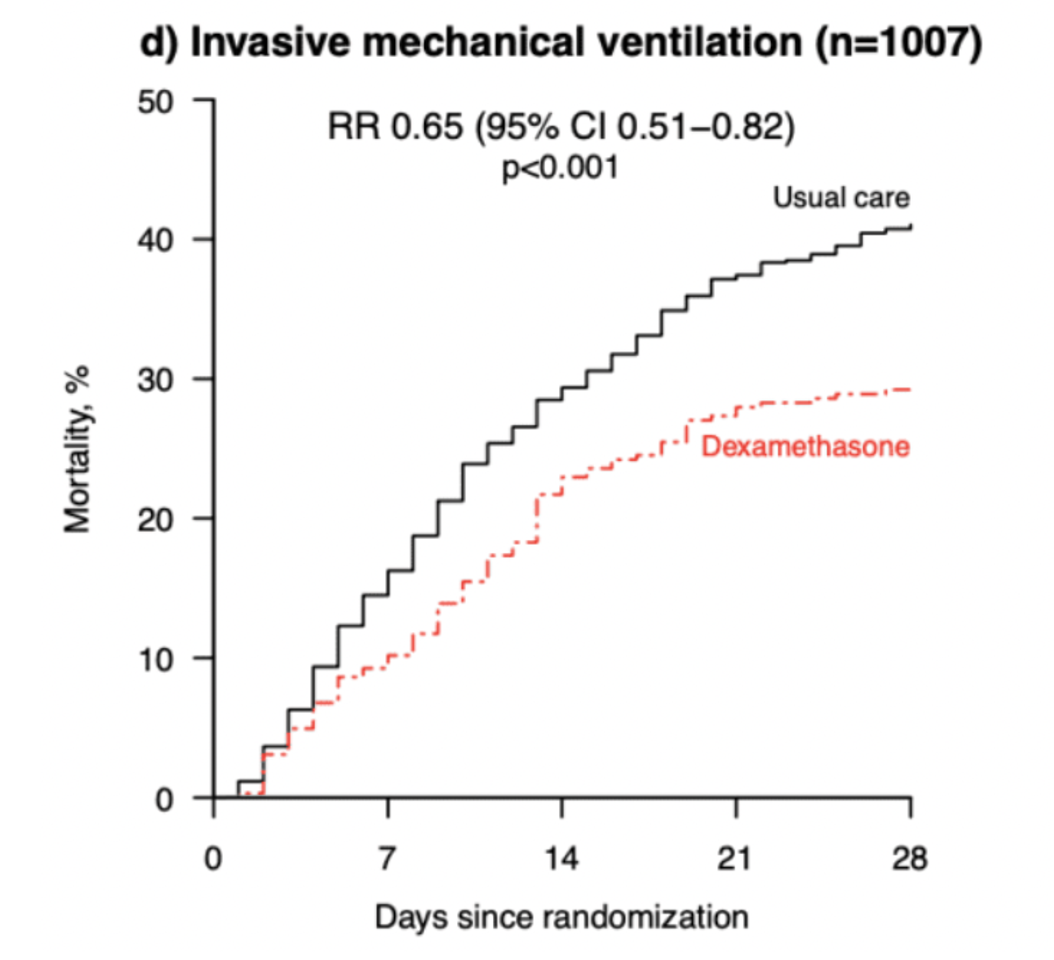
- 28-day mortality in patients on getting supplemental oxygen without IMV
- Dexamethasone + usual care 21.5%
- Usual care 25%%
- RR 0.80 (95% CI 0.70-0.92)

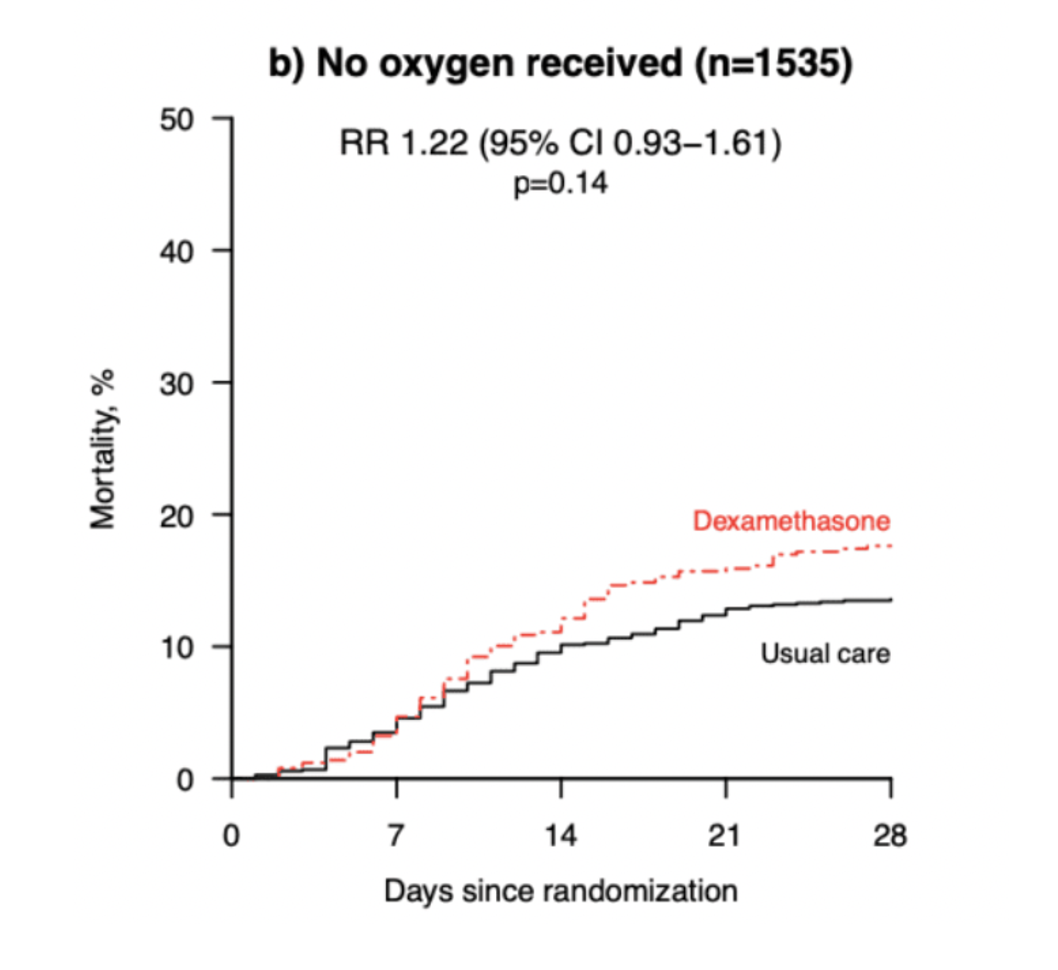
- 28-day mortality in patients without need for oxygen or IMV
- Dexamethasone + usual care 17%
- Usual care 13.2%%
- RR 1.22 (95% CI 0.93-1.61)
Take away:
- Positive trial. This study utilized a patient-centered outcome and used allocation concealment, intention to treat analysis, and had near complete follow-up.
- There was a statistically significant benefit in reduction of 28-day mortality in all admitted patients.
- Preplanned subgroup analysis shows big wins for critically ill intubated patients with a NNT of about 9 for 28-day mortality and a NNT of 29 in patients requiring oxygen supplementation.
- However, subgroup analysis suggests trends toward harm in patients with no supplemental oxygen requirement.
- The study was unblinded and open-label, and usual care was not standardized.
- It is unclear why they chose 6mg of dexamethasone. 6 mg of dexamethasone equates to about 32 mg of methylprednisolone. What about other doses such as 10 mg? This article doesn’t assess larger doses. It would be nice if the dose picked more closely reflected the common practice we use in the ED, which is often 10 mg.
My take:
I will give my SARS-CoV-2 infected patients 6 mg dexamethasone if they have a need to be admitted to the hospital with supplemental O2 or IMV based on this trial. Especially if they need IMV.
Reference:
- RECOVERY Collaborative Group, Horby P, Lim WS, et al. Dexamethasone in Hospitalized Patients with Covid-19. N Engl J Med. 2021;384(8):693-704. doi:10.1056/NEJMoa2021436






