Authors: John H. Priester, MD (EM Resident Physician, University of Vermont), Mark Bisanzo, MD (EM Attending Physician, University of Vermont) // Reviewed by: Alex Koyfman, MD (@EMHighAK) and Brit Long, MD (@long_brit)
Case
A 57-year-old male presents to the emergency department endorsing a three-day history of severe low back pain. The pain began suddenly as the patient was moving boxes while at work. His back pain has been accompanied by bilateral pain in the posterior legs, a tingling sensation near the midline of his buttocks, and difficulty voiding urine. Vital signs include a HR of 87, BP of 138/90, RR of 14, temperature of 98.4 F, and oxygen saturation of 98%. Physical examination is significant for midline tenderness of the L4-L5 lumbar spine, decreased reflexes bilaterally in the lower extremities, decreased sensation to pinprick in the buttocks and posterior thighs, and a distended lower abdomen. Urethral catheterization following voiding returns 550 ml of urine. What are the next steps in the evaluation and management of this patient?
Introduction
Cauda equina syndrome (CES) is a rare disorder, accounting for 1/370 of patients presenting with back pain1. While definitions of CES vary, it typically includes back pain accompanied by at least one of the following: bowel and/or bladder dysfunction, saddle anesthesia, sexual dysfunction, and lower limb neurologic deficit (e.g. motor/sensory loss, changes in reflexes)2. While back pain is usually present in CES, up to 30% of patients may not experience pain and instead present with lower extremity neurologic deficits3. CES may arise from a wide range of etiologies, including those listed below:

The cauda equina itself is a collection of nerve roots at the L2-L5, S1-S5, and coccygeal levels, responsible for lower limb movement, lower limb sensation, bladder control, anal sphincter control, perineal sensation, and coccygeal sensation11. The syndrome occurs when nerve roots in the lumbar vertebral canal are injured by one of the etiologies described above. The most common cause is compression of the nerve roots at the L4-L5 or L5-S1 level by disc herniation12. Disc herniation has been reported to cause up to 45% of cases of CES13. Prompt recognition of CES and surgical management is critical, as neurologic deficits may be permanent if treatment is delayed14. Diagnosis of CES in the emergency department remains challenging, as symptoms may be variable and with onset ranging from sudden, to occurring in the setting of chronic back pain15. While data on the percentage of CES cases missed in the emergency department is sparse, there is evidence that CES diagnosis and subsequent treatment is often delayed. In a review of CES cases caused by lumbar disc herniation 55% of patients received a delay in definitive treatment4. The majority of delays (83%) were attributed to physician-related causes, including missed diagnosis and delays in imaging. In a similar study, 79% of CES cases were diagnosed > 48 hours after the onset of symptoms, with a median of 11 days16. Such delays in diagnosis and treatment may mean the difference between return of function and permanent disability.
Classification
Classification schemes for CES are based on the clinical onset of symptoms. Predictably, surgical outcomes are improved in cases diagnosed in the earliest stages. In one meta-analysis17, improvements in sensory, motor, urinary and rectal function were more pronounced if treated surgically in the first 48 hours. While no universal classification system is currently in place for CES, several have been proposed12,18-19. Shi et al.18 found that pre-clinical and early manifestations of CES included bulbocavernosus and ischiocavernosus reflex abnormalities, sciatica, and saddle sensory disturbance. Middle and late manifestations included saddle sensory disturbance/loss, reduction or loss of sexual function, bladder and/or bowel dysfunction, and lower extremity weakness. Todd19 proposed a CES classification scale based upon three factors: perineal (saddle) sensation, anal tone, and bladder function. Points are assigned in a manner similar to the Glasgow Coma Scale, with a lower total score representing more severe disease. Lavy et al.12 describes CES in three categories, with type 1 presenting acutely as the first manifestation of lumbar disc herniation, type 2 presenting as the outcome of chronic back pain, and type 3 presenting in a chronic pattern, with slow onset of symptoms. Further classification as complete (with urinary retention) or incomplete (urinary dysfunction without retention) CES has also been proposed12. While no clinical consensus is currently in place for the classification of CES, the schemes described above underscore the importance of history and physical examination in the evaluation of this syndrome.
Risk Factors
Risk factors for CES are generally attributable to conditions that lead to compression of the lumbar or sacral nerve roots20. As discussed above, these include lumbar disc herniation, traumatic injury, malignancy of the spine, and infection. Additionally, disorders that cause inflammatory or degenerative changes to the spine, such as ankylosing spondylitis21 and lumbar spinal stenosis22, respectively, may increase the risk of CES.
Clinical Presentation
The diagnosis of CES in the emergency department is often difficult, as back pain is a common presenting symptom in the ED, and this disorder manifests as a non-specific constellation of signs and symptoms. While definitions of CES vary2, the following signs and symptoms are most commonly present11-12,15,23-25:
- Bladder dysfunction; incomplete emptying with progression to urinary retention and overflow incontinence
- Bowel dysfunction; may include both constipation and incontinence
- Saddle sensory deficit; involves the perineal region, buttocks and/or posterior thighs
- Sexual dysfunction; may include erectile dysfunction, priapism, dyspareunia, and urination during intercourse
- Sciatica; often bilateral
- Lumbar pain
- Diminished lower extremity reflexes
- Lower extremity weakness
Some combination of the first four criteria listed above (i.e. bladder/bowel dysfunction, saddle sensory deficit, and sexual dysfunction) is suggestive of CES2. Additionally, the lack of neurologic deficits in the upper extremities is considered supportive of the diagnosis CES11. In a study of patients with CES identified by MRI, saddle sensory deficit was the only clinical feature that had a statistically significant association with the diagnosis26. This highlights the importance of addressing saddle anesthesia during the patient interview, as deficits may be subtle and manifest as changes in sensation while sitting, wiping or urinating. While the above clinical indicators may be present in CES, it has been proposed that certain factors in the patient’s history and initial evaluation be prioritized. In his review on clinical features of CES, Todd14 identified “red flags” that, if detected, could lead to favorable post-surgical outcomes, and “white flags” that may indicate permanent loss of function:

Evaluation for spinal cord injury often includes assessment of the sacral reflexes (bulbocavernosus reflex and anal reflex)27. While an absence of one or both of these reflexes supports the diagnosis of CES, it is important to note that sensitivity is relatively low. In a study by Domen et al.28 loss of the anal reflex was present in only 37.5% of MRI confirmed cases of CES. In a similar report, abnormal anal reflexes were present in only 38.7% of CES cases23. The clinical features described above highlight the importance of recognizing CES in its early stages with a thorough neurologic examination, perhaps even before the classic symptoms of saddle anesthesia and urinary retention manifest. The diagnosis of CES based on clinical assessment alone, however, is often not adequate. In a study of 80 individuals with signs and symptoms concerning for CES, only 15 (18%) had the disorder as confirmed by MRI26. This is consistent with the 13-22% reported elsewhere28-29. Thus, if CES is suspected clinically, further confirmatory evaluation must be pursued in a timely manner to avoid potential treatment delays.
Diagnosis
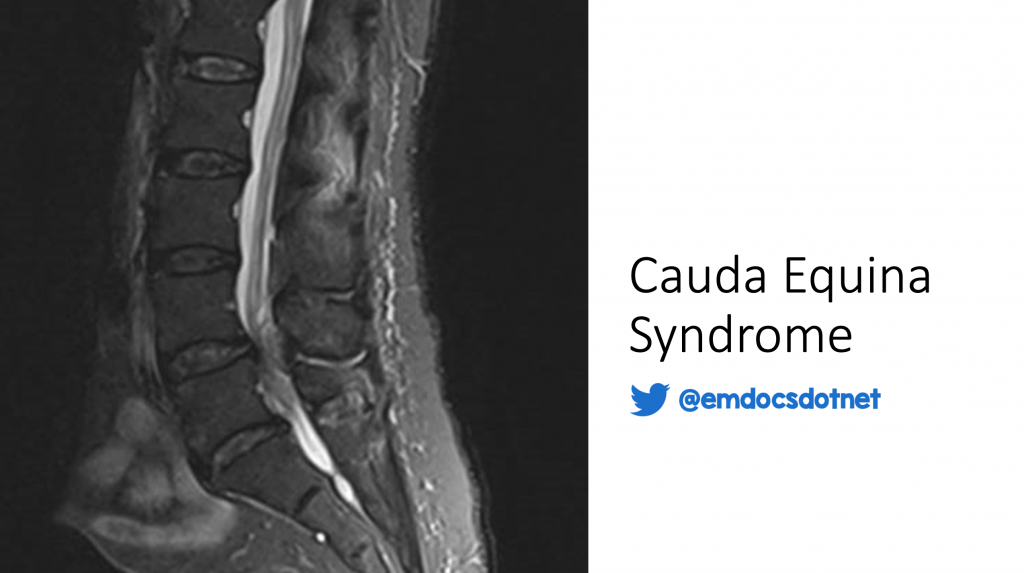
While history and physical examination are crucial in the diagnosis of CES30, additional modalities are required for definitive diagnosis31. Of particular interest to emergency providers is the recent use of point-of-care ultrasound as an aid in the diagnosis of CES. Specifically, post-void residual volume (PVR) measured by ultrasonography is a tool that can be used to risk stratify patients with suspicion for CES. While consensus on exact PVR cutoffs has yet to be established, both low volume (rule out)32 and high volume (rule in)28 strategies have been described. In a study by Venkatesan et al.32, a PVR of <200 mL had a negative predictive value of 97% in the diagnosis of CES. Domen et al.28 found that PVR >500 mL was a reliable predictor of CES. Thus, ultrasound can be an important tool in risk-stratifying patients for further evaluation by MRI. Magnetic resonance imaging of the lumbar spine remains the gold standard for the diagnosis of CES11-12,33. Based on American College of Radiology recommendations, MRI without contrast is typically appropriate, though contrast may be useful depending on the clinical setting34. Situations where contrast is recommended include suspected malignancy or infection35. The most common sequences included in MRI evaluation of CES are sagittal and axial T1, T2 and STIR36. Ancillary studies such as x-ray11, electromyography (EMG)11-12, and CSF analysis are sometimes used11, though their utility as a first-line diagnostic tool is uncertain. These are typically not as useful in the ED setting for diagnosis of CES but may be used to evaluate for other conditions. Although not currently in widespread use, evaluation for thecal sac impingement using spine CT with IV contrast has been explored as a potential future lower-cost modality, with results comparable to MRI for ruling out CES in patients with low pretest probability37. This may be a useful tool in patients with contraindication to MRI (e.g. metallic implants, severe claustrophobia).
Mimics
Diagnosis in the emergency department is often difficult due to the assortment of illness and injury that present with at least one of the symptoms of CES. In addition to lumbar back pain with radiculopathy, reported mimics of CES in the literature include: hypokalemia38, multiple sclerosis39, intracranial metastasis40, intracranial hematoma40, and transverse myelitis41.
In an evaluation of patients who presented clinically with symptoms of CES, but who had negative MR imaging, their presentation was attributable to spinal cord inflammation, infection, neurovascular disease, neoplasm, neurodegenerative disease, and/or medications (including anticholinergics, benzodiazepines, and opiates)42. Symptoms consistent with CES may also be present in conus medullaris syndrome (CMS). CMS, however, often also presents with upper motor neuron (UMN) signs, which are absent in CES43. These UMN signs include increased muscle tone, hyperreflexia, and upgoing plantar response (positive Babinski test)44.
Treatment
Once the diagnosis of CES is made, definitive treatment is surgical decompression at the affected level of the spinal cord45. The timing of surgery is largely dependent on the extent and duration of clinical symptoms. Treatment within the first 48 hours following symptom onset is generally preferred and may be associated with improved outcomes17. For patients with complete deficits, however, it has been suggested that surgery may be delayed, as return of function following spine decompression is considered unlikely46. Prior to surgical management analgesia should be provided for patient comfort. In some patients, particularly those in which malignancy with subsequent compression is suspected47, an IV dose of dexamethasone (typically 8-10 mg) may provide symptom relief in the emergency department48.
Pitfalls and How to Improve
CES is a rare syndrome with a constellation of symptoms that overlap with many common disorders. Given the potential long-term neurologic deficits that may occur, prompt recognition and treatment of CES is essential. A major pitfall with CES in the emergency department is the failure to include this disorder in the differential diagnosis, as well as failure to perform a thorough assessment once suspected. An additional pitfall is the failure to evaluate suspected cases with appropriate imaging. Over reliance on faster, less expensive modalities such as plain films or CT may cause providers to miss cord compression that would otherwise be visible on MRI. While CES is often considered in patients presenting with severe low back pain, emergency providers should also consider it in those arriving to the ED with sciatica, urinary symptoms (e.g. retention, incontinence), bowel symptoms (e.g. constipation, incontinence), saddle sensory deficit, lower extremity weakness, and sexual dysfunction. A thorough patient history and physical exam should be performed, focusing on bowel/bladder function, sexual function, neurologic deficits, and perineal sensation, along with appropriate confirmatory imaging (typically MRI) if suspicion remains high. PVR measurement by ultrasonography can also be helpful and is a rapid means of assessing for urinary retention. Following diagnosis, prompt surgical consultation should be placed for further evaluation and definitive management.
Case Conclusion
MRI of the spine is performed and reveals a large midline disc herniation at the L4-L5 level. IV ketorolac is administered for analgesia and neurosurgery is consulted for further evaluation. The patient is diagnosed with CES and is taken to the operating room for spinal decompression.
Take Home Points
- Cauda equina syndrome (CES) is a rare but important diagnosis, as failure to recognize this disorder may result in delayed treatment and permanent neurologic deficits.
- CES results from compression of lower lumbar and sacral nerve roots, with L4-L5 disc herniation as the most common etiology.
- While CES is typically associated with low back pain, it also manifests with urinary retention, overflow bladder incontinence, constipation, bowel incontinence, saddle anesthesia, sexual dysfunction, sciatica, lower extremity weakness, and diminished lower extremity reflexes.
- Diagnosis of CES is primarily made by MRI.
- Point of care ultrasound to assess for urinary retention can be used to help guide the diagnosis of CES.
- Treatment of CES is by surgical decompression, with the best results occurring within 48 hours of symptom onset.
- CES may be missed by emergency providers if they fail to recognize the signs and symptoms that differentiate it from other sources of back pain. CES should be considered in the differential of all patients reporting back pain, urinary retention, incontinence, bowel dysfunction, sexual dysfunction, and lower extremity weakness.
- CES may also be missed in the emergency department if inappropriate imaging modalities are chosen. In the absence of absolute contraindications, MRI should be pursued in suspected cases instead of faster, less expensive choices such as plain films or CT.
References/Further Reading:
- Hoeritzauer I, Wood M, Copley PC, et al. What is the incidence of cauda equina syndrome? A systematic review. J Neurosurg Spine. 2020;32:832-841.
- Fraser S, Roberts L, Murphy E. Cauda equina syndrome: a literature review of its definition and clinical presentation. Arch Phys Med Rehabil. 2009;90:1964-1968.
- Bednar DA. Cauda equina syndrome from lumbar disc herniation. Can Med Assoc J. 2016;188(4):284.
- Shapiro S. Medical realities of cauda equina syndrome secondary to lumbar disc herniation. Spine. 2000;25(3):348-352.
- Bennett SJ, Katzman GL, Roos RP, et al. Neoplastic cauda equina syndrome: a neuroimaging-based review. Pract Neurol. 2015;16:35-41.
- Harrop JS, Hunt Jr GH, Vaccaro AR. Conus medullaris and cauda equina syndrome as a result of traumatic injuries: management principles. Neurosurg Focus. 2004;16(6):19-23.
- Neal JM, Kopp SL, Pasternak JJ, et al. Anatomy and pathophysiology of spinal cord injury associated with regional anesthesia and pain medicine. Reg Anesth Pain Med. 2015;40(5):506-525.
- Cohen DB. Infectious origins of cauda equina syndrome. Neurosurg Focus. 2004;16(6):5-10.
- He F, Xing T, Yu F, et al. Cauda equina syndrome: an uncommon symptom of aortic diseases. Int J Clin Exp Med. 2015;8(7):10760-10766.
- Tamburrelli FC, Gentitiempo M, Logroscino CA. Cauda equina syndrome and spine manipulation: a case report and review of the literature. Eur Spine J. 2011;20(suppl 1):s128-s131.
- Goodman BP. Disorders of the cauda equina. Continuum (Minneap Minn). 2018;24(2):584-602.
- Lavy C, James T, Wilson-MacDonald J, et al. Cauda equina syndrome. Brit Med J. 2009;338(7699):881-884.
- Kapetanakis S, Chaniotakis C, Kazakos C, et al. Cauda equina syndrome due to lumbar disc herniation: a review of literature. Folia Med. 2017;59(4):377-386.
- Todd NV. Guidelines for cauda equina syndrome. Red flags and white flags. Systematic review and implications for triage. Br J Neurosurg. 2017;31(3):336-339.
- Long B, Koyfman A, Gottlieb M. Evaluation and management of cauda equina syndrome in the emergency department. Am J Emerg Med. 2020;38:143-148.
- Fuso FAF, Dias ALN, Letaif OB, et al. Epidemiological study of cauda equina syndrome. Acta Ortop Bras. 2013;21(3):159-162.
- Ahn UM, Ahn NU, Buchowski JM, et al. Cauda equina syndrome secondary to lumbar disc herniation. Spine. 2000;25(12):1515-1522.
- Shi J, Jia L, Yuan W, et al. Clinical classification of cauda equina syndrome for proper treatment. Acta Orthop. 2010;81(3):391-395.
- Todd NV. Quantifying the clinical aspects of the cauda equina syndrome-the cauda scale (TCS). Br J Neurosurg. 2018;32(3):260-263.
- Kaiser R, Krajcova A, Waldauf P, et al. Are there any risk factors associated with the presence of cauda equina syndrome in symptomatic lumbar disk herniation? World Neurosurg. 2020;141:E600-E605.
- Tang C, Moser FG, Reveille J, et al. Cauda equina syndrome in ankylosing spondylitis: challenges in diagnosis, management, and pathogenesis. J Rheumatol. 2019;46(12):1582-1588.
- Comer C, Finucane L, Mercer C, et al. Shades of grey-the challenge of ‘grumbling’ cauda equina symptoms in older adults with lumbar spinal stenosis. Musculoskelet Sci Pract. 2020;45:102049.
- Korse NS, Pijpers JA, van Zwet E, et al. Cauda equina syndrome: presentation, outcome, and predictors with focus on micturition, defecation, and sexual dysfunction. Eur Spine J. 2017;26:894-904.
- Grasso G, Munakomi S, Salli M. Red flag for cauda equina syndrome in symptomatic lumbar disc herniation. World Neurosurg. 2020;143:232-234.
- Pronin S, Hoeritzauer I, Statham PF, et al. Are we neglecting sexual function assessment in suspected cauda equina syndrome? Surgeon. 2020;18:8-11.
- Balasubramanian K, Kalsi P, Greenough CG, et al. Reliability of clinical assessment in diagnosing cauda equina syndrome. Br J Neurosurg. 2010;24(4):383-386.
- Kirshblum S, Eren F. Anal reflex versus bulbocavernosus reflex in evaluation of patients with spinal cord injury. Spinal Cord. 2020;6:2-4.
- Domen PM, Hofman PA, van Santbrink H, et al. Predictive value of clinical characteristics in patients with suspected cauda equina syndrome. Eur J Neurol. 2009;16:416-419.
- Bell DA, Collie D, Statham PF. Cauda equina syndrome-what is the correlation between clinical assessment and MRI scanning? Br J Neurosurg. 2007;21(2):201-203.
- Greenhalgh S, Finucane L, Mercer C, et al. Assessment and management of cauda equina syndrome. Musculoskelet Sci Pract. 2018;37:69-74.
- Dionne N, Adefolarin A, Kunzelman D, et al. What is the diagnostic accuracy of red flags related to cauda equina syndrome (CES), when compared to magnetic resonance imaging (MRI)? A systematic review. Musculoskelet Sci Pract. 2019;42:125-133.
- Venkatesan M, Nasto L, Tsegaye M, et al. Bladder scans and postvoid residual volume measurement improve diagnostic accuracy of cauda equina syndrome. Spine. 2019;44(18):1303-1308.
- Quaile A. Cauda equina syndrome-the questions. Int Orthop. 2019;43:957-961.
- Patel ND, Broderick DF, Burns J, et al. Low back pain. ACR appropriateness criteria low back pain. J Am Coll Radiol. 2016;13(9):1069-1078.
- Gitelman A, Hishmeh S, Morelli BN, et al. Cauda equina syndrome: a comprehensive review. Am J Orthop. 2008;37(11):556-562.
- Balasubramanya R, Selvarajan SK. Lumbar spine imaging. StatPearls. 2021;available at https://www.statpearls.com/ArticleLibrary/viewarticle/67763″https://www.statpearls.com/ArticleLibrary/viewarticle/67763. Accessed 4/13/2021.
- Peacock JG, Timpone VM. Doing more with less: diagnostic accuracy of CT in suspected cauda equina syndrome. Am J Neuroradiol. 2017;38:391-397.
- Quinlan CS, Walsh JC, Moran AM, et al. Gitelman’s syndrome a rare presentation mimicking cauda equina syndrome. J Bone Joint Surg. 2011;93(B):266-268.
- Vinceti G, Zini A, Nichelli P, et al. Sensory loss mimicking cauda equina syndrome due to cervical spinal lesion in a patient with clinically isolated syndrome. Case Rep Neurol. 2012;4:97-100.
- Kumaria A, Haider Z, Ali A, et al. Intracranial mimics of cauda equina syndrome: heads or tails? World Neurosurg. 2020;144:e643-e647.
- Huh Y, Park E-J, Jung J-W, et al. Clinical insights for early detection of acute transverse myelitis in the emergency department. Clin Exp Emerg Med. 2015;2(1):44-50.
- Hoeritzauer I, Pronin S, Carson A, et al. The clinical features and outcome of scan-negative and scan-positive cases in suspected cauda equina syndrome: a retrospective study of 276 patients. J Neurol. 2018;265:2916-2926.
- McKinley W, Santos K, Meade M, et al. Incidence and outcomes of spinal cord injury clinical syndromes. J Spinal Cord Med. 2007;30:215-224.
- Kunam VK, Velayudhan V, Chaudhry ZA, et al. Incomplete cord syndromes: clinical and imaging review. Radiographics. 2018;38(4):1201-1222.
- Spector LR, Madigan L, Rhyne A, et al. Cauda equina syndrome. J Am Acad Orthop Surg. 2008;16(8):471-479.
- Gleave JRW, MacFarlane R. Cauda equina syndrome: what is the relationship between timing of surgery and outcome? Br J Neurosurg. 2002;16(4):325-328.
- Kumar A, Weber MH, Gokaslan Z, et al. Metastatic spinal cord compression and steroid treatment: A systematic review. Clin Spine Surg. 2017;30(4):156-163.
- Balakrishnamoorthy R, Horgan I, Perez S, et al. Does a single dose of intravenous dexamethasone reduce symptoms in emergency department patients with low back pain and radiculopathy (SEBRA)? A double-blind randomised controlled trial.Emerg Med J. 2015;32:525-530.