Authors: Emilie Lothet, MD (EM Resident Physician, Washington University School of Medicine in St. Louis); Aaron Lacy, MD (@aaronlacyMD, EM Attending Physician, Washington University School of Medicine in St. Louis) // Reviewed by: Jessica Pelletier, DO (EM Education Fellow, Washington University School of Medicine in St. Louis); Marina Boushra, MD (Cleveland Clinic Foundation, EM-CCM Attending)
Case
A 33-year-old female with no significant past medical history presents to the emergency department (ED) with left-sided face and neck pain. The pain is unilateral, moderate, and has been progressive over the last 24 hours. The patient reports that last night she was the driver involved in a moderate-speed motor vehicle collision in which airbags did not deploy. She was wearing her seatbelt and did not lose consciousness. The patient was able to ambulate after the incident and declined EMS transport for medical evaluation. Her pain developed overnight and was still present upon waking this morning.
Initial vitals are within normal limits. Examination is notable for bruising over her left neck and asymmetrical pupils, with her left pupil measuring 2 mm, and her right 6 mm. On examination, she has lid lag of her left eye. The rest of her neurologic examination is normal, and the patient’s visual acuity is at her baseline. When asked about the difference in pupil size, the patient reports that she first noticed it this morning.
What is the relevance of the patient’s pupillary size difference and how does it pertain to this patient’s workup? How is anisocoria evaluated in the ED and how is the clinician to determine the relevance of this physical examination finding?
Introduction
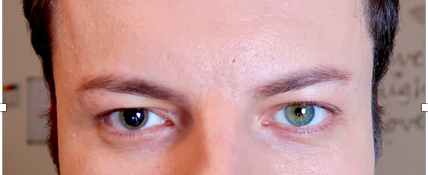
Anisocoria is a condition defined by pupils of unequal sizes (Figure 1).1-4 The possible etiologies for anisocoria are broad and can range from benign conditions to potentially life-threatening pathologies.3,4 It is estimated that physiologic anisocoria, also called simple or essential anisocoria, is present in 10-30% of the population.1-3,5 Physiologic anisocoria is the most common cause of anisocoria, with a pupillary size difference most commonly less than or equal to 1 mm.1-4 Physiologic anisocoria is not associated with a disease process and can be persistent or self-limiting and unilateral or of variable laterality.1-6 However, anisocoria can point to dangerous pathologies, so its presence on physical examination should prompt a thorough evaluation. For that reason, the emergency clinician must understand how to obtain the appropriate history, examination, and workup when anisocoria is found on examination.
Figure 1. Unequal pupils found on examination consistent with anisocoria.7
Pathophysiology
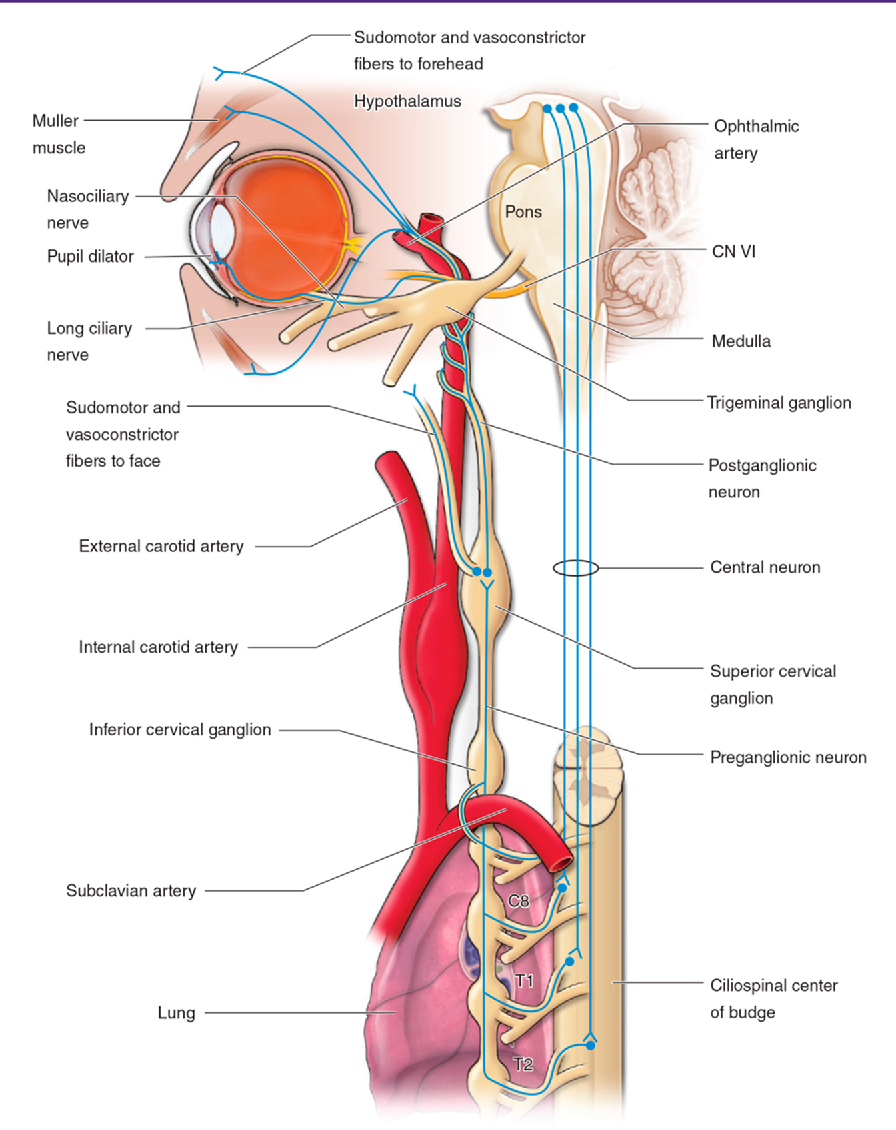
Pupillary constriction is initiated in the brainstem and is controlled by parasympathetic signals. The parasympathetic fibers travel alongside the oculomotor nerve (CN III) before innervating the iris sphincter muscles across an acetylcholine neuromuscular end plate (Figure 2). Upon iris sphincter contraction, the pupil constricts.8,9
Figure 2. Parasympathetic and sympathetic pathways with associated anatomy responsible for pupillary constriction and dilation.11

While pupillary dilation is also initiated in the brainstem, it results from sympathetic input. The sympathetic fibers start in the hypothalamus and extend down the spinal cord before exiting at C8-T2 and synapsing on the paravertebral sympathetic chain.2,10 From there, the sympathetic chain extends cephalad before giving rise to the carotid plexus.2,10 The carotid plexus then follows the path of the external carotid artery through the cavernous sinus before innervating the dilator papillae muscle across a norepinephrine synapse (Figure 3).10 Upon contraction of the dilator papillae, the pupil dilates.2,10 Unilateral interruptions anywhere along these pathways, including the brain, nerve chains, neuromuscular junction, or muscles can lead to anisocoria.4
Figure 3. Autonomic innervation of the eye and associated anatomic structures.9
Categorizing Anisocoria
Anisocoria can be subdivided int0 three broad categories: (1) pathologic large pupil, (2) pathologic small pupil, or (3) physiologic pupil size difference.2,6,9 An initial focused history and examination can help delineate between these three categories. Key history to obtain include: recent head, neck, or eye trauma; past ocular procedures; use of eyedrops or other topical medication; and the presence of other neurologic symptoms.2 Physical examination should include a full ocular assessment, including visual acuity, visual fields, extraocular eye movements, and assessment for an afferent pupillary defect (APD).9
Determining if the anisocoria is acute or chronic is an important first step. This can be facilitated by looking at old photographs, such as those found on social media or a driver’s license, as anisocoria can go unnoticed by the patient or family. If the anisocoria is found to be new, the next step is to identify which of the pupils is ‘pathologic.’6 This can be done by evaluating the sizes of the pupils under different light conditions.9,13 A stepwise evaluation of new anisocoria which can be found in Figure 4.
Figure 4. Stepwise approach to the examination of anisocoric pupils to guide a differential diagnosis and management.6

Differential Diagnosis
After full history, examination, and differentiation between the three categories of anisocoria, a differential diagnosis can be formulated (Table 1).
Table 1. Differential diagnosis of new anisocoria by pathologic pupil identified on clinical examination.2,6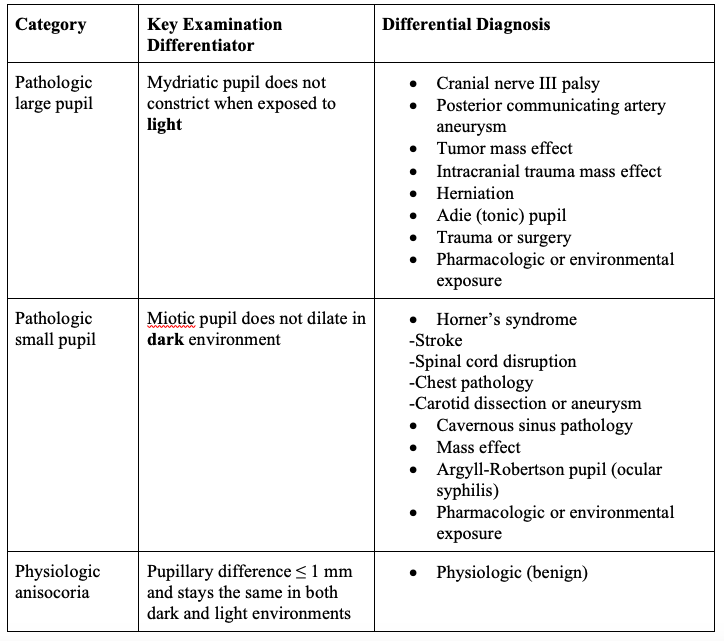
The Pathologic Large Pupil
A pathologically large pupil is caused by impaired parasympathetic signaling leading to unopposed sympathetic output.6 The differential diagnosis for this condition is discussed in detail below:
A. Cranial nerve III (CN III) palsy: Because the parasympathetic innervation of the eye courses along CN III, mydriasis due to external compression of the nerve bundle can present with oculomotor dysfunction. A classic CN III palsy includes extraocular movement dysfunction (“down and out” appearance of the eye), ptosis ( impaired CN III innervation of the levator palpebrae superioris muscle of the eyelid), and mydriasis.3,12 Most CN III palsies do not present with all three classic findings.14-16 The most common cause of CN III palsy (42%) however is microvascular disease-induced mononeuropathy, and will classically not involve the pupil.15,16
Posterior communicating artery aneurysm is an important cause of CN III palsy found in 6% of cases.2,16,17 If this diagnosis is suspected, the next indicated study is computed tomography angiography (CTA) of the head and neck to evaluate for an aneurysm.16,17 While isolated oculomotor dysfunction without pupillary involvement usually points to a mononeuropathy, a pure CN III palsy can be the early presentation of an aneurysm in up to 14% of cases; thus, a low threshold for further investigation is warranted.15 Deficit as result of neurosurgical procedures (10%), neoplastic compression (11%), and traumatic hemorrhage (12%) are other important etiologies of CN III palsy, which can be elucidated with history and computed tomography of the head.3,15,16
B. Adie (tonic) pupil: An Adie pupil arises from the parasympathetic denervation of the constrictor muscles, leading to a large pupil with segmental paralysis when exposed to light.19-21 Classically, the pupil constricts well with near sight but has a sluggish, sometimes irregular re-dilation, therefore exhibiting light-near dissociation.20 The vast majority of Adie pupils (90%) are found in young female patients aged 20-40 years.20,22 Tonic pupil is most often idiopathic and unilateral but can also be seen in the setting of trauma, surgery, or autoimmune disease. In these settings, the cause of initial pupillary denervation leads to non-uniform reinnervation of the sphincter muscles.19,20,22
C. Disruption of the iris: Direct disruption of iris itself is will lead to an appearance of pupillary asymmetry. This can occur in the setting of direct ocular trauma, recent ocular surgery, tumors altering the architecture of the eye, acute angle closure glaucoma, and inflammatory conditions such as iritis and uveitis.20 Acute angle closure glaucoma is a true ocular emergency that is vision-threatening. When this diagnosis is suspected, the emergency provider should check intraocular pressure and obtain ophthalmologic consultation.23,24
D. Pharmacologic and environmental exposures: Direct inoculation with either anticholinergics or sympathomimetic drugs can lead to anisocoria. Anticholinergics inhibit parasympathetic signaling at the neuromuscular junction of the pupillary sphincter and ciliary muscles, leading to mydriasis.20 Scopolamine (most often found in a patch form), belladonna, Jimson weed, and angel’s trumpet are all examples of anticholinergic exposures implicated in anisocoria.25,26
Sympathomimetics cause mydriasis by acting on alpha-1 receptors of the pupillary dilator muscle.20 Implicated exposures include adrenaline and phenylephrine although those are more likely to be ingested systemically and therefore affect both eyes symmetrically.20
The Pathologic Small Pupil
A pathologically small pupil is caused by impaired sympathetic signaling leading to unopposed parasympathetic output.6 The differential diagnosis for the pathologically small pupil is discussed below. The most serious and time-sensitive causes of the pathologic small pupil include stroke, spinal trauma, carotid dissection and aneurysm, and cavernous sinus thrombosis. Carotid artery disruption is responsible for up to 30% of Horner syndromes.
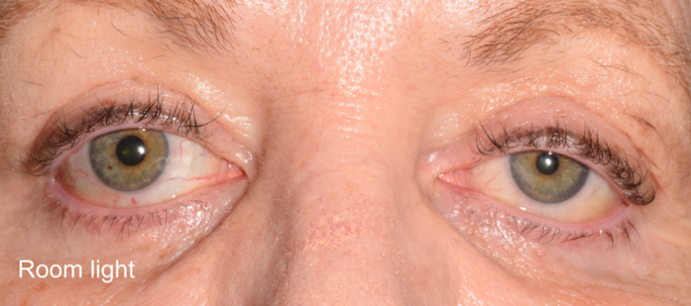
A. Horner syndrome: Horner syndrome is an umbrella term referring to compressive pathology of the oculomotor sympathetic pathway anywhere along its course.9,27 The ‘classic’ triad of Horner syndrome includes ptosis, miosis, and anhidrosis (Figure 4), although the combination of all three findings is rarely present.9,27 Causes of Horner syndrome can be divided into (1) central (first-order); (2) preganglionic (second-order); (3) or postganglionic (third-order) etiologies.9 This nomenclature is based on the relationship of the lesion in relation to the superior orbital ganglion.9 Only two-thirds of patients presenting with Horner syndrome have an identifiable cause, and when found, 13% are central, 44% preganglionic, and 43% postganglionic.28 Notable causes of the pathologic small pupil which can be associated with Horner syndrome are discussed in detail below.
Figure 4. Example of Horner syndrome. The left pupil is miotic compared to the right with associated lid lag of the left eye.9
- Stroke: First-order neurons of the oculomotor sympathetic pathway reside in the hypothalamus and descend to the level of C8-T2, meaning a stroke along this pathway can lead to pathologic miosis.9 However, stroke rarely presents with isolated anisocoria, and there are often other concurrent neurologic findings that can help localize the lesion.9,29 Lesions in the hypothalamus and thalamus cause ipsilateral miosis and ptosis with contralateral hemiparesis and numbness.30 Midbrain lesions will often have a contralateral CN IV palsy, while lesions in the pons may have associated ipsilateral or bilateral CN VI palsy.31 Wallenberg syndrome – a constellation of ipsilateral Horner syndrome, ataxia, contralateral nystagmus, facial weakness, dysphagia, and vertigo – is a result of infarction of the lateral medullary plate.32
- Spinal Cord Disruption: An important first-order cause of pathologic miosis is disruption to the cervical or upper thoracic spinal cord due to trauma, demyelination, tumors, or a syrinx.33-36 Brown-Sequard syndrome is a notable cause in which patients present with pathologic miosis, ptosis, and concurrent ipsilateral motor hemiplegia with contralateral numbness.37 Again, pathology at this level would rarely present with isolated anisocoria and concurrent neurological findings help localize the lesion.
- Chest pathology: Pancoast tumor (superior sulcus tumor) is a preganglionic ‘classic’ cause of Horner syndrome, where a cancerous lesion in the lung apex compresses the oculomotor sympathetic ganglion as it travels out from the C8-T2 spinal nucleus caudally.35-38 However, any mass effect in the apical lung, including infection or pneumothorax, can lead to pathologic miosis.9,35,39 Rarely would this etiology present with isolated pathologic miosis, as apical compression is often associated with pulmonary symptoms, ipsilateral referred shoulder pain, or symptoms of thoracic outlet syndrome.9,40
- Carotid artery dissection and aneurysm: After synapsing at the superior chain ganglion the ocular sympathetic chain follows the course of the carotid artery.9,41 Carotid artery dissection is responsible for 20-30% of diagnosed Horner syndromes, and therefore emergency clinicians must have a low threshold to evaluate for carotid artery pathology in pathologic miotic anisocoria.41-43 Trauma, including chiropractic manipulation, is a strong risk factor for carotid artery dissection and aneurysm.44-45 Carotid artery dissection often manifests with pain in the neck, eye, or head, and may have other associated neurologic deficits.46 If carotid artery pathology is a suspected cause of pathologic isolated miosis, the emergency clinician should pursue computed tomographic angiography (CTA) of the neck and head.43,46
B. Cavernous sinus pathology: As the postganglionic carotid plexus courses toward its final destination, it traverses through the cavernous sinus.9 This makes it susceptible to compression from either thrombosis, aneurysm, or infection in the cavernous sinus.9,20,47 Cranial nerves III, IV, V1, V2, and VI accompany the internal carotid artery through the cavernous sinus; thus, cavernous sinus pathology often presents with palsies of these nerves (most notably CN IV and CN VI) but can also exhibit anisocoria.9,47
C. Other mass effect and end-organ ischemia: The course of sympathetic innervation to the ocular system predisposes it to a wide plethora of possible pathology. Less common causes of pathologic miosis include tumors or iatrogenic injury after procedures of the skull base, nasopharyngeal, and ocular spaces.9,48,49 End-organ ischemia caused by giant cell arteritis, microvascular ischemia, and autonomic neuropathies are also relatively rare causes of pathologic miosis but have been reported in the literature.9,50 Lastly, through mostly unknown mechanisms, cluster headaches and trigeminal neuralgia have been associated with miotic anisocoria.9
Late-stage syphilis can present with an ‘Argyll-Robertson’ pupil.51,52 These patients present with bilateral asymmetric miosis and light-near dissociation, having an appearance similar to anisocoria. The exact pathophysiology of the Argyll-Robertson pupil is unclear, and in the right clinical context, disseminated late-stage syphilis should be considered as a possible etiology for the finding of unequal pupils.51
D. Pharmacologic and environmental exposures: Opioids, pilocarpine, and organophosphates are known to cause miosis. These agents are most often encountered in a systemic fashion, leading to bilateral miosis; however, unilateral pathologic miosis may occur if an exposure physically occurred in a single eye.2,53,54
Pharmacologic Evaluation in the Setting of Diagnostic Uncertainty
If diagnostic uncertainty is present in differentiating the cause of anisocoria, pharmacologic testing can be conducted to assist in diagnosis.2,55 Pharmacologic testing (through eye drops) is nuanced; thus, this post will focus on pharmacologic testing to help in the identification of emergent pathology. Pharmacologic testing for both the pathologic large and small pupils is susceptible to pitfalls and limitations, so the emergency clinician should be cautious to rely solely on pharmacologic testing. Rather, a thorough history and physical examination should be the cornerstone of anisocoria evaluation.
Pharmacologic evaluation of the pathologic large pupil
The most serious cause of a pathologic large pupil to rule out is CN III palsy secondary to external compression on the nerve tract. While most CN III palsies will not present with isolated anisocoria, up to 14% may have anisocoria as the only neurological finding.15 Pilocarpine, a cholinergic muscarinic receptor agonist, can stimulate the iris sphincter muscle causing contraction, and is used in the evaluation of the pathologic large pupil.55
Evaluation should start with a weakly concentrated (0.05-0.15%) pilocarpine eye drop. Dilute pilocarpine is not potent enough to cause pupillary constriction on normal pupils. However, due to cholinergic hypersensitivity, an Adie pupil will constrict when exposed to dilute pilocarpine. 2,20,55 Therefore, constriction with 0.05-0.15% pilocarpine eye drops can help rule in Adie pupil as the etiology for a pathologic large pupil.2,20,55
If there is no constriction with dilute pilocarpine, the next step is to use concentrated pilocarpine of at least 1%.2,55 A large pupil secondary to external nerve compression will have an intact neuromuscular junction and, when exposed to 1% pilocarpine, should constrict.55 If the neuromuscular junction is saturated by other pharmacologic agents, the pupil will not constrict. Therefore, lack of constriction when exposed to concentrated 1% pilocarpine eye drops indicate a pharmacologic etiology to the pathologic large pupil.55 If there is damage to the neuromuscular junction secondary to trauma or infection there also may be no constriction when exposed to concentrated pilocarpine.55 Direct iris damage secondary to trauma or infection is best evaluated via thorough slit lamp examination.20,55
Pharmacologic evaluation of the pathologic small pupil
The most serious causes of a pathologic small pupil to rule out are those leading to Horner syndrome. Historically, 10% cocaine drops have been used in the evaluation of the pathologic small pupil, but due to abuse potential and side effect profile, using cocaine drops in the emergency setting is not practical.55,56 1% apraclonidine, which contains alpha-1 adrenergic properties, will cause pupillary dilation and is an adequate and more accessible substitute.55
The miotic pupil due to Horner syndrome has been observed to be hypersensitive to the presence of norepinephrine in the neuromuscular cleft.55 Therefore, when exposed to 1% apraclonidine, a miotic pupil due to Horner syndrome will exhibit a profound response and dilate while the normal pupil will respond only minimally.2,55,57 However, this test relies on the hypersensitization of the pupil, a process which develops over a minimum of 36 hours and sometimes up to multiple days.2,58-60 This is an important limitation for the emergency clinician to keep in mind.
Case Conclusion
After the patient’s initial presentation, an ocular examination to categorize her anisocoria was undertaken. When examined with the lights turned on, the larger right pupil constricted appropriately, excluding the diagnosis of pathologic mydriasis. When the light source was turned off, the miotic left pupil did not dilate, raising concern for a pathologic small pupil. At this point, the differential diagnosis for anisocoria included pathologies causing Horner syndrome, including vascular compression. Given her concerning bruising of the neck and her associated pathologic anisocoria, CTA of the head and neck was undertaken, revealing a carotid artery dissection. This was thought to be traumatic secondary to her cervical whiplash injury. Vascular surgery was consulted for further management and recommendations, and the patient was admitted to the trauma service for further workup and evaluation.
Pearls:
-Anisocoria can be classified into three groups based on pupillary reaction to light: (1) Physiologic anisocoria, ( (2) a pathologically large pupil, or (3) a pathologically small pupil
–Up to 30% of anisocoria is physiologic and asymptomatic, but this must be a diagnosis of exclusion.
-The most concerning cause of the pathologic large pupil is a cranial nerve III palsy, which can be a marker of compressive neoplasm or aneurysm.
-The most concerning cause of the pathologic small pupil is Horner syndrome, most commonly caused by carotid artery pathology.
-Pharmacologic evaluations can help differentiate between etiologies of anisocoria when the physical exam is inconclusive, with limitations in the emergency setting.
References:
- Lam BL, Thompson HS, Corbett JJ. The prevalence of simple anisocoria. Am J Ophthalmol.1987. 104(1):69–73.
- Gross JR, McClelland CM, Lee MS. An approach to anisocoria. Curr Opin Ophthalmol. 2016. Nov;27(6):486-492
- Payne WN, Blair K, Barrett MJ. Anisocoria. 2022 Aug 15. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan.
- Falardeau J. Anisocoria. Int Ophthalmol Clin. 2019. Summer;59(3):125-139.
- George AS, Abraham AP, Nair S, Joseph M. The Prevalence of Physiological Anisocoria and its Clinical Significance – A Neurosurgical Perspective. Neurol India. 2019. Nov-Dec;67(6):1500-1503.
- Thompson H, Sydney F.J. Pilley, Unequal pupils. A flow chart for sorting out the anisocorias. Survey of Ophthalmology. 1976. 21(1):45-48.
- Anisocoria. WikiMedia Commons. https://commons.wikimedia.org/wiki/Category:Anisocoria#/media/File:Anisocoroa.jpg
- Belliveau AP, Somani AN, Dossani RH. Pupillary Light Reflex. 2022 Jul 25. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan.
- Martin TJ. Horner Syndrome: A Clinical Review. ACS Chem Neurosci. 2018. Feb 21;9(2):177-186.
- Lykstad J, Reddy V, Hanna A. Neuroanatomy, Pupillary Dilation Pathway. 2022 Aug 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan.
- Pupillary Response by Moises Domingez, MD. https://step1.medbullets.com/neurology/113082/pupillary-response
- Wilhelm H. Neuro-ophthalmology of pupillary function–practical guidelines. J Neurol. 1998. Sep;245(9):573-83.
- Steck RP, Kong M, McCray KL, Quan V, Davey PG. Physiologic anisocoria under various lighting conditions. Clin Ophthalmol. 2018. Jan 4;12:85-89.
- Green WR, Hackett ER, Schlezinger NS. Neuro-Ophthalmologic Evaluation of Oculomotor Nerve Paralysis. Arch Ophthalmol. 1964. 72(2):154–167.
- Kissel JT, Burde RM, Klingele TG, Zeiger HE. Pupil-sparing oculomotor palsies with internal carotid-posterior communicating artery aneurysms. Ann Neurol. 1983. 13(2):149–154.
- Fang C, et al. Incidence and etiologies of acquired third nerve palsy using a population-based method. JAMA Ophthalmology. 2017. 135:23
- Lemos J, Eggenberger E. Neuro-ophthalmological emergencies. The Neurohospitalist. 2015. 5(4):223-33.
- Thompson HS. Segmental Palsy of the Iris Sphincter in Adie’s Syndrome. Arch Ophthalmol. 1978. 96(9):1615–1620.
- Xu SY, Song MM, Li L, Li CX. Adie’s Pupil: A Diagnostic Challenge for the Physician. Med Sci Monit. 2022. Mar 8;28:e934657.
- Wilhelm H. Disorders of the pupil. Handb Clin Neurol. 2011. 102:427-66.
- Thompson HS. Adie’s syndrome: some new observations. Trans Am Ophthalmol Soc. 1977. 75:587-626.
- Kanzaria HK, Farzan N, Coralic Z. Adie’s Tonic Pupil. West J Emerg Med. 2012. Dec;13(6):543.
- Weinreb RN, Aung T, Medeiros FA. The pathophysiology and treatment of glaucoma: a review. JAMA. 2014. May 14;311(18):1901-1911.
- Flores-Sánchez BC, Tatham AJ. Acute angle closure glaucoma. Br J Hosp Med (Lond). 2019. Dec 2;80(12):C174-C179.
- Thompson HS. Cornpicker’s pupil: Jimson weed mydriasis. J Iowa Med Soc. 1971. Aug;61(8):475-7.
- Firestone D, Sloane C. Not your everyday anisocoria: Angel’s trumpet ocular toxicity. JEM. 2007. 33(1):21-24.
- Wilhelm H, Ochsner H, Kopycziok E, Trauzettel KS, Schiefer U, Zrenner E. Horner’s syndrome: a retrospective analysis of 90 cases and recommendations for clinical handling. Ger J Ophthalmol. 1: 96–102.
- Maloney WF, Younge BR, and Moyer NJ. Evaluation of the causes and accuracy of pharmacologic localization in Horner’s syndrome. J. Ophthalmol. 1980. 90:394−402.
- de Seze J, Vukusic S, Viallet-Marcel M, et al. Unusual ocular motor findings in multiple sclerosis. J Neurol Sci. 2006. Apr 15;243(1-2):91-5.
- Ortiz de Mendivil A, Alcalá-Galiano A, Ochoa M, Salvador E, Millán JM. Brainstem stroke: anatomy, clinical and radiological findings. Semin Ultrasound CT MR. 2013. Apr;34(2):131-41.
- Kellen RI, Burde RM, Hodges FJ 3rd, Roper-Hall G. Central bilateral sixth nerve palsy associated with a unilateral preganglionic Horner’s syndrome. J Clin Neuroophthalmol. 1988. Sep;8(3):179-84.
- Day GS, Swartz RH, Chenkin J, Shamji AI, Frost DW. Lateral medullary syndrome: a diagnostic approach illustrated through case presentation and literature review. CJEM. 2014. Mar;16(2):164-70.
- Harsfort D, Hagen EM, Hansen RM. Autonomic dysreflexia and concurrent Horner’s Syndrome: a rare presentation in a patient with spinal cord injury. Spinal Cord Ser Cases. 2021. May 28;7(1):47.
- Agarwal PK, Lim LT, Park S, Spiteri-Cornish K, Cox A. Alternating Horner’s syndrome in multiple sclerosis. Semin Ophthalmol. 2012. Jan-Mar;27(1-2):40-1.
- Glassman LR, Hyman K. Pancoast tumor: a modern perspective on an old problem. Curr Opin Pulm Med. 2013. Jul;19(4):340-3.
- Pomeranz H. Isolated Horner syndrome and syrinx of the cervical spinal cord. Am J Ophthalmol. May;133(5):702-4.
- Moskowitz E, Schroeppel T. Brown-Sequard syndrome. Trauma Surg Acute Care Open. 2018 Feb 20;3(1):e000169.
- Gundepalli SG, Tadi P. Lung Pancoast Tumor. 2022 Jul 18. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan.
- White HD, White BA, Boethel C, Arroliga AC. Pancoast’s syndrome secondary to infectious etiologies: a not so uncommon occurrence. Am J Med Sci. 2011. Apr;341(4):333-6.
- Masocatto NO, Da-Matta T, Prozzo TG, Couto WJ, Porfirio G. Thoracic outlet syndrome: a narrative review. Rev Col Bras Cir. 2019. Dec 20;46(5):e20192243.
- Dziewas R, Konrad C, Drager B et al. Cervical artery dissection – clinical features, risk factors, therapy and outcomes in 126 patients. J Neurol. 2003. 250:1179-1184.
- Baumgartner, R. W., and Bogousslavsky, J. Clinical manifestations of carotid dissection. Front Neurol Neurosci. 20:70−76.
- Beebe, J. D., Kardon, R. H., and Thurtell, M. J. The Yield of Diagnostic Imaging in Patients with Isolated Horner Syndrome. 2017. Neurol Clin. 35:145−151.
- Fassett DR, Dailey AT, Vaccaro AR. Vertebral artery injuries associated with cervical spine injuries: a review of the literature. J Spinal Disord Tech. 2008. 21(4):252–8.
- Chen Y, Mofatteh M, Nguyen TN, et al. Carotid Artery Dissection and Ischemic Stroke Following Cervical Chiropractic Manipulation: Two Case Reports. Vasc Endovascular Surg. 2022. Apr;56(3):303-307.
- Hakimi R, Sivakumar S. Imaging of Carotid Dissection. Curr Pain Headache Rep. 2019 Jan 19;23(1):2.
- Munawar K, Nayak G, Fatterpekar GM, Sen C, Zagzag D, Zan E, Hagiwara M. Cavernous sinus lesions. Clin Imaging. Dec;68:71-89.
- Sabbagh MA, De Lott LB, Trobe JD. Causes of Horner Syndrome: A Study of 318 Patients. J Neuroophthalmol. 2020. Sep;40(3):362-369.
- Jimenez-Caballero P, Marsal-Alonso C, Alvarez-Tejerina A. Horner syndrome as the first symptom of nasopharyngeal cancer. Two case reports. Neurol. 2005. 40:541−543.
- Lacy A, Nelson R, Koyfman A, Long B. High risk and low prevalence diseases: Giant cell arteritis. Am J Emerg Med. Aug;58:135-140.
- Dichter SL, Shubert GS. Argyll Robertson Pupil. 2021 Dec 3. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan.
- Thompson HS, Kardon RH. The Argyll Robertson pupil. J Neuroophthalmol. 26:134-138
- Slattery A, Liebelt E, Gaines LA. Common ocular effects reported to a poison control center after a systemic absorption of drugs in therapeutic and toxic doses. Curr Opin Ophthalmol. 25:519-523.
- Dinslage S, Diestelhorst M, Kuhner H, Krieglstein GK. The effect of latanoprost 0.005% on pupillary reaction of the human eye. 2000. 97:396-401.
- Antonio-Santos AA, Santo RN, Eggenberger ER. Pharmacological testing of anisocoria, Expert Opinion on Pharmacotherapy. 2005 Oct 6:12, 2007-2013.
- Smith D. Pharmacological testing in Horner’s syndrome – a new paradigm. SAMJ. 2010. Nov. 100(11):738-740.
- Cambron M, Maertens H, Crevits L. Apraclonidine and my pupil. Clin Auton Res. 2011 Oct;21(5):347-51.
- McDougal DH, Gamlin PD. Autonomic control of the eye. Compr Physiol. 2015 Jan;5(1):439-73.
- Lebas M, Seror J, Debroucker T. Positive apraclonidine test 36 h after acute onset of Horner syndrome in dorsolateral pontomedullary stroke. J Neuroophthalmol. 30:12–17.
- Kauh CY, Bursztyn LL. Positive apraclonidine test in Horner syndrome caused by thalamic hemorrhage. J 2015. 35:287–288.







2 thoughts on “Anisocoria in the ED: Pathophysiology, Evaluation, and Management”
Pingback: Quiz 180, February 17th, 2022 – The FOAMed Quiz
Pingback: Nerdfallmedizin.de - Nerdwoche am 19.02.2023