Author: Brit Long, MD (@long_brit, EM physician at SAUSHEC, USAF) // Edited by: Alex Koyfman, MD (@EMHighAK, EM Attending Physician, UTSW / Parkland Memorial Hospital)
Pulmonary embolism (PE) is classically a life-threatening diagnosis, often considered in the work-up of patients with chest pain or dyspnea. Initial mortality rates of missed, untreated PE has been quoted as high as 26%, based on a 1960 study.1 This disease is common, with 400,000 patients affected with nonfatal PE and another 200,000 patients in the U.S. dying each year from this disease. PE is the third most common cause of death in cardiovascular disease after myocardial infarction and stroke.2,3
With this risk of mortality, physicians through the years have been tasked with diagnosing and managing PE. Unfortunately, no individual risk factor, symptom, or clinical sign can definitively diagnose or exclude PE.4,5 Thus, evaluation for PE often includes clinical decision rules, laboratory tests, and several imaging modalities. These tools have been developed to provide physicians with avenues for the evaluation and diagnosis of PE. However, the availability of these tests has resulted in increased test use and number of PE diagnoses. In particular, the use of D-dimer and computed tomography pulmonary angiography (CTPA) has remarkably increased. However, mortality from PE has not changed with increased rates of diagnosis. With the increased testing for PE and sensitivity of CTPA, the diagnosis of subsegmental PE and incidental PE is increasing.5-7 Controversy currently exists in the use of CTPA for PE and treatment of these lesions.7 Recent literature and guidelines have sought to answer these questions, illuminating the path for proper diagnosis, evaluation, and management.7-11
Diagnosis and Testing
The primary diagnostic utility until the late 1990s was ventilation-perfusion (VQ) scanning. However, CT angiography became widely popular after the Prospective Investigation of Pulmonary Embolism Diagnosis (PIOPED) studies, which demonstrated the ease of use, convenience, and adequate sensitivity of CTPA in the evaluation for PE.12,13 The test could reliably diagnose central PE, but the modality was limited in imaging peripheral pulmonary vasculature.13,14 With the advent of multidetector row CTPA, imaging the subsegmental vasculature became feasible.8,9,13-15
CTPA is the test of choice for the majority of physicians in the evaluation for PE. A survey of emergency physicians found a strong preference for CT in PE evaluation, especially as a first line test for the disease.8,9,15-18 Unfortunately, many physicians order imaging without risk stratification first through utilization of clinical gestalt, Wells criteria, or revised Geneva score (RGS).9,16 Studies have demonstrated the benefit of risk stratification in association with the pulmonary embolism rule-out criteria (PERC). In low risk patients with negative PERC, no further testing is required. If the patient is PERC positive or intermediate risk, then testing with D-dimer is warranted. However, even D-dimer testing in patients with low to intermediate risk is often not completed in preference for imaging.8,9,11 Literature has demonstrated that providers do not adhere to PIOPED II recommendations concerning PE imaging. In one study, CTPA was completed in 45.5% of patients meeting imaging recommendation, defined by revised Geneva score pulmonary embolism likely or if PE unlikely with positive D-dimer.19 In the majority of emergency departments, CTPA is the default test, as it is available at all hours and affords higher diagnostic capability for other diseases that can present with chest pain or dyspnea.8,9
An increase in CT use is immediately apparent upon review of recent years. From 1998 to 2006, the detection of PE rose by approximately 80% (62.1 to 112.3 per 100,000 US adults).8,9,16 However, no change in US mortality from PE was found in the same time period.16,21 As scanning technology has advanced with improved resolution, this test is able to detect smaller clots, particularly small filling defects in subsegmental arteries 2-3 mm in diameter.22 These scanners can now cover the entire thorax in one breath-hold, and imaging of peripheral pulmonary arteries down to fifth order branches is possible.22-24 High quality CTPAs require contrast opacification of 200 Hounsfield units in the pulmonary artery. Normal scanning technique requires the patient to hold his or her breath for several seconds. During the scan, injection of 120 ml of intravenous contrast is completed, which requires a 20 gauge or larger intravenous line.25,26 Four detector CTPA technology demonstrated initial diagnosis rates of 5% for isolated subsegmental pulmonary embolism (SSPE).6 Updated imaging technology with 64-detector CTPA have resulted in rates as high as 12% for SSPE.27
The Dangers of Increased Testing
No improvement in patient outcomes has occurred with increased testing for PE in the inpatient, outpatient, and emergency settings. These tests may be associated with actual patient harm, and more than one third of patients will undergo a second CTPA within 5 years of the first scan.28 Radiation from CT scanning has been associated with higher incidence of malignancy in several populations.29,30 One scan imparts 10-20 mSv of radiation, creating a lifetime risk of fatal cancer of 1 in 500 per CT. In particular, women are at greatest risk due to the presence of radiosensitive breast and thoracic tissue.31-33 In addition to increased radiation, other complications include anaphylaxis and anaphylactoid reactions to contrast. Prior allergy to iodinated contrast is associated with a 6-15% risk of recurrence with second exposure, as compared to 1% risk in patients with no prior reactions.34 Extravasation of contrast into a limb occurs in less than 1 per 500 patients, but this can lead to severe pain and compartment syndrome.35
The intravenous contrast utilized is associated with nephropathy, with greater risk in patients of older age and co-morbidities such as renal disease.35-37 One study demonstrated nephropathy due to contrast in 14% of patients undergoing CTPA for PE evaluation.37 Contrast nephropathy is defined by a rise of serum creatinine by 25% from baseline value, which has been associated with worse outcomes.36-38
Overdiagnosis and treatment can also increase healthcare costs, as the average charge for PE admission increased by $19,000 from 1998 to 2006.39 Warfarin anticoagulation with testing and clinic visits approached $2,700 in 2006, while the new oral anticoagulants approached $3,000 per year.40,41 A 2015 study found the cost of medical care to be lower for low-risk patients with VTE immediately discharged from the ED with rivaroxaban therapy compared with patients receiving heparin and warfarin. Costs at 6 months for the heparin-warfarin group were over $11,000, while those for the new oral anticoagulant approximated $4,800.42
The Quandary: Incidental and Subsegmental PE
Incidental PE
Numerous studies have demonstrated the increased prevalence of incidental emboli with rates varying from 0.5% to 5%, depending largely on the scanning technology, the reviewer of the test, and the population under study (cancer versus non-cancer, as well as inpatient versus outpatient).43-49 One study evaluated incidental PE found in consecutive inpatients imaged with MDCT (the majority 16-slice scanners). Nine out of 28 scans were found to be positive by a thoracic radiologist which were initially read negative by the first reporting radiologist. All of these thrombi were located in segmental or subsegmental vasculature. Unsuspected emboli were found in 5.7% of scans.50 Other studies have found comparable numbers with 64 slice scanners, with a prevalence of unsuspected PE of 4.3% in an inpatient population. Interestingly, these unsuspected PE were present in almost 17% of those over 80 years, with none found in those below 50 years.47,48
These two studies found a strong correlation with malignant disease and incidental PE as well, with rates of 70% in the Gosselin et al. and 83% in the Storto et al. studies.47,48 However, these patients often receive more CT imaging due to the need for disease staging. Tertiary cancer centers have reported similar numbers as these other two studies, with rates of 4% in asymptomatic patients.50 Interestingly, Ritchie found no significant difference in incidental PE prevalence in patients with history of malignancy and patients without cancer.51 However, with these numbers, two points require discussion. First, though these studies have a high prevalence of patients with malignancy and incidental pulmonary embolism, it does not necessarily mean that patients with malignancy are predisposed to PE. Other risk factors require consideration. Second, the finding of incidental PE in a patient with no malignancy should not trigger a hunt for malignancy.53
Subsegmental PE
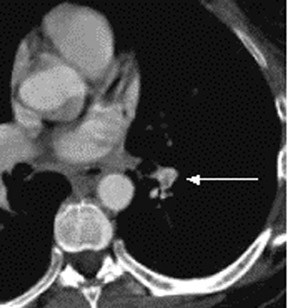
Subsegmental lesions also present a quandary, as the opacity could represent a true PE or artifact with no disease. Numerous studies demonstrate this difficulty. The positive predictive value of CTPA for SSPE may be only 25%, with low inter-observer agreement between radiologists (K 0.38, 95 % CI 0.0–0.89).12,52 A prior study found 11% of SSPE diagnoses were false positive upon a second read by thoracic radiologists.54 One study in Ireland evaluated 937 CTPAs, with PE the initial diagnosis in 174 (18.6% of scans). Investigators found a 25.9% false positive rate, with 59.4% of subsegmental PE read as false positive.55 Studies of PE diagnosis using five different radiologists found that for subsegmental PE, one reviewer dissented with the initial reviewer’s interpretation in 60% of cases. False positive rates were 15% for subsegmental PE and 3.6% for segmental PE.56,57
Suboptimal images are obtained in approximately 10% of formal CTPA interpretations.8,9,12,58 Several reasons exist for this, producing increased false positive rates and decreased specificity. Obesity increases risk of inadequate imaging due to greater amounts of soft tissue. As the pulmonary vasculature tapers, the contrast capture and opacification quality decrease. This increases the risk of artifact, either beam-hardening or with motion due to patient respiration. Increases in false positive rates occur with more peripheral location of lesions, decreased size of the lesion (defined by the short-axis diameter), and decreasing quality of the CT study.9,57
Are Incidental and Subsegmental PEs Harmful?
Controversy exists whether these emboli, including incidental and subsegmental PE specifically, are associated with harm if left untreated. Some argue that small clots do not require treatment, as the lungs function as a sieve to prevent these clots from traveling to the arterial circulation, potentially causing issues such as stroke. Emboli in the lungs are thought to be resorbed by the body without clinical effect.59,60
Postmortem studies have demonstrated a rate of incidental PE ranging from 9% to 63%, with the authors suggesting these emboli were likely not related to the cause of death.60-64 Patients with missed PE who are not anticoagulated may not have an adverse outcome.65 In 25 untreated patients with subsegmental PE, no deaths attributable to PE were found.66 One study suggested that for patients with unacceptable risks of anticoagulation, treatment for incidental PE may be held if adequate pulmonary reserve is present and no lower extremity deep venous thrombus (DVT) is discovered.59 A 2010 study evaluating patients with subsegmental PE found a SSPE rate of 4.7%. Three month VTE risks in patients receiving no treatment were 0.9% (95% CI: 0.4-1.4) and 1.1% (95% CI: 0.7-1.4) for patients who underwent single and multidetector CTPA, respectively.6 These studies support the low risk of adverse outcome and recurrent VTE in patients with isolated SSPE with adequate pulmonary reserves and no DVT.
However, a study by den Exter et al. challenged the notion subsegmental PE is not harmful.66 The authors found that among 116 patients with subsegmental PE and 632 patients with proximal PE, recurrent VTE rates did not differ between the two groups at 3 months, with 3.6% of the SSPE and 2.4% of the proximal PE groups experiencing recurrent VTE. The risk of all-cause mortality was 10.3% for SSPE, 6.3% for proximal PE, and 5.4% for patients without PE. These rates were not statistically significant, though the investigators stated patients with SSPE to be at increased risk for VTE at follow up with a hazard ratio 3.8 (95% CI: 1.3-11.1). The authors of this study conclude that SSPE has similar clinical course and outcomes to that of proximal PE, specifically mortality and recurrent VTE.66 These results should be considered carefully, as the patients in the SSPE group experienced greater mortality when compared to patients with proximal PE. An important aspect of this study is the prevalence of comorbidities present in the SSPE and proximal PE patients, including higher rates of malignancy than the standard population. Many of the patients included had concomitant DVT as well, and most providers would agree DVT in the setting of SSPE requires treatment.
A Cochrane review released in 2016 evaluating the literature up to December 2015 found no studies that met inclusion criteria. The investigators state there is no randomized control trial evidence for anticoagulation versus no treatment in patients with SSPE or incidental PE. No conclusions were provided.67
Treatment
The standard of care in the initial management of VTE has been heparin since the late 1950s.1 Until recently, all patients with PE have been anticoagulated in a similar manner, no matter the location, number, and size of the thrombus, and the majority of patients were admitted for treatment. However, recent literature has now supported outpatient treatment for patients low risk utilizing risk stratification and shared decision making. New oral anticoagulants are gaining popularity for anticoagulation, and patients at low-risk for adverse outcome may be safe for discharge with these anticoagulant agents.68-70 Little argument exists for treating segmental PE.8,9 However, the dilemma occurs when considering management of the patient with subsegmental PE.
Some guidelines recommend anticoagulation for all emboli found, but others acknowledge that anticoagulation may not be warranted in all cases due to the uncertainty in treating isolated subsegmental PE.71,72 Many patients with SSPE may be treated without benefit, as well as increased risk.73 Donato in 2010 found patients with SSPE not anticoagulated experienced a recurrence and mortality rate of 0% at 3 month follow up.74 Experts in thromboembolism have recommended not anticoagulating patients with subsegmental PE with adequate pulmonary reserve.75
In the setting of subsegmental PE, providers must balance the risks and benefits of treatment, namely increased bleeding risk versus reduced recurrent VTE. In some studies, these bleeding complications are more common than recurrent thromboembolism, which is the condition treatment is meant to prevent. 74-76 In one case series of patients with subsegmental PE anticoagulated, risk of major bleeding was 5.3%, compared to recurrent thromboembolism at 0.7%.74 Another study found increased complications from anticoagulation in patients admitted to hospital from PE increased to 5.3% from 3.1 % in an 8 year period.21
The American College of Chest Physicians (ACCP) has recently updated treatment guidelines, published in 2016, providing recommendations on treatment of SSPE:10
-In patients with subsegmental PE (no proximal pulmonary artery involvement), no proximal DVT, and low risk for recurrent VTE, clinical surveillance over anticoagulation is recommended (Grade 2C).
-If high risk for recurrent VTE, anticoagulation is recommended (Grade 2C).* In this setting, ultrasound of the lower extremities should be obtained to exclude proximal DVT.
*The following are at risk for recurrent VTE: patients who are hospitalized or have reduced mobility for another reason; have active cancer (metastatic or being treated with chemotherapy); or have no reversible risk factor for VTE such as recent surgery. A low cardiopulmonary reserve or marked symptoms that cannot be attributed to another condition favor anticoagulant therapy, whereas a high risk of bleeding favors no anticoagulant therapy.
The ACCP cites two reasons for uncertainty in anticoagulation SSPE. First, the abnormality is small and may be a false-positive, rather than a segmental PE. Second, if a true subsegmental PE resulted from a small DVT, these are likely to have small risk of progression or recurrent VTE.10
A diagnosis of true subsegmental PE is more likely a true positive in the setting of high quality CTPA with good contrast opacification of distal pulmonary vasculature, multiple intraluminal defects are present, the defects also include proximal vessels, defects are present on multiple slices/images, contrast surrounds the defects (rather than the defect adhering to the vascular wall), patients are experiencing symptoms, a high-pretest probability is present for PE, and the D-dimer level is elevated.4,5,8-10,75
Treatment is recommended if the patient has multiple SSPE, symptoms are present, or if a DVT is also present. On the other hand, if the SSPE is a single lesion, a DVT is not present, and the patient has no symptoms related to PE, treatment may be deferred. 4,5,8-10,75
What can the provider do?
How can providers improve the current state of PE diagnosis and management?11 The first, and most important step, involves engaging the patient in a shared decision making model for the diagnostic process. This may reduce testing, which has been demonstrated in patients with chest pain.77
The second aspect of PE management involves understanding that PEs will be missed. Unfortunately, emergency physicians cannot diagnose every PE. However, in the appropriate patient who is low risk for adverse outcome, this may not harm the patient, but increased testing and over treatment is associated with risk. Over testing can potentially harm the patient through increased radiation and anticoagulation.
When evaluating the patient with concern for PE, patient risk stratification is essential utilizing Wells criteria, RGS, or clinical gestalt. Approximately two thirds of patients require no testing with adequate risk stratification.8,9 A combination of low risk and low pretest probability in association with negative PERC rule produces a probability of PE of less than 2% (approximately 0.3%), where the risk of further testing outweighs the risk of failure to diagnose PE. 78-81 The PERC rule has demonstrated sensitivities of 97% in a meta-analysis of 14,844 patients.78 However, the PERC rule should not be applied to intermediate or high risk patients.8,9
If the patient is intermediate risk or PERC positive, obtain quantitative D-dimer. The D-dimer possesses a serum half-life of 8 hours and is abnormally elevated for 3 days following symptomatic thrombus.82-85 Ensure the D-dimer is age-adjusted for patients over age 50 years. A threshold of age X 10 ng/mL results in sensitivities greater than 97% in all patients, no matter with ages over 50 years, while also increasing the specificity of the test.86,87
With high-risk patients, imaging with CTPA is recommended for evaluation. If concern for radiation, contrast nephropathy, or contrast reaction is present, consider the use of bilateral DVT study and/or ventilation-perfusion scan.8,9
If a SSPE is discovered, several components should be evaluated: the patient, the scan, and other tests.10 This can be completed through the consideration of the following: 1) Evaluate the patient for risk factors (immobility, hospitalization, comorbidities) and symptoms. 2) Speak with the radiologist about the study obtained, and question whether artifacts could be present. 3) Consider other imaging tests such as bilateral DVT studies. If CTPA demonstrates SSPE, obtain DVT studies to evaluate for other thrombi. Presence of DVT is a marker for recurrent VTE and predictor of mortality for patients with PE.4,5,75
In the setting of SSPE, treatment is likely warranted for patients with symptoms or risk factors, multiple SSPE on high quality scan, or a DVT with SSPE. On the other hand, if the patient has adequate cardiopulmonary reserve, no DVT is found on other imaging modalities, and persistent risk factors are not present, further treatment may be held until follow up.4,5,7,10
Summary
CTPA is the first-line imaging modality for PE and has developed the ability to image pulmonary vasculature down to fifth order branches. However, this test is not without risk including radiation, contrast reaction, and contrast nephropathy. With the ever-increasing imaging capability of CTPA, the diagnosis of incidental and subsegmental PE is increasing, Controversy has occurred concerning the potential dangers of these lesions and whether treatment is warranted. The literature differs in demonstrating harm of SSPE. Fortunately, the ACCP recently released updated guidelines for the treatment of SSPE taking into account the patient, the scan, and other imaging modalities. Ultimately, providers must reign in testing and use risk stratification, in association with shared decision making, in the evaluation and treatment of PE.
References/Further Reading
- Barritt DW, Jordan SC. Anticoagulant drugs in the treatment of pulmonary embolism: a controlled trial. Lancet 1960;1(7138):1309–12.
- Cushman M, Tsai AW, White RH, Heckbert SR, Rosamond WD, Enright P, et al. Deep vein thrombosis and pulmonary embolism in two cohorts: the longitudinal investigation of thromboembolism etiology. Am J Med 2004 Jul;117(1):19-25.
- Heit JA. The epidemiology of venous thromboembolism in the community: implications for prevention and management. J Thromb Haemost 2006 Feb;21(1):23-29.
- Chunilal SD, Eikelboom JW, Attia J, Miniati M, Panju AA, Simel DL, et al. Does this patient have pulmonary embolism? JAMA 2003;290:2849-58.
- Carrier M, Righini M, Le GG (2012) Symptomatic subsegmental pulmonary embolism: what is the next step? J Thromb Haemost 10:1486–1490
- Carrier M, Righini M, Wells PS, Perrier A, Anderson DR, Rodger MA, Pleasance S, Le GG. Subsegmental pulmonary embolism diagnosed by computed tomography: incidence and clinical implications. A systematic review and meta-analysis of the management outcome studies. J Thromb Haemost 2010;8:1716–1722.
- Raskob GE. Importance of subsegmental pulmonary embolism. Blood 2015; 122(7):1095-1096.
- Raja AS, Greenberg JO, Qaseem A, Denberg TD, Fitterman N, Schuur JD, et al. Evaluation of Patients With Suspected Acute Pulmonary Embolism: Best Practice Advice From the Clinical Guidelines Committee of the American College of Physicians. Ann Intern Med 2015;163:701-711.
- Kline JA and Kabrhel C. Emergency Evaluation for Pulmonary Embolism, Part 2: Diagnostic Approach. JEM 2015;49(1):104-117.
- Kearon C, Akl EA, Ornelas J, Blaivas A, et al. Antithrombotic Therapy for VTE Disease: CHEST Guideline. Chest 2016. DOI: 10.1016/j.chest.2015.11.026
- Green SM, Yealy DM. Right-sizing testing for pulmonary embolism: recognizing the risks of detecting any clot. Ann Emerg Med 2012; 59:524-526.
- Stein PD, Fowler SE, Goodman LR, et al, for the PIOPED II Investigators. Multidetector computed tomography for acute pulmonary embolism. N Engl J Med 2006;354:2317-2327.
- Mullins MD, Becker DM, Hagspiel KD, Philbrick JT. The role of spiral volumetric computed tomography in the diagnosis of pulmonary embolism. Arch Intern Med 2000;160(3):293-8.
- Rathbun SW, Raskob GE, Whitsett TL. Sensitivity and specificity of helical computed tomography in the diagnosis of pulmonary embolism: a systematic review. Annals of Internal Medicine 2000;132(3):227–32.
- Le Gal G, Righini M, Parent F, van Strijen M, Couturaud F. Diagnosis and management of sub-segmental pulmonary embolism. Journal of Thrombosis and Haemostasis 2006;4 (4):724-31.
- Wiener RS, Schwartz LM, Woloshin S. When a test is too good: how CT pulmonary angiograms find pulmonary emboli that do not need to be found. The BMJ 2013;347:f3368
- Weiss CR, Scatarige JC, Diette GB, Haponik EF, Merriman B, Fishman EK. CT pulmonary angiography is the first-line imaging test for acute pulmonary embolism: a survey of US clinicians. Acad Radiol 2006;13:434-46.
- Smith-Bindman R, Miglioretti DL, Johnson E, Lee C, Feigelson HS, Flynn M, et al. Use of diagnostic imaging studies and associated radiation exposure for patients enrolled in large integrated health care systems, 1996-2010. JAMA 2012;307:2400-9.
- Adams DM, Stevens SM, Woller SC, Evans RS, Lloyd JF, Snow GL, Allen TL, Bledsoe JR, Brown LM, Blagev DP, Lovelace TD, Shill TL, Conner KE, Aston VT, Elliott CG. Adherence to PIOPED II investigators’ recommendations for computed tomography pulmonary angiography. Am J Med 2013 Jan;126(1):36-42.
- Ikesaka R and Carrier M. Clinical significance and management of subsegmental pulmonary embolism. J Thromb Thrombolysis 2015;39:311–314.
- Wiener RS, Schwartz LM, Woloshin S. Time trends in pulmonary embolism in the United States: evidence of overdiagnosis. Arch Intern Med 2011;171:831-7.
- Ghaye B. Peripheral pulmonary embolism on multidetector CT pulmonary angiography. JBR-BTR 2007;90:100-8.
- Ghaye B, Szapiro D, Mastora I, et al. Peripheral pulmonary arteries: how far in the lung does multi- detector row spiral CT allow analysis? Radiology 2001;219:629–36.
- Raptopoulos V, Boiselle PM. Multi-detector row spiral CT pulmonary angiography: comparison with single-detector row spiral CT. Radiology 2001;221:606–13.
- Burnside P, Kline JA. Indirect computed tomography venography: quality of vascular opacification and diagnostic implications. Emerg Radiol 2010;17:195–202.
- Mos IC, Klok FA, Kroft LJ, et al. Safety of ruling out acute pulmonary embolism by normal computed tomography pulmonary angiography in patients with an indication for computed tomography: systematic review and meta-analysis. J Thromb Haemost 2009;7: 1491–8.
- Pesavento R, de Conti G, Minotto I, Filippi L, Mongiat M, de Faveri D, Maurizi F, Dalla VF, Piovella C, Pagnan A, Prandoni P (2011) The value of 64-detector row computed tomography for the exclusion of pulmonary embolism. Thromb Haemost 105:901–907.
- Kline JA, Courtney DM, Beam DM, et al. Incidence and predictors of repeated computed tomographic pulmonary angiography in emergency department patients. Ann Emerg Med 2009;54:41–8.
- Amis ES Jr,Butler PF,Applegate KE, Birnbaum SB, Brateman LF, Hevezi JM, et al, American College of Radiology. American College of Radiology white paper on radiation dose in medicine. J Am Coll Radiol. 2007;4:272-84.
- Brenner DJ, Hall EJ. Computed tomography—an increasing source of radiation exposure. N Engl J Med. 2007;357:2277-84.
- Einstein AJ, Henzlova MJ, Rajagopalan S. Estimating risk of cancer associated with radiation exposure from 64-slice computed tomography coronary angiography. JAMA 2007;298:317–23.
- Health Physics Society. Radiation exposure from medical diag- nostic imaging procedures Health Physics Society fact sheet. McLean, VA: Health Physics Society; 2008.
- Parker MS, Hui FK, Camacho MA, et al. Female breast radiation exposure during CT pulmonary angiography. AJR Am J Roentgenol 2005;185:1228–33.
- Kopp AF, Mortele KJ, Cho YD, et al. Prevalence of acute reactions to iopromide: postmarketing surveillance study of 74,717 patients. Acta Radiol 2008;49:902–11.
- Mitchell AM, Jones AE, Tumlin JA, et al. Immediate complications of intravenous contrast for computed tomography imaging in the outpatient setting are rare. Acad Emerg Med 2011;18:1005–9.
- Mitchell AM,Jones AE,Tumlin JA, Kline JA. Incidence of contrast-induced nephropathy after contrast-enhanced computed tomography in the outpatient setting. Clin J Am Soc Nephrol. 2010;5:4-9.
- Mitchell AM, Jones AE, Tumlin JA, Kline JA. Prospective study of the incidence of contrast-induced nephropathy among patients evaluated for pulmonary embolism by contrast-enhanced computed tomography. Acad Emerg Med 2012;19:618-25
- Sinert R, Brandler E, Subramanian RA, et al. Does the current definition of contrast-induced acute kidney injury reflect a true clinical entity? Acad Emerg Med 2012;19:1261–7.
- Park B, Messina L, Dargon P, Huang W, Ciocca R, Anderson FA. Recent trends in clinical outcomes and resource utilization for pulmonary embolism in the United States: findings from the nationwide inpatient sample. Chest 2009;136:983-90.
- Bullano MF, Willey V, Hauch O, Wygant G, Spyropoulos AC, Hoffman L. Longitudinal evaluation of health plan cost per venous thromboembolism or bleed event in patients with a prior venous thromboembolism event during hospitalization. J Manag Care Pharm 2005;11:663-73.
- Gage BF. Cost of dabigatran for atrial fibrillation. BMJ 2011;343:d6980.
- Kahler ZP, Beam DM, Kline JA, Hiestand B. Cost of Treating Venous Thromboembolism With Heparin and Warfarin Versus Home Treatment With Rivaroxaban. Acad Emerg Med 2015;22(7):796-802.
- Sebastian AJ, Paddon AJ. Clinically unsuspected pulmonary embolism: an important secondary finding in oncology CT. Clin Radiol 2006;61:81–5.
- Verschakelen JA, Vanwijck E, Bogaert J, et al. Detection of unsuspected central pulmonary embolism with conventional contrast-enhanced CT. Radiology 1993;188:847–50.
- Patel JJ, Chandrasekaran K, Maniet AR, et al. Impact of incidental diagnosis of clinically unsuspected central pulmonary artery thromboembolism in treatment of critically ill patients. Chest 1994;105:986–90.
- Winston CB, Wechsler RJ, Salazar AM, et al. Incidental pulmonary emboli detected at helical CT: effect on patient care. Radiology 1996;201:23–7.
- Gosselin MV, Rubin GD, Leung AN, et al. Unsuspected pulmonary embolism: prospective detection on routine helical CT scans. Radiology 1998;208:209–15.
- Storto ML, Di Credico A, Guido F, et al. Incidental detection of pulmonary emboli on routine MDCT of the chest. AJR Am J Roentgenol 2005;184:264–7.
- Engelke C, Manstein P, Rummeny EJ, et al. Suspected and incidental pulmonary embolism on multidetector-row CT: analysis of technical and morphological factors influencing the diagnosis in a cross-sectional cancer centre patient cohort. Clin Radiol 2006;61:71–80.
- Gladish GW, Choe DH, Marom EM, et al. Incidental pulmonary emboli in oncology patients: prevalence, CT evaluation, and natural history. Radiology 2006;240:246–55.
- Ritchie G, McGurk S, McCreath C, et al. Prospective evaluation of unsuspected pulmonary embolism on contrast enhanced CT (MDCT) scanning. Thorax 2007;62:536–40.
- Ghanima W, Nielssen BE, Holmen LO, Witwit A, Al-Ashtari A, Sandset PM. Multidetector computed tomography (MDCT) in the diagnosis of pulmonary embolism: interobserver agreement among radiologists with varied levels of experience. Acta Radiol 2007;48:165–170.
- Desai SR. Unsuspected pulmonary embolism on CT scanning: yet another headache for clinicians? Thorax 2007;62:470–472.
- Perrier A, Desmarais S, Miron MJ, de Moerloose P, Lepage R, Slosman D, Didier D, Unger PF, Patenaude JV, Bounameaux H. Non-invasive diagnosis of venous thromboembolism in outpatients. Lancet 1999;353:190–195.
- Hutchinson BD, Navin P, Marom EM, et al. Overdiagnosis of pulmonary embolism by pulmonary CT angiography. AJR Am J Roentgenol 2015;205:271-277.
- Gurney JW. No fooling around: direct visualisation of pulmonary embolism. Radiology 1993;188:618–9.
- Miller WT Jr, Marinari LA, Barbosa E Jr, et al. Small pulmonary artery defects are not reliable indicators of pulmonary embolism. Ann Am Thorac Soc 2015;12:1022-1029
- Courtney DM, Miller CD, Smithline HA, et al. Prospective multicenter assessment of interobserver agreement for radiologist inter- pretation of multidetector CT angiography for pulmonary embo- lism. J Thromb Haemost 2010;8:533–40.
- Goodman LR. Small pulmonary emboli: what do we know? Radiology 2005;23:654-8.
- Stein PD, Henry JW. Prevalence of acute pulmonary embolism among patients in a general hospital and at autopsy. Chest 1995;108:978–81.
- Bergqvist D, Lindblad B. A 30-year survey of pulmonary embolism verified at autopsy: an analysis of 1274 surgical patients. Br J Surg 1985;72:105–8.
- Cullen DJ, Nemeskal AR. The autopsy incidence of acute pulmonary embolism in critically ill surgical patients. Intensive Care Med 1986;12:399–403.
- Lindblad B, Sternby NH, Bergqvist D. Incidence of venous thromboembolism verified by necropsy over 30 years. BMJ 1991;302:709–11.
- Lindblad B, Eriksson A, Bergqvist D. Autopsy- verified pulmonary embolism in a surgical department: analysis of the period from 1951 to 1988. Br J Surg 1991;78:849–52.
- Engelke C, Rummeny EJ, Marten K. Pulmonary embolism at multi-detector row CT of chest: one- year survival of treated and untreated patients. Radiology 2006;239:563–75.
- Eyer BA, Goodman LR, Washington L. Clinicians’ response to radiologists’ reports of isolated subsegmental pulmonary embolism or inconclusive interpretation of pulmonary embolism using MDCT. AJR Am J Roentgenol 2005;184:623–8.
- den Exter PL, van Es J, Klok FA, Kroft LJ, Kruip MJ, Kamphuisen PW, Büller HR, Huisman MV. Risk profile and clinical outcome of symptomatic subsegmental acute pulmonary embolism. Blood. 2013 Aug 15;122(7):1144-9; quiz 1329.
- Buller HR, Prins MH, Lensing AW, et al; EINSTEIN–PE Investigators. Oral rivaroxaban for the treatment of symptomatic pulmonary embolism. N Engl J Med 2012;366(14):1287-1297.
- Agnelli G, Buller HR, Cohen A, et al; AMPLIFY- EXT Investigators. Apixaban for extended treatment of venous thromboembolism. N Engl J Med 2013;368(8): 699-708.
- Beam DM, Kahler ZP, Kline JA, Hiestand BC. Immediate Discharge and Home Treatment With Rivaroxaban of Low-risk Venous Thromboembolism Diagnosed in Two U.S. Emergency Departments: A One-year Preplanned Analysis. Acad Emerg Med 2015;22(7):788-795.
- Remy-Jardin M, Pistolesi M, Goodman LR, Gefter WB, Gottschalk A, Mayo JR, et al. Management of suspected acute pulmonary embolism in the era of CT angiography: a statement from the Fleischner Society. Radiology 2007;245:315-29.
- Torbicki A, Perrier A, Konstantinides S, Agnelli G, Galie N, Pruszczyk P, et al. Guidelines on the diagnosis and management of acute pulmonary embolism: the Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology (ESC). Eur Heart J 2008;29:2276-315.
- Prasad V, Rho J, Cifu. The diagnosis and treatment of pulmonary embolism. A metaphor for medicine in the evidence-based medicine era. Arch Intern Med 2012;172(12):955–8.
- Donato AA, Khoche S, Santora J, Wagner B. Clinical outcomes in patients with isolated subsegmental pulmonary emboli diagnosed by multidetector CT pulmonary angiography. Thromb Res 2010;126:e266-70.
- Stein PD, Goodman LR, Hull RD, Dalen JE, Matta F. Diagnosis and management of isolated subsegmental pulmonary embolism: review and assessment of the options. Clin Appl Thromb Hemost 2012;18:20-6.
- Wysowski DK, Nourjah P, Swartz L. Bleeding complications with warfarin use: a prevalent adverse effect resulting in regulatory action. Arch Intern Med 2007;167:1414-9.
- Hess EP, Knoedler MA, Shah ND, Kline JA, Breslin M, Branda ME, Pencille LJ, Asplin BR, Nestler DM, Sadosty AT, Stiell IG, Ting HH, Montori VM. The chest pain choice decision aid: a randomized trial. Circ Cardiovasc Qual Outcomes 2012 May;5(3):251-9.
- Singh B, Mommer SK, Erwin PJ, et al. Pulmonary embolism rule- out criteria (PERC) in pulmonary embolism–revisited: a systematic review and meta-analysis. Emerg Med J 2013;30:701–6.
- Fesmire FM, Brown MD, Espinosa JA, et al. Critical issues in the evaluation and management of adult patients presenting to the emergency department with suspected pulmonary embolism. Ann Emerg Med 2011;57:628–52.
- Penaloza A, Verschuren F, Dambrine S, et al. Performance of the pulmonary embolism rule-out criteria (the PERC rule) combined with low clinical probability in high prevalence population. Thromb Res 2012;129:e189–93.
- Bokobza J, Aubry A, Nakle N, et al. Pulmonary embolism rule-out criteria vs D-dimer testing in low-risk patients for pulmonary em- bolism: a retrospective study. Am J Emerg Med 2014;32:609–13.
- Couturaud F, Kearon C, Bates SM, Ginsberg JS. Decrease in sensitivity of D-dimer for acute venous thromboembolism after starting anticoagulant therapy. Blood Coagul Fibrinolysis 2002; 13:241–6.
- Taira T, Taira BR, Carmen M, et al. Risk of venous thromboembolism in patients with borderline quantitative D-dimer levels. Am J Emerg Med 2010;28:450–3.
- Kutinsky I, Blakley S, Roche V. Normal D-dimer levels in patients with pulmonary embolism. Arch Intern Med 1999;159:1569–72.
- Runyon MS, Gellar MA, Sanapareddy N, et al. Development and comparison of a minimally-invasive model of autologous clot pul- monary embolism in Sprague-Dawley and Copenhagen rats. Thromb J 2010;8:3.
- Schouten HJ, Geersing GJ, Koek HL, Zuithoff NP, Janssen KJ, Douma RA, et al. Diagnostic accuracy of conventional or age adjusted d-dimer cut-off values in older patients with suspected venous thromboembolism: systematic review and meta-analysis. BMJ 2013; 346:f2492.
- Righini M, Van Es J, Den Exter PL, Roy PM, Verschuren F, Ghuysen A, et al. Age-adjusted d-dimer cutoff levels to rule out pulmonary embolism: the ADJUST-PE study. JAMA 2014; 311:1117-24.






5 thoughts on “Controversies in Pulmonary Embolism Imaging and Treatment of Subsegmental Thromboembolic Disease”
Pingback: Subsegmentell Lungemboli | Mind palace of an ER doc
Pingback: Pulmonary Embolism Challenges in Diagnosis Part 1 | Emergency Medicine Cases
Pingback: PE Challenges in Diagnosis | Imaging, Subsegmental, Pregnancy | EM Cases
Pingback: 2018'in en iyi makaleleri | Acilci.Net
Pingback: Should We Anticoagulate Subsegmental PE? – JournalFeed