Author: Lloyd Tannenbaum, MD (Emergency Medicine Resident, San Antonio, TX) // Edited by: Jamie Santistevan, MD (@jamie_rae_EMdoc – EM Physician, Presbyterian Hospital, Albuquerque, NM); Manpreet Singh, MD (@MPrizzleER – Assistant Professor of Emergency Medicine / Department of Emergency Medicine – Harbor-UCLA Medical Center); and Brit Long, MD (@long_brit – EM Attending Physician, San Antonio, TX)
Welcome to this edition of ECG Pointers, an emDOCs series designed to give you high yield tips about ECGs to keep your interpretation skills sharp. For a deeper dive on ECGs, we will include links to other great ECG FOAMed!
Welcome back to emDOCS ECG points. This week we are continuing our Syncope Series with a discussion of Hypertrophic Cardiomyopathy (HCM). Interestingly, this disease process has gone by many other names, such as Hypertrophic Obstructive Cardiomyopathy (HOCM) or Idiopathic Hypertrophic Subaortic Stenosis (IHSS), but currently, HCM is the preferred term for this disease state. As a quick refresher, any patient that presents with syncope should have a thorough history and physical exam performed and an ECG. These recommendations are based on the ACC/AHA/HRS Guideline for the Evaluation and Management of Patients With Syncope, which has been endorsed by ACEP as guidelines for clinical practice [1]. As previously discussed, I screen every ECG for with I get my Dairy Queen At HEB (DQ@HEB) [2]:
D: Delta wave (Wolff-Parkinson-White)
Q: QT Prolongation
@: AV nodal blocks
H: HCM (look for dagger Q waves)
E: Epsilon wave (Arrhymogenic Right Ventricular Dysplasia)
B: Brugada
The Case:
An 20-year-old basic trainee presents with a chief complaint of syncope. He states that he was running on the track and racing his buddy to finish a mile when he was suddenly on the ground with 7 people standing over him screaming his name. He has never had anything like this happen before, but his brother would pass out sometimes when they worked out together. ECG compliments of Dr. Amber Cibrario and is shown below:

This ECG shows normal sinus rhythm with higher than expected voltages for the patient’s age and lack of other medical comorbidities. There are also dagger Q-Waves (thin, pointy) in the precordial and inferior leads, raising concerns for hypertrophic cardiomyopathy.
Discussion:
In 1957, Dr. Donald Teare presented a case series of 8 young patients (age 14-44) who died unexpectedly. Dr. Teare was a British pathologist, and when he performed the autopsies, he noted that all of the patients had inexplicable asymmetric hypertrophy of their cardiac septums (see figure below from his paper). When examined microscopically, they all exhibited seemingly random and disorganized muscle fiber growth. Dr Teare thought this may be some kind of strange cardiac tumor that he had stumbled upon, but it was actually the first recorded case series of Hypertrophic Cardiomyopathy [3].

This image, taken from Dr. Teare’s landmark paper in 1957, shows one of his patient’s hearts [3]. Note the extremely hypertrophied septum! It barely leaves room for the ventricles.
A significant amount of progress has been made in diagnosing and treating Hypertrophic Cardiomyopathy since it was first described by Dr. Teare in 1957. HCM is a genetic disorder that is inherited in an autosomal dominant fashion, and it affects approximately 0.2% of the population (that’s 1 in 500 people!) [4]. There are several different mutations that have been described that cause HCM, but all of the mutations cause a malfunction of the cardiac sarcomeres [5].
In 2011, a panel of experts put together a consensus statement on HCM. Taken directly from their paper, HCM is diagnosed as:
“Maximal LV wall thickness ≥15 mm, with wall thickness of 13 to 14 mm considered borderline, particularly in the presence of other compelling information (e.g., family history of HCM), based on echocardiography” [6].
So, if an echo is the way to make the diagnosis, why do we get an ECG? Great question. ECGs are much cheaper and quicker to get than echos. Also, 75-95% of patients with HCM will have an abnormal ECG, which can help determine which patients with syncope will require further work up [6].
ECG Findings:
A study in 2012 by Dr. Delcrè et al looked at the ECGs of 257 patients with known HCM to evaluate for abnormal findings. Only 6% of patients had a normal ECG, which is consistent with prior studies [6]. This paper screened the ECG for multiple different conditions including prolonged QRS, bundle branch blocks, LVH, prolonged QTc, repolarization abnormalities, ST segment elevation in V1/V2, pathological Q-waves, and absent Q-waves in aVL, V5, and V6. What they found was that 60% of HCM patients have an ECG consistent with LVH, 53% also had pathological Q-waves [7].
What are pathological Q-waves seen in HCM? The table below shows the difference between the typical pathological Q-waves seen in patients with a prior MI and those typical of HCM [8]:
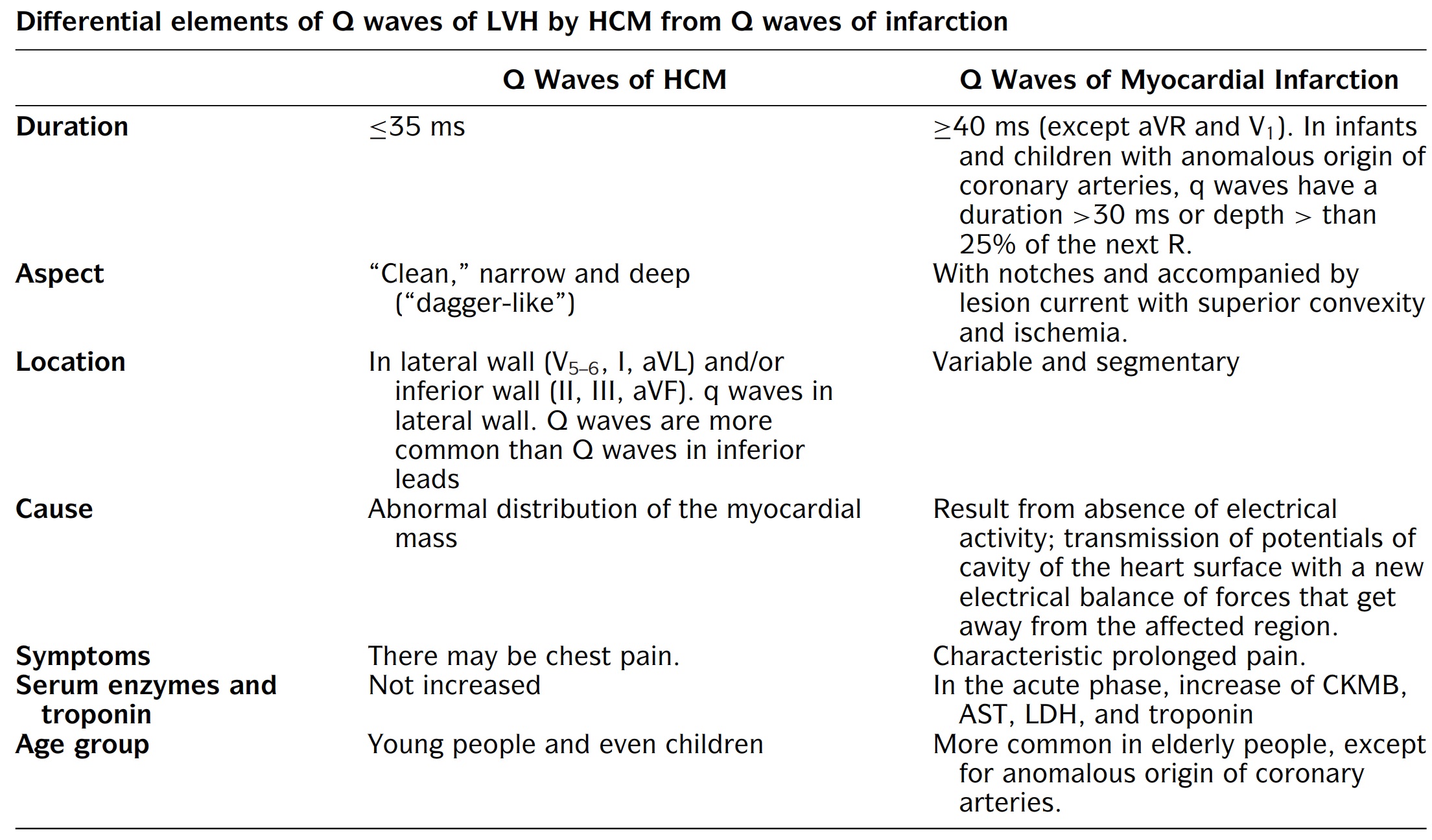
In summary, the Q-waves of HCM will be shorter in duration (making them narrow) and localized to the lateral and sometimes inferior leads. The Q-waves of acute MI will be associated with ischemic changes and ischemic chest pain. The patient population is also different with HCM occurring in children and adolescents.
What are Dagger Q Waves?
WikiEM has an excellent example of Dagger Q-Waves (Q waves that are deep, narrow, and <40 msec), which has been copied below. This seems to be a favorite of board exams [9]:
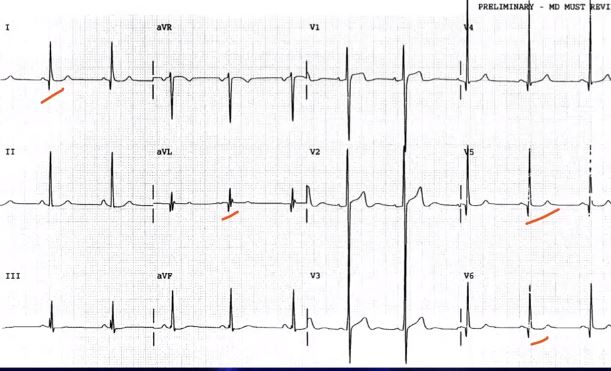
Classic Physical Exam Findings:
Remember back when there was confusion between HOCM and HCM? Where did the “O” go? Maybe you learned about the loss of the “O” while you were standing on turnover rounds, pimping an intern about HOCM, and then the intern turns to you and says, “Well, you know, the proper term is actually HCM…” Well the “O” stands for obstructive, and only 25-40% of patient with HCM have an obstructive component, so cardiologists decided to ditch the “O.” That won’t stop board exams from asking about it though. Let’s review the murmur of HOCM:
During systole, there is an obstruction to aortic outflow, due to the mitral valve and the ventricular septum being so close together. This will cause a systolic murmur with HCM. Since murmurs increase when flow across them increases, the murmur of HCM will INCREASE with a Valsalva or suddenly standing from squatting and will DECREASE with squatting or handrip maneuver [10]. This is because valsalva maneuver acts to decrease venous return, and therefore there is less volume in the left ventricle, which worsens outflow obstruction, making the murmur louder. The same is true when standing suddenly from squatting, venous return decreases and obstruction worsens. Squatting does the opposite by increasing venous return (more blood in the LV), which decreases the amount of obstruction making the murmur quieter. Hand grip works by increasing afterload, which means the amount of blood in the LV is greater, decreasing obstruction and murmur. When there is no obstruction, there shouldn’t be a murmur. Often, due to alterations in the myocardial structure, an S3 or S4 can be heard [10].
How often does syncope occur in patients with HCM? Up to 25% of patients with HCM will experience at least one episode of syncope according to one study [11].
Treatment:
Treating HCM is a topic best left to the cardiologists. As emergency medicine physicians, our job is to catch patients who could have HCM and send them to a cardiologist for further work up and treatment. For completeness sake, a copy of the flowsheet from the 2011 consensus statement is included below to show the wide variety of treatments that cardiologists may use to treat HCM patients. Note the large spectrum from observation to pharmacotherapy to surgical removal of part of the heart muscle wall [6].

Case Conclusions:
This patient was referred to cardiology and had an echocardiogram done the next day, which showed a LV wall thickness of 23 mm. He was diagnosed with HCM, and further treatment was guided by the cardiologists.
References:
- Shen W-K, Sheldon RS, Benditt DG, Cohen MI, Forman DE, Goldberger ZD, Grubb BP, Hamdan MH, Krahn AD, Link MS, Olshansky B, Raj SR, Sandhu RK, Sorajja D, Sun BC, Yancy CW, 2017 ACC/AHA/HRS Guideline for the Evaluation and Management of Patients With Syncope, Journal of the American College of Cardiology (2017), doi: 10.1016/j.jacc.2017.03.003.
- Kann, Kristen. Master Mneumonicer.
- Teare, Donald. Asymmetrical hypertrophy of the heart in young adults. British heart journal 1958;20.1.
- Maron, Barry J. Hypertrophic cardiomyopathy: a systematic review. Jama 2002;287(10):1308-1320.
- Wigle ED, et al. Hypertrophic cardiomyopathy: clinical spectrum and treatment. Circulation 1995;92(7): 1680-1692.
- Gersh, B, Maron, B, Bonow, R et al. 2011 ACCF/AHA Guideline for the Diagnosis and Treatment of Hypertrophic Cardiomyopathy. Journal of the American College of Cardiology Dec 2011;58 (25):e212-e260.
- Delcrè, Sara Dalila Luisella, et al. Relationship of ECG findings to phenotypic expression in patients with hypertrophic cardiomyopathy: a cardiac magnetic resonance study. International journal of cardiology. 2013; 167(3): 1038-1045.
- Pérez‐Riera, Andrés Ricardo, et al. Value of electro‐vectorcardiogram in hypertrophic cardiomyopathy. Annals of Noninvasive Electrocardiology. 2013;18(4): 311-326.
- Eggeman, D, Basso, M, Zonnoor, B, et al. Hypertrophic Cardiomyopathy. https://wikem.org/wiki/File:Needle-like_q-waves.JPG.
- Watrous RL, et al. Computer-assisted detection of systolic murmurs associated with hypertrophic cardiomyopathy: a pilot study. Tex Heart Inst J. 2004; 31(4): 368–375.
- Williams L, Frenneaux M. Syncope in hypertrophic cardiomyopathy: mechanisms and consequences for treatment. Europace. 2007 Sep;9(9):817-22.





