Authors: Geoffrey William Fisher, DO & Carolyne Riehle, OMS-IV UNECOM // Edited by: Alex Koyfman, MD (@EMHighAK) and Brit Long, MD (@long_brit)
Case
A 31-year-old female arrives with a chief complaint of “Cough”. She has recently experienced fits of coughing that are non-productive. She admits to feeling “warm” at home but has not taken her temperature. She’s also had some rhinorrhea and congestion.
The patient has also noted some changes in her vision, which has never happened before. She states that 4 or 5 weeks ago she had symptoms of “pins and needles” in her legs that lasted about a week in duration but denies any traumatic event or changes in activity that could have incited this event. She denies any current paresthesias or gait/coordination disturbances.
Initial VS: BP 114/76, HR 77, T 100.7F Oral, RR 16, and SpO2 100% on room air. The physical exam is normal except for her neuro exam. She has a slight delay in eye adduction upon lateral gaze, bilaterally. However, upon convergence both eyes adduct briskly. Aside from this finding, cranial nerves II-XII appear intact. Reflexes are 2/4 throughout, sensory is intact, and the patient demonstrates no gait abnormalities on ambulation.
Lab Results are pertinent for WBC 14,000 cells/mcL, with negative pregnancy test and normal electrolytes and liver function tests.
Based on what you’ve seen so far, what might be going on?
Differential Diagnosis
A young female with neurologic symptoms and signs of viral infection can have a long differential diagnoses. Fortunately, the differential diagnosis for internuclear opthalmoplegia (INO) is exceedingly short!
- Multiple Sclerosis; where the demyelinating lesion has targeted the medial longitudinal fasciculus (MLF)
- Often presents in a younger population
- Presents as bilateral adduction delays with lateral gaze but convergence is preserved (because the MLF is not involved in convergent gaze)
- Cerebrovascular Disease involving lacunar disease and occlusion of the small penetrating arteries stemming from the basilar artery
- Often in the older population with multiple comorbidities
- Usual presents unilaterally
- May or may not have preserved convergence
- Partial Third Nerve Palsy – not true INO
- Usually presents with unilateral weakness of elevation, ptosis, pupil dilation, and impaired convergence
- Infection
- Patient’s often will have photophobia, nystagmus and other symptoms of meningitis/encephalitis
- Subdural Hematoma
- Presents with a history of head trauma
- Drug intoxications: tricyclic antidepressants, phenothiazines, narcotics, lithium, barbiturates, propranolol
- Patients often have a history of anxiety/depression with previous attempts at self-harm.
- Patient’s often present in various states of depressed mental status along with other signs of medication overdose.
Answer: Multiple Sclerosis, flare secondary to URI.
What is the role of an EM doc when it comes to Multiple Sclerosis?
Management and control of life-threatening complications come first. With an initial non-life-threatening presentation, however, not only will making the diagnosis get patients the resources they need sooner through a neurology referral, but early treatment at time of presentation has been shown to significantly impact progression of the disease, in some instances delaying the recurrence and progression by up to 5 years.1
Epidemiology
Secondary to trauma, MS is the most common cause of neurologic disability in young adults.2 The incidence is generally 2-3 times greater in females than in males, with an average age of onset 28-31 years. MS is believed to be an autoimmune condition, and like many autoimmune conditions, it has an increased prevalence in patients suffering from other autoimmune conditions, the most common being psoriasis and thyroid disease (7.7 and 6.4%, respectively).3
Pathophysiology
MS is a demyelinating disorder of the CNS leading to diffuse axonal injury. The pathogenesis of this is somewhat debated, though the predominant theory is T-Cell mediate autoimmune response directed against structural components of the myelin sheath found in the CNS. This patient is presenting with bilateral INO, which is classic in MS patients due to the exceptionally dense myelination, as well as the periventricular location of the MLF.
INO is a disorder of conjugate lateral gaze in which the affected eye, or eyes, show impairment of adduction. When an attempt is made to gaze contralaterally (relative to the affected eye), the affected eye fails to adduct. The contralateral eye will abduct but display nystagmus. Divergence of the eyes will lead to horizontal diplopia or “double vision”. For example, if the right eye is affected the patient will “see double” when looking to the left. Convergence is generally preserved.
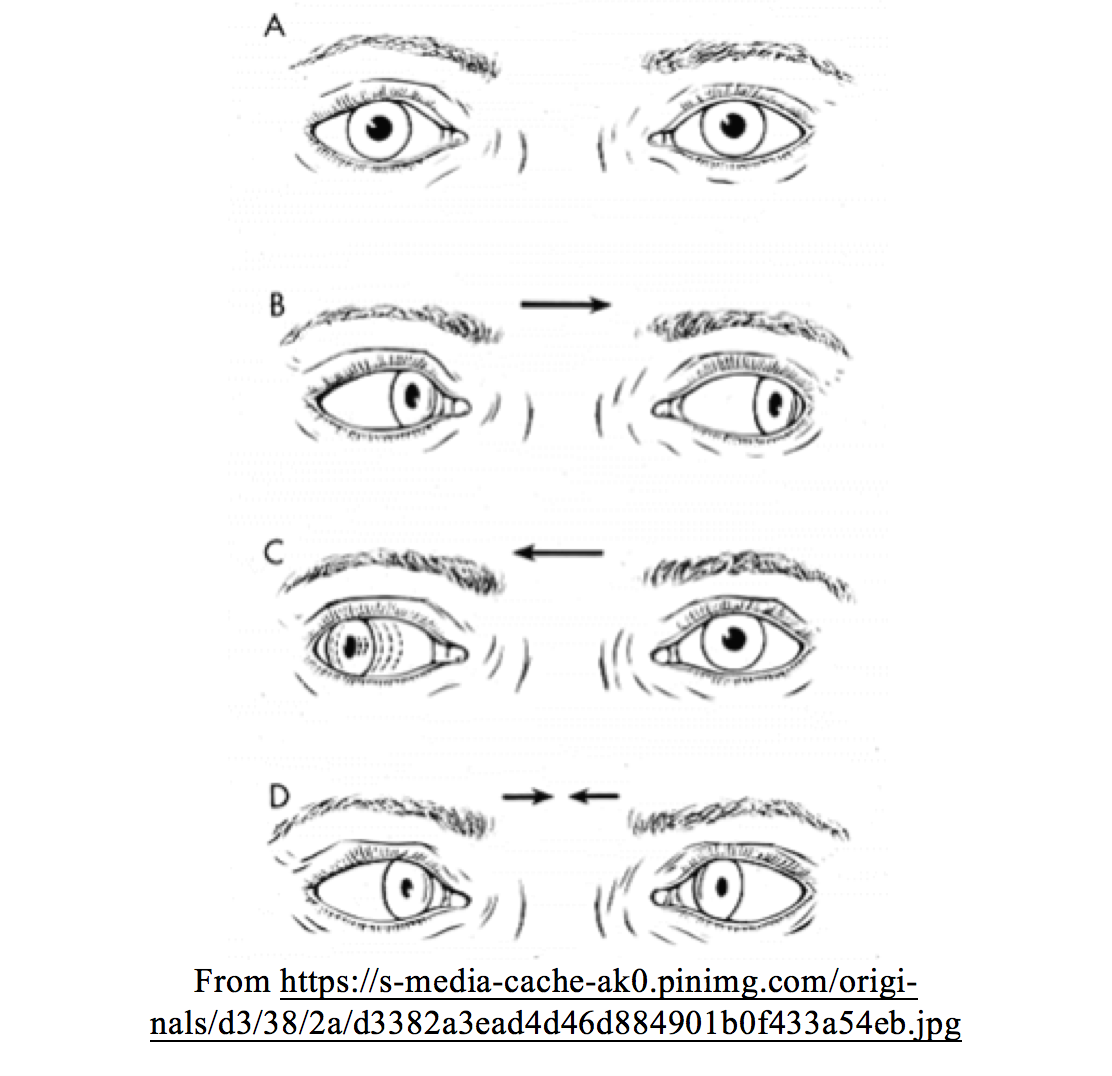
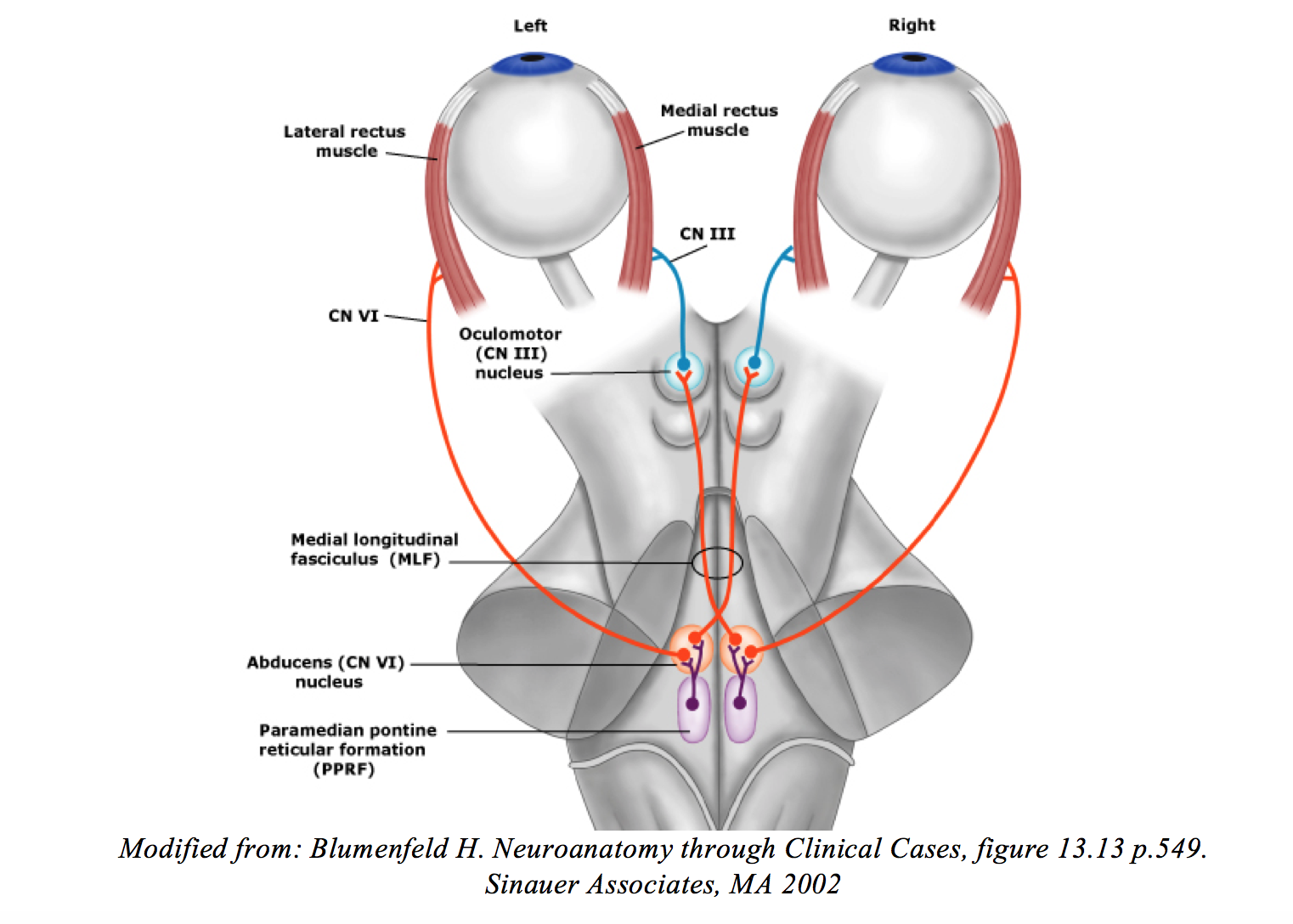
INO is caused by disruption of the MLF, specifically its connection between CN VI nucleus on one side of the pons to the medial rectus subnucleus of CN III in the contralateral midbrain. The MLF connects the vestibular nuclei with the oculomotor, trochlear, and abducens nuclei as well as motor neurons controlling cervical musculature. Specific to horizontal gaze, excitatory interneurons project contralaterally from the abducens (CN VI) nucleus via the MLF to the opposite oculomotor nucleus, to synapse with motor neurons supplying the medial rectus muscle. The combined effect is to adduct the eye (Figure 2).
What about the patient’s URI?
Studies suggest that systemic infectious and inflammatory responses often found in URIs and UTIs are a mediating factor for MS flares.4 Patients with MS are known to have defective receptors on T-regulatory cells, which are responsible in preventing antibody production against native host tissues. With these autoimmune safeguards defective, antibodies directed towards myelin are created either incidentally or through T cells autoreactivity, molecular mimicry, or superantigen-driven immune cell activation.5 Hence, it is important to be suspicious of MS in a patient with new neurological complaints in the presence of infection.
MS Subtypes and Clinical Presentation
Normally, patients initially present with a “Clinically Isolated Syndrome” (CIS) that demonstrates symptoms consistent with demyelinating disease, but which may not meet all the characteristics of a classic MS presentation. The most clinically relevant symptoms often seen in these patients are listed in Table 1.
Approximately 15% of patients presenting with a CIS will never demonstrate a second relapse or further progression, called “Benign MS”. In the rest of the patient population, 85% will ultimately suffer from the Relapsing-and-Remitting (RRMS) type, characterized by clearly defined episodes of neurologic insult, often without residual deficit upon recovery. The majority of RRMS patients will suffer further periodic neurologic flares the rest of their lives. A small subset of patients with RRMS will progress with what’s known as Secondary Progressive MS (SPMS), where there are no relief periods of symptoms remission, and instead the patient experiences a gradual progression of symptoms. SPMS typically occurs 19 years after the patient’s initial CIS. Lastly, 10% of patients will experience a form of MS known as Primary Progressive (PPMS), characterized as progressive disability, with periodic plateaus in symptoms (and rarely very mild and brief states of remission).
PEARL: Patients with PPMS classically exhibit spastic paralysis during their CIS and are often initially diagnosed with transverse myelitis.

i. One study found that nearly 18% of patients presenting with acute MS flares displayed urine dipstick analysis consistent with UTI.5
ii. Frank dementia occurs in less than 5% of patients; however, neuropsychological tests usual demonstrate some impairment in complex attention, efficiency of information processing, executive functioning, processing speed, or long-term memory.
Handy “Rule-Out” Clinical Presentations16
Not only are there classic clinical presentations indicative of MS, there are several key patient presentations that allow you to place MS much further down on your differential, specifically: Hearing loss (especially bilateral), presentation of paresthesias in patients less than 10 years of age or greater than 50, serum ESR > 80, CSF protein > 100, and CSF WBC > 50.
Diagnosis: TIME and SPACE
- The diagnosis of MS is largely clinical and utilizes the McDonald Criteria, explained below.
- Lumbar punctures are no longer required for the diagnosis of MS.
- LPs look for oligoclonal bands and elevated IgG as indicators of MS, and while no longer necessary, may still has some utility in certain situations.
McDonald Criteria: These were originally established to aid the clinical diagnosis of MS based on symptom recurrence (dissemination through TIME) and the presence of new symptoms different from those of the CIS (dissemination through SPACE of the CNS). This required observation over the course of years to prove dissemination of the disease through “time” and “space” and delayed the diagnosis. In 2010 a revised version was made, and current guidelines have a respective sensitivity and specificity of 77% and 92%, respectively.6

What is the real value of analyzing a patient’s CSF?
The presence of IgG (and less frequently, IgM) and Oligoclonal Bands (OCBs) upon electrophoresis serve as markers of immune activity within the CNS. Immunoglobulins do not readily cross the Blood Brain Barrier (BBB), so an elevated ratio of CSF to serum Ig is a hallmark of MS pathology. In patients diagnosed with MS, the presence of OCBs is unique to CSF when compared to serum. In these same patients, 70-90% have a significantly elevated CSF:Serum IgG index.7
In CIS, OCBs are found in roughly 60% of patients. However, studies demonstrated that the amount of OCBs in CIS strongly correlates with the number of lesions present on a baseline MRI.8
While this evidence shows that there is some diagnostic utility in an IgG Index, remember that there are many other causes of CNS inflammation, and BBB disruption that can lead to an elevated IgG index, including viral meningitis, PML, lymphoma, neurosyphilis, acute inflammatory polyradiculoneuropathy, and subacute sclerosing panencephalitis. Basically, you should look for OCBs and an elevated IgG Index when an MRI is unavailable only when you can safely rule out other causes of potentially positive results.
Need and application of the MRI
An MRI with and without contrast should be obtained in all patients suspected of having CIS or MS. Patients will need a baseline for follow-up with neurology. That being said, it’s unreasonable to think that all EDs have access to an MRI right away. In these cases, you can perform an LP looking for evidence of OCBs and an elevated Ig Index. If the patient does not have a known diagnosis of MS with focal deficits, admission is warranted.
If you do have access to an MRI here’s what you need to consider: In a T1 image, grey matter will give off a low signal brightness, while white matter will give off brightest signal (and CSF, which is mostly water by comparison, gives off almost no signal). T2 is pretty much an inverse image of this (think of it as the negative) and FLAIR (fluid attenuated inversion recovery) cleverly selects out and discards signal produced by the CSF. With a T2-FLAIR image normal healthy myelinated white matter will look dark grey, while demyelinated regions representative of old flares will look bright white! In contrast, active plaques are best visualized with T1 gadolinium enhancement, because during an active flare the BBB is compromised allowing permeation of the lesion by contrast circulating in the blood. This is a good way to utilize an MRI to meet your Dissemination-in-Time criteria.
MRI diagnosis focuses on dissemination in space, and diagnosis of MS includes lesions in at least 2 of the 5 following locations: Periventricular: ≥3 lesions, Cortical-Juxtacortical: ≥1 lesion, Infratentorial: ≥1 lesion, Spinal cord: ≥1 lesion, Optic nerve: ≥1 lesion.19,20-24, 27-32

Treatment
- Acute MS Flare17-18
- In patients with the diagnosis of MS, IV methylprednisolone or oral prednisone are the treatments of choice for flares.
- Preferred Method: A 5-day course intravenous methylprednisolone 1000 mg daily without an oral taper, commonly completed as an inpatient.
- Alternative Method: A 3-7 day course of oral prednisone, 625 to 1250 mg daily, with or without a short taper can also be used in patients desiring outpatient therapy who don’t want to return to the hospital for daily infusions.
- Pearls:
- If the patient has signs of systemic infection, treatment with glucocorticoids should be delayed until infection has been treated and is resolved.
- Patients with osteoporosis/penia, other bone demineralization disorders, or history of fractures will need to be counseled on their increase risk of fracture while on glucocorticoids.
- In patients with the diagnosis of MS, IV methylprednisolone or oral prednisone are the treatments of choice for flares.
- CIS: Treat like an acute MS Flare +/- β Interferon therapy
- In randomized controlled trials treatment with beta interferons, glatiramer acetate, and IV immune globulin has been shown to delay the conversion of CIS to MS by up to 5 years.1,9-15 However, it is still unknown whether such treatments have any effect on long-term disability.
- The role of interferon therapy in the ED should be done at the behest of a neurology consult. If the patient’s symptoms warrant discharge, neurology should be contacted to confirm whether an initial dose of interferon therapy should be given prior to discharge.
Disposition
Reasons for admission include several considerations. Patients with focal neurologic deficits or for those in who the diagnosis of MS is not confirmed should be admitted. Other reasons for admission include disabling/life-threatening symptoms, patients with pre-existing diabetes (due to the high-dose glucocorticoid treatment, these patients may experience severe hyperglycemia, and will require strict inpatient glucose monitoring with insulin control), patients with severe inciting event such as sepsis, and those unable to obtain follow-up or with poor social situations. Patients may be appropriate for discharge if the diagnosis of MS is confirmed and neurology and primary care follow is ensured within the next several days.
Take Home Points
- History and presentation are perhaps the most important and may provide you with enough evidence of disease progression “through time and space” to reach a diagnosis.
- MRI will be needed at some point to look for asymptomatic lesions and establish a baseline for future monitoring.
- LPs can be used if an MRI is unavailable or unequivocal, but sensitivity is often low in CIS, and specificity is only high when you can rule out other causes of an elevated Ig Index.
- Treatment consists of high dose steroids +/- β Interferon if you have reason to believe this is a CIS.
References/Further Reading:
- Kinkel RP, Kollman C, O’Connor P, et al., CHAMPIONS Study Group. IM interferon beta-1a delays definite multiple sclerosis 5 years after a first demyelinating event. Neurology. 2006;66(5):678. Epub 2006 Jan 25
- Ramagopalan SV, Sadovnick AD. Epidemiology of multiple sclerosis. Neurologic Clinics, May 29, 2011. Volume 29, Issue 2, Pages 207-217
- Marrie RA, Reider N, Cohen J, et al. A systematic review of the incidence and prevalence of autoimmune disease in multiple sclerosis. Multiple Sclerosis. 2015 Mar; 21(3): 282–293
- Correale J, Fiol M, Gilmore W. The risk of relapses in multiple sclerosis during systemic infections. Neurology. 2006 Aug 22;67(4):652-9
- Mahadeva A, Tanasescu R, and Gran B. Urinary tract infections in multiple sclerosis: under-diagnosed and under-treated? A clinical audit at a large University Hospital. American Journal of Clinical and Experimental Immunology. 2014; 3(1): 57–67
- Swanton JK, Fernando K, Dalton CM, et al. Modification of MRI criteria for multiple sclerosis in patients with clinically isolated syndromes. Journal of Neurology, Neurosurgery & Psychiatry. 2006;77 (7): 830-3.
- Luzzio C, Dangond F. Multiple Sclerosis – Lumbar Puncture. emedicine.medscape.com Jun 02, 2017
- Kuhle J, et al. Conversion from clinically isolated syndrome to multiple sclerosis: A large multicentre study. Multiple Sclerosis Journal. 2015, Vol. 21(8) 1013–1024
- Jacobs LD, Beck RW, Simon JH, et al. Intramuscular interferon beta-1a therapy initiated during a first demyelinating event in multiple sclerosis. CHAMPS Study Group. New England Journal of Medicine. 2000 Sep 28;343(13):898-904
- Comi G, Filippi M, Barkhof F, et al, Early Treatment of Multiple Sclerosis Study Group. Effect of early interferon treatment on conversion to definite multiple sclerosis: a randomised study. Lancet. 2001;357(9268):1576.
- Filippi M, Rovaris M, Inglese M, et al. Interferon beta-1a for brain tissue loss in patients at presentation with syndromes suggestive of multiple sclerosis: a randomised, double-blind, placebo-controlled trial. 2004;364(9444):1489.
- Kappos L, Polman CH, Freedman MS, et al. Treatment with interferon beta-1b delays conversion to clinically definite and McDonald MS in patients with clinically isolated syndromes. 2006;67(7):1242. Epub 2006 Aug 16.
- Clerico M, Faggiano F, Palace J, et al. Recombinant interferon beta or glatiramer acetate for delaying conversion of the first demyelinating event to multiple sclerosis. Cochrane Database Syst Rev. 2008
- Kappos L, Freedman MS, Polman CH, et al, BENEFIT Study Group. Effect of early versus delayed interferon beta-1b treatment on disability after a first clinical event suggestive of multiple sclerosis: a 3-year follow-up analysis of the BENEFIT study. 2007;370(9585):389.
- Kappos L, Freedman MS, Polman CH, et al. BENEFIT Study Group. Long-term effect of early treatment with interferon beta-1b after a first clinical event suggestive of multiple sclerosis: 5-year active treatment extension of the phase 3 BENEFIT trial. Lancet Neurol. 2009;8(11):987.
- Westover B, DeCroos E, Awad K, Bianchi M. Pocket Neurology: Second Edition. Wolters Kluwer Publishing.
- Kupersmith MJ, Kaufman D, Paty DW, Ebers G, McFarland H, Johnson K, Reingold S, Whitaker J. Megadose corticosteroids in multiple sclerosis. 1994 Jan;44(1):1-4.
- Murray TJ. Diagnosis and treatment of multiple sclerosis. 2006;332(7540):525.
- Kang H, Metz LM, Traboulsee AL, Eliasziw M, Zhao GJ, Cheng Y, Zhao Y, Li1 DKB and the Minocycline in CIS Study Group. Application and a proposed modification of the 2010 McDonald criteria for the diagnosis of multiple sclerosis in a Canadian cohort of patients with clinically isolated syndromes. Multiple Sclerosis Journal 2014, Vol. 20(4) 458–463
- Filippi M, Rocca MA, Ciccarelli O, et al. MRI criteria for the diagnosis of multiple sclerosis: MAGNIMS consensus guidelines. The Lancet Neurology, Volume 15, Issue 3, 2016, Pages 292-303, ISSN 1474-4422
- Absinta M, Rocca MA, Colombo B, et al. Patients with migraine do not have MRI visible cortical lesions. Journal of Neurology, 259 (2012) pp. 2695-2698
- Miller D, Barkhof F, Montalban X, Thompson A, Filippi M. Clinically isolated syndromes suggestive of multiple sclerosis, part I: natural history, pathogenesis, diagnosis, and prognosis. The Lancet Neurology, Volume 4, Issue 5, 2005, Pages 281-288, ISSN 1474-4422
- Tintore M, Rovira A, Rio J, et al. Defining high, medium and low impact prognostic factors for developing multiple sclerosis. Brain, 138 (2015), pp. 1863-1874
- Calabrese M, Oh MS, Favaretto A, et al. No MRI evidence of cortical lesions in neuromyelitis optica. Neurology, 79 (2012), pp. 1671-1676
- Favaretto A, Poggiali D, Lazzarotto A, Rolma G, Causin F, Gallo P. The parallel analysis of phase sensitive inversion recovery (PSIR) and double inversion recovery (DIR) images significantly improves the detection of cortical lesions in multiple sclerosis (MS) since clinical onset. PLoS One, 10 (2015), p. e0127805
- Calabrese M, De Stefano N, Atzori M, et al. Detection of cortical inflammatory lesions by double inversion recovery magnetic resonance imaging in patients with multiple sclerosis. Arch Neurol, 64 (2007), pp. 1416-1422
- Sombekke MH, Wattjes MP, Balk LJ, et al. Spinal cord lesions in patients with clinically isolated syndrome: a powerful tool in diagnosis and prognosis. Neurology, 80 (2013), pp. 69-75
- Bot JC, Barkhof F, Polman CH, et al. Spinal cord abnormalities in recently diagnosed MS patients: added value of spinal MRI examination. Neurology, 62 (2004), pp. 226-233
- Weier K, Mazraeh J, Naegelin Y, et al. Biplanar MRI for the assessment of the spinal cord in multiple sclerosis. Mult Scler, 18 (2012), pp. 1560-1569
- Nair G, Absinta M, Reich DS. Optimized T1-MPRAGE sequence for better visualization of spinal cord multiple sclerosis lesions at 3T. AJNR Am J Neuroradiol, 34 (2013), pp. 2215-2222
- Giorgio A., et al. Location of brain lesions predicts conversion of clinically isolated syndromes to multiple sclerosis. Neurology, 80 (3) (2013), pp. 234-241
- Droby A, Fleischer V, Carnini M, Zimmermann H, Siffrin V, Gawehn J, Erb M, Hildebrandt A, Baier B, Zipp F. The impact of isolated lesions on white-matter fiber tracts in multiple sclerosis patients. NeuroImage: Clinical, Volume 8, 2015, Pages 110-116, ISSN 2213-1582







2 thoughts on “Multiple Sclerosis in the ED: Pearls & Pitfalls”
Pingback: Multiple Sclerosis in the ED: Pearls & Pitfalls – Global Intensive Care
Pingback: Weekly Update – 29 March 2021 – NewYork-Presbyterian Emergency Medicine