Authors: Mitchell Blenden, MD (EM Resident Physician, Northwestern) and Kelly Williamson, MD (EM Attending Physician, Northwestern) // Reviewed by: Alexander Y. Sheng MD MHPE (@TheShenger); Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit)
Case
A 64-year-old male with a past medical history of hypertension presents to the emergency department with right lower back pain. He states the pain started when he bent down to reach for dishwasher detergent yesterday evening. He feels the pain starting in his lower back and radiating down the leg. He denies any recent trauma or similar symptoms in the past. He denies bowel or bladder incontinence, weakness, sensation changes, fevers, or recent weight loss. You suspect his presentation is consistent with sciatica. How is sciatica diagnosed and what are some mimics that comprise the differential diagnosis?
What is Sciatica?
Sciatica, also known as lumbosacral radiculopathy, is characterized by radiating leg pain that occurs when the lumbosacral nerve roots are compressed or inflamed. Sciatica is a clinical diagnosis based on symptoms. Patients commonly describe aching and sharp pain radiating in one leg without the presence of neurologic deficits on exam. The most common cause is disc herniation due to age related degenerative disc disease. Risk factors for sciatica include older age, obesity, an occupation that requires carrying heavy loads or a lot of twisting, a history of smoking and a sedentary lifestyle 1. Symptoms that may suggest sciatica include unilateral pain that is more severe than low back pain, pain that radiates posteriorly down the leg, and paresthesias involving the lower leg. Symptoms that may suggest another etiology and prompt further investigation for more serious causes of pain include severe or progressive neurologic deficits, a suspicion of malignancy, bowel or bladder dysfunction, a history of trauma, and symptoms that have persisted for longer than 12 weeks 2. Below is a list of some pathology that can mimic sciatica type pain and requires further investigation.
Sciatica Mimics
Spinal Cord Compression / Cauda Equina Syndrome
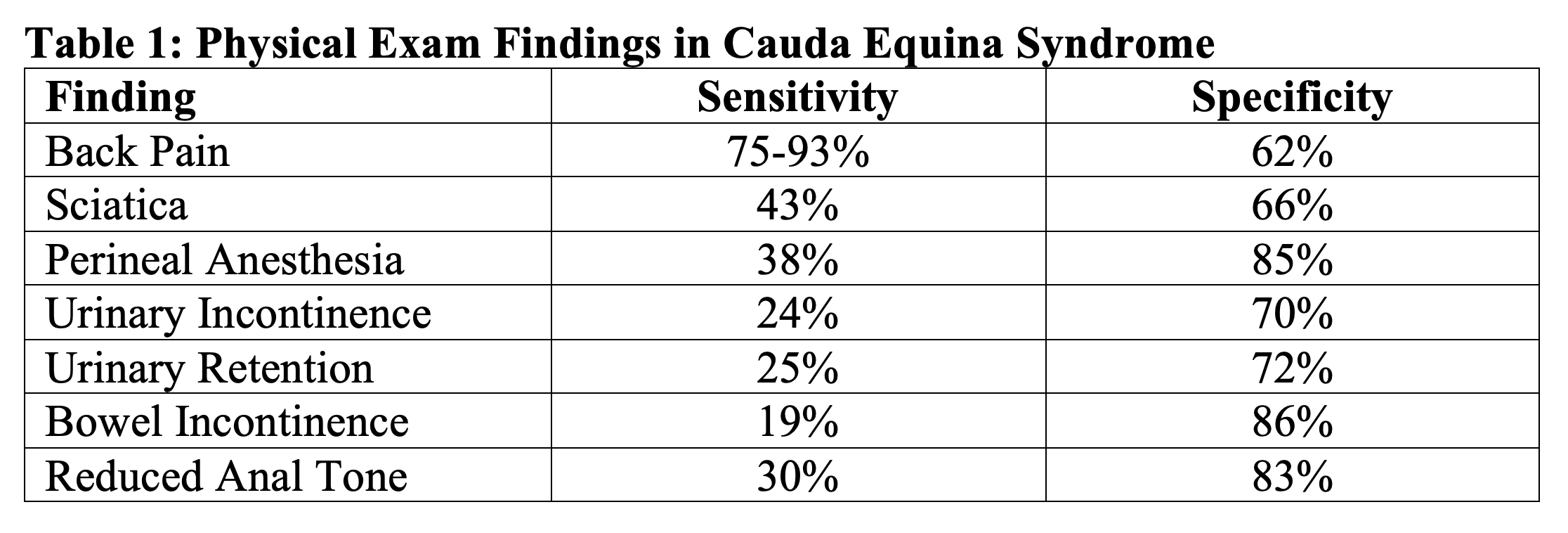
The collection of nerves at the end of the spinal cord is known as the cauda equina. Cauda equina syndrome (CES) occurs when multiple lumbar and sacral nerve roots are compressed hindering sensation and movement 3. The most common cause of cauda equina syndrome is a herniated lumbar disc. Other conditions associated with cauda equina include tumors, trauma, spinal stenosis, spinal epidural abscess, a history of spinal surgery, malignancy, and spinal manipulation 4. CES can occur as an acute pathology or can be more chronic. While there is no agreed upon definition for CES, the most characteristic features include severe lower back pain, lower extremity weakness, reduced perineal sensation (saddle anesthesia), bowel or bladder dysfunction, and loss of sexual function5. Physical exam should focus on assessing the L2-S4 nerve distributions, which can be accomplished by testing strength and sensation of the lower extremities, patellar and Achilles reflex, perineal sensation, anal wink reflex, post void residual with bedside ultrasound, and bulbocavernosus reflex 4. The sensitivity and specificity for any single characteristic feature of CES is poor (Table 1), therefore a good physical exam and history is imperative. If CES is suspected the imaging modality of choice is MRI. When CES is suspected, a prompt neurosurgical or orthopedic spine consult is warranted. In the emergency department, analgesia should be given. Only in cases of spinal compression due to a tumor should steroids also be administered 5-7.
Spinal Epidural Abscesses and Vertebral Osteomyelitis
Vertebral osteomyelitis is an infection of the vertebrae secondary to pyogenic organisms, which can include bacterial, fungi, and mycobacteria 8. A spinal epidural abscess (SEA) is an abscess that is enclosed within the spinal column and can compress the spinal cord, thus causing pain and associated neurologic symptoms. Both are usually seen in patients over 50 and both have a predilection for males. Other risk factors that should increase suspicion for either infectious process includes a history of spinal disease, previous spinal surgeries, paraspinal injections diabetes, trauma, intravenous drug use, malnutrition, alcoholism, tattooing, contiguous spread from another infection and immunosuppression 9-13. Clinically, back pain is the most common symptom and spinal tenderness is often found in conjunction 14. The pain is generally insidious in onset and approximately 30% of patients will have an associated neurologic deficit. The neurologic deficit, when present, can include a wide variety of symptoms including weakness, bowel and bladder dysfunction, nerve root pain similar to the presentation of sciatica, paralysis, sensory changes, and radiculopathy 15. Other nonspecific findings are common, including fatigue, fever, nausea, and vomiting. Like sciatica, this pain may radiate to different locations including the leg. Local tenderness to percussion is the most useful clinical sign. The classic triad of a SEA is fever, back pain, and a neurologic deficit. It is rare, however, for all three symptoms to be present (~13% of cases) 14,15. A full neurological exam is important in diagnosis. In a patient without the above risk factors and a baseline neurologic exam no further workup is needed. If a neurologic deficit is present an emergent MRI of the spine with and without contrast should be obtained. If a patient has one or more of the risk factors above it is important to obtain laboratory studies which should include inflammatory markers (e.g., ESR and CRP). These have been reported to have high sensitivities (> 95%), but they do have low specificities. Blood cultures should be obtained as well. The most sensitive imaging modality for diagnosis is MRI with a sensitivity of 96% and specificity of 94% for vertebral osteomyelitis and 90% specificity and sensitivity for SEA. When imaging the spine, it is important to image the entire spine, as there is a possibility for skip lesions particularly in an SEA 16. On the other hand, CT is 67% sensitive and 50% specific for vertebral osteomyelitis and is only 6% sensitive for SEA 10,17-20. While definitive treatment should be guided by biopsy or culture, empiric treatment in the emergency department should be directed toward the most common causes of spinal infections including staphylococci (most common), streptococci, and gram-negative bacilli. An appropriate regimen includes vancomycin and a cephalosporin generally ceftriaxone, or in those with a severe allergy vancomycin and ciprofloxacin or moxifloxacin can be used 21.
Necrotizing Skin and Soft Tissue Infections
Necrotizing skin and soft tissue infections (NSTI) are characterized by fulminant tissue destruction. This may include involvement of the fascia and muscle, which can mimic sciatica in the early phases. Risk factors for a necrotizing infection can include any skin or mucosal breach, recent surgery, malignancy, obesity, alcoholism, immunosuppression, and diabetes, the latter of which is a particularly important risk factor 22-24. The clinical findings of necrotizing infections can include those typically found in skin infections including soft tissue edema, crepitus, erythema, pain, and tenderness, as well as fever and skin bullae. Laboratory results are often non-specific but in addition to basic laboratory tests inflammatory markers, a lactic acid, blood cultures, and creatinine kinase should be drawn. If there is crepitus on exam and a necrotizing infection is suspected, imaging should not delay surgical intervention, which is the mainstay of treatment alongside antibiotics. If the diagnosis is unclear, CT is the test of choice. These infections are often polymicrobial and treatment should be against gram positive, gram negative, and anaerobic pathogens. An empiric regimen can include a carbapenem or piperacillin-tazobactam as well as vancomycin and clindamycin 22.
Malignancy/Metastatic Cancer
The spine is the most common site for musculoskeletal tumors. More commonly, the spine serves as a site for metastatic cancer to spread, which often comes from breast, lung, and prostate cancer. Red flag symptoms that should prompt consideration or evaluation for underlying malignancy include a history of prior malignancy, unexplained weight loss, pain that is worse at night, pain that is present at rest, pain in multiple sites, pain that has been present for over a month, older age (generally over 50), general malaise, and multiple cancer risk factors 16. The best imaging study for spinal tumors is an MRI. A plain radiograph will often not show a spinal tumor until a significant amount of destruction has occurred 25. For these patients, emergency department management is often centered around pain control. Definitive management is often based on the type of cancer, extent of the malignancy, and whether cord compression is present.
Acute Limb Ischemia/Acute Aortic Occlusion
Acute limb ischemia (ALI) is defined as a rapid decrease in blood flow to a limb usually due to an arterial occlusion. Occasionally this may be due to an acute aortic occlusion, specifically of the infrarenal abdominal aorta 26. Classically ALI is characterized by the “6 P’s”. These are pain, pulselessness, pallor, poikilothermia, paresthesia, and paralysis 27,28. Given the distribution and description of the pain that patients may provide, an ALI or acute aortic occlusion can be mistaken for sciatica pain. Risk factors for ALI and aortic occlusion include a history of atrial fibrillation, cardiac surgery, structural heart defects, valvular heart disease, peripheral arterial disease, tobacco use, diabetes, aneurysmal disease, and recent catheterization procedure 26,27. It is important to examine both lower extremities especially in terms of assessing their strength, sensation, and pulses. If there is difficulty palpating pulses manually a doppler can be used for better assessment. In patients where there is suspicion for ALI, an ankle-brachial index (ABI) should be obtained. This can be done at the bedside by dividing the highest ankle systolic pressure by the highest brachial systolic pressure per leg. If the value of the ABI is <0.90 there is a decline in limb perfusion 28. Depending upon the degree of ischemia, vascular imaging is generally performed and in most cases a CT angiogram is the best imaging study 28,29. In the case of an immediately threatened extremity in a facility with vascular surgery available, the patient should preferentially undergo further evaluation and treatment in a surgical suite. Treatment of ALI and aortic occlusion in the emergency department includes systemic anticoagulation usually with unfractionated heparin (in the absence of contraindications) to prevent progression of the thrombus. Analgesia is often needed as well. A vascular surgery consult should be initiated promptly once the diagnosis is suspected as ALI/aortic occlusion can be rapidly progressive and lead to ischemia of the lower extremities, the spinal cord, mesenteric ischemia and renal failure and thus requires emergent treatment 26,28,29
Aortic dissection
An aortic dissection occurs when there is a loss of integrity of the intima of the aorta and blood flows into the media of the aorta 30. Aortic dissection has high morbidity and mortality, and up to 38% of cases are missed on initial evaluation 31. Most cases occur in men and the risk increases with age. The most important risk factor for the development of an acute aortic dissection is poorly controlled hypertension. Other common risk factors include a family or genetic history (particularly connective tissues diseases), aortic and aortic valve disease, a history of cardiac surgery, and current or previous trauma. The most common symptom of an aortic dissection is chest pain, but back and abdominal pain are also common. Symptoms are often described as abrupt in onset and severe 32. Aortic dissection can rarely present with sciatic pain, so it is important to keep it on the differential and investigate further in the appropriate situations 31. On physical exam the blood pressure should be measured in both arms and legs as a dissection can lead to a differential in blood pressure. It can also lead to other perfusion deficits including a peripheral pulse deficit and/or neurologic deficits, and thus a thorough neurovascular examination is warranted. If a dissection is suspected, CT aorta is needed emergently, as mortality increases 1-2% per hour. In a hemodynamically unstable patient, an ultrasound can be used to investigate the aorta 32. Consultation with cardiothoracic surgery is recommended in all cases of dissection. A type A dissection (involving the ascending aorta) should be treated surgically. While awaiting surgery, initial treatment should be aimed at preventing aortic rupture by controlling heart rate and blood pressure. For a type B dissection (descending aorta) the same medical management should be followed, and in those with organ injury, surgery may be required 31.
Abdominal aortic aneurysm
An abdominal aortic aneurysm (AAA) is defined as a maximum diameter of the abdominal aorta of greater than 3 cm. A ruptured AAA is characterized by the presence of blood outside the adventitia into the peritoneal cavity or into the retroperitoneum 33. Abdominal aortic aneurysms are more common in males, and the incidence increases with age. Tobacco smoking is the primary modifiable risk factor. Other risk factors include Caucasian race, a family history of a AAA (including a history of connective tissue diseases), a history of other large vessel aneurysms, and atherosclerotic diseases. Symptomatic AAAs most commonly present with severe and persistent pain in the back, abdomen, flank, or groin. This pain may indicate compression or erosion into surrounding structures including the spine 34. Misdiagnosis of a ruptured AAA occurs in up to 60% of cases as symptoms are frequently attributed to other causes including musculoskeletal pathology. On physical exam a palpable pulsatile abdominal mass is the classic exam finding. However, this is only 68% sensitive and 75% specific, and the majority of AAA’s cannot be detected on physical exam 34,35. In symptomatic patients in the ED, US can be utilized to rapidly diagnose a AAA. In the stable patient, CT aorta is recommended, but if unstable, US is the modality of choice 34. If a ruptured AAA is discovered, emergent vascular surgery consultation is warranted, as the treatment of choice is surgical repair. In the ED it is important to establish two large bore IV’s. Management also involves permissive hypotension with a goal systolic blood pressure between 70-90 as long as a patient remains conscious 36.
Evaluation and Management
Sciatica is a clinical diagnosis and a diagnosis of exclusion. It is generally diagnosed by a thorough history and physical exam in combination with consideration of other pathology, including the diagnoses discussed above. While there is no perfect physical exam maneuver to confirm the diagnosis of sciatica, the most commonly used tests include the straight leg raise test and the seated slump test. Below are the reported test characteristics for these tests (Table 2) 37-39.

For patients with presentations consistent with sciatica, the objective in the emergency department is aimed at pain management. While sciatica can be extremely disconcerting for the patient in pain, for the majority of patients, sciatica improves and resolves without any intervention. The most common initial treatment is pain control with medications. Non-steroidal anti-inflammatory medications may provide temporary relief. Physical therapy has also been shown to be beneficial in some patients 40. Corticosteroids may be of limited benefit as they showed modestly improved function but no improvement in pain in acute radiculopathy due to a herniated disc 41. Benzodiazepines and opioids are not recommended in treatment 2,42.
Case Conclusion
The patient had a reassuring neurological and vascular examination, and upon a more detailed history did not provide any red flag or worrisome risk factors. He received a shot of ketorolac and a lidocaine patch which significantly improved his pain. He was subsequently discharged from the emergency department.
Key Points
– Sciatica is a diagnosis of exclusion in the emergency department. It is important to assess for red flag symptoms that should prompt further evaluation.
-Important diagnoses to consider that may mimic sciatica include spinal cord compression, infectious processes, malignancy, and acute limb ischemia.
– There is no perfect test for sciatica. History and physical exam should be used together to make this diagnosis.
– The treatment of sciatica is pain control with NSAIDs. The utility of corticosteroids is controversial. Opioids and benzodiazepines have no proven benefit.
References
- Cook, C. E., et al. (2014). “Risk factors for first time incidence sciatica: a systematic review.” Physiother Res Int 19(2): 65-78.
- Jensen, R. K., et al. (2019). “Diagnosis and treatment of sciatica.” BMJ 367: l6273.
- Park D. Cauda Equina Syndrome 2020;https://orthoinfo.aaos.org/en/diseases–conditions/cauda-equina-syndrome/#:~:text=Cauda%20equina%20syndrome%20occurs%20when,are%20especially%20vulnerable%20to%20damage.
- Greenhalgh, S., et al. (2018). “Assessment and management of cauda equina syndrome.” Musculoskelet Sci Pract 37: 69-74.
- Barraclough K. Cauda equina syndrome. BMJ. 2021;372:n32. doi: 10.1136/bmj.n32.
- Long, B., et al. (2020). “Evaluation and management of cauda equina syndrome in the emergency department.” The American Journal of Emergency Medicine 38(1): 143-148.
- Small, S. A., et al. (2005). “Orthopedic pitfalls: cauda equina syndrome.” The American Journal of Emergency Medicine 23(2): 159-163.
- Momodu II, Savaliya V. Osteomyelitis. [Updated 2022 May 12]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK532250/
- Cook, T. M., et al. (2009). “Major complications of central neuraxial block: report on the Third National Audit Project of the Royal College of Anaesthetists.” Br J Anaesth 102(2): 179-190.
- Cornett, C. A., et al. (2016). “Bacterial Spine Infections in Adults: Evaluation and Management.” JAAOS – Journal of the American Academy of Orthopaedic Surgeons 24(1): 11-18.
- Gaul, C., et al. (2005). “Iatrogenic (para-) spinal abscesses and meningitis following injection therapy for low back pain.” Pain 116(3): 407-410.
- Krishnamohan, P. and J. R. Berger (2014). “Spinal epidural abscess.” Curr Infect Dis Rep 16(11): 436.
- Sethna, N. F., et al. (2010). “Incidence of epidural catheter-associated infections after continuous epidural analgesia in children.” Anesthesiology 113(1): 224-232.
- Bond, A. and F. A. Manian (2016). “Spinal Epidural Abscess: A Review with Special Emphasis on Earlier Diagnosis.” Biomed Res Int 2016: 1614328.
- Davis, D. P., et al. (2004). “The clinical presentation and impact of diagnostic delays on emergency department patients with spinal epidural abscess.” Journal of Emergency Medicine 26(3): 285-291.
- Verhagen, A. P., et al. (2016). “Red flags presented in current low back pain guidelines: a review.” European Spine Journal 25(9): 2788-2802.
- Pineda, C., et al. (2009). “Radiographic imaging in osteomyelitis: the role of plain radiography, computed tomography, ultrasonography, magnetic resonance imaging, and scintigraphy.” Seminars in plastic surgery 23(2): 80-89.
- Rausch, V. H., et al. (2017). “Diagnostic Yield of Multidetector Computed Tomography in Patients with Acute Spondylodiscitis.” Rofo 189(4): 339-346.
- Yang, X., et al. (2019). “Challenges in diagnosis of spinal epidural abscess: A case report.” Medicine 98(5): e14196-e14196.
- Schwab, J. H. and A. A. Shah (2020). “Spinal Epidural Abscess: Diagnosis, Management, and Outcomes.” JAAOS – Journal of the American Academy of Orthopaedic Surgeons 28(21): e929-e938.
- Matsuo, T., et al. (2021). “The STAPH Score: A Predictor of Staphylococcus aureus as the Causative Microorganism of Native Vertebral Osteomyelitis.” Open Forum Infect Dis 8(1): ofaa504.
- Anaya, D. A. and E. P. Dellinger (2007). “Necrotizing soft-tissue infection: diagnosis and management.” Clin Infect Dis 44(5): 705-710.
- Miller, L. G., et al. (2005). “Necrotizing fasciitis caused by community-associated methicillin-resistant Staphylococcus aureus in Los Angeles.” N Engl J Med 352(14): 1445-1453.
- Stevens, D. L. and A. E. Bryant (2017). “Necrotizing Soft-Tissue Infections.” N Engl J Med 377(23): 2253-2265.
- Siemionow, K., et al. (2008). “Identifying serious causes of back pain: cancer, infection, fracture.” Cleve Clin J Med 75(8): 557-566.
- Hines, G. L. and H. H. Liu (2021). “Acute Aortic Occlusion and Its Sequelae: Metabolic, Pathologic Etiology, and Management.” Cardiology in Review 29(2).
- Obara, H., et al. (2018). “Acute Limb Ischemia.” Ann Vasc Dis 11(4): 443-448.
- Olinic, D. M., et al. (2019). “Acute Limb Ischemia: An Update on Diagnosis and Management.” J Clin Med 8(8).
- Acar, R. D., et al. (2013). “One of the most urgent vascular circumstances: Acute limb ischemia.” SAGE Open Medicine 1: 2050312113516110.
- Teo, E. P. and E. M. Isselbacher (2019). 34 – Diseases of the Aorta. Essential Echocardiography. S. D. Solomon, J. C. Wu and L. D. Gillam, Elsevier: 354-368.e351.
- Bekele, E., et al. (2017). “More than just muscle spasms: a rare presentation of aortic dissection.” BMJ Case Reports2017: bcr2016218432.
- Silaschi, M., et al. (2017). “Aortic dissection: medical, interventional and surgical management.” Heart 103(1): 78.
- Gawenda, M. and J. Brunkwall (2012). “Ruptured Abdominal Aortic Aneurysm.” Dtsch Arztebl International109(43): 727-732.
- Ullery, B. W., et al. (2018). “Epidemiology and contemporary management of abdominal aortic aneurysms.” Abdominal Radiology 43(5): 1032-1043.
- Calero, A. and K. A. Illig (2016). “Overview of aortic aneurysm management in the endovascular era.” Seminars in Vascular Surgery 29(1): 3-17.
- Chaikof, E. L., et al. (2018). “The Society for Vascular Surgery practice guidelines on the care of patients with an abdominal aortic aneurysm.” Journal of Vascular Surgery 67(1): 2-77.e72.
- Majlesi, J., et al. (2008). “The sensitivity and specificity of the Slump and the Straight Leg Raising tests in patients with lumbar disc herniation.” J Clin Rheumatol 14(2): 87-91.
- Suri, P., et al. (2011). “The accuracy of the physical examination for the diagnosis of midlumbar and low lumbar nerve root impingement.” Spine (Phila Pa 1976) 36(1): 63-73.
- van der Windt, D., et al. (2010). “Physical examination for lumbar radiculopathy due to disc herniation in patients with low-back pain.” Cochrane database of systematic reviews (Online) 2: CD007431.
- Fritz, J. M., et al. (2021). “Physical Therapy Referral From Primary Care for Acute Back Pain With Sciatica: A Randomized Controlled Trial.” Ann Intern Med 174(1): 8-17.
- Goldberg, H., et al. (2015). “Oral Steroids for Acute Radiculopathy Due to a Herniated Lumbar Disk: A Randomized Clinical Trial.” JAMA 313(19): 1915-1923.
- Ropper, A. H. and R. D. Zafonte (2015). “Sciatica.” N Engl J Med 372(13): 1240-1248






