Authors: Elizabeth G. Olson, MD (EM Resident Physician, Atrium Health’s Carolinas Medical Center) and Christine Murphy, MD (EM Attending Physician, Medical Toxicologist, Atrium Health’s Carolinas Medical Center) // Reviewed by: Cynthia Santos, MD (@Cynthia Santos, MD); Alex Koyfman, MD (@EMHighAK); and Brit Long, MD (@long_brit)
Case presentation:
A 39-year-old man is brought to the ED by his wife, moaning and clutching his eyes. He smells terrible. He explains that he had been working outside when he startled a skunk and got sprayed in the face and eyes!
That’s not the worst part. He reached for a jug of vinegar in an attempt to neutralize the stench, only to realize too late that what he was dousing himself with was… bleach. He immediately had intense pain with light sensitivity and blepharospasm.
HR 90, BP 139/88, RR 16, T 98, O2 saturation 97% on room air.
Question: How do we manage caustic eye injuries?
Background:
- Alkali or basic compounds have a high pH
- These cause the most serious ocular chemical injuries through liquefactive necrosis.
- Pain is not a good indicator of degree of injury, as alkali substances cause nerve damage.
- Alkalis erode through the corneal epithelium and penetrate into the anterior chamber.
- These compounds denature proteins and saponify fatty acid, causing collagen breakdown and lysis of keratocytes, the cells responsible for corneal transparency.
- Bleach, lye, lime (present in cement or plaster dust), ammonia, and even airbag deployment can cause alkali injury. 1
- Acidic compounds have a low pH
- These injuries may initially look worse than an alkali burn but are often less severe.
- Classically acids cause coagulation necrosis.
- Common sources of injury include industrial chemical exposure, automobile battery explosions (causing both thermal injury and sulfuric acid exposure), hair care products, and disinfectants.
- Hydrofluoric acid is an exception – it is a weak acid that behaves more like a base, causing liquefactive necrosis, and chelates calcium and magnesium ions from the body, potentially leading to severe cardiac arrhythmias.
Pearls:
- Prompt irrigation is the most important initial therapy, regardless of whether the caustic substance is acidic or alkali.
- Tetracaine ophthalmic drops are safe to use in chemical injuries and will increase the tolerability (and therefore the effectiveness) of the irrigation. Oral pain and anxiety treatment may also be necessary.
- Do not delay irrigation for any reason. It is better to immediately start irrigating away the caustic substance than delay initial care to remove contact lenses and administer tetracaine. Once your supplies are gathered, briefly pause irrigation to remove contacts and administer tetracaine, then resume irrigation.
- Data supports the use of warm lactated ringer’s as the solution best tolerated by patients. 2,3
- However, there is no data to suggest that selection of LR vs normal saline or room-temperature vs warmed fluids has superior outcomes in terms of long-term outcomes such as visual acuity.
- The classic equipment for eye irrigation is the Morgan lens.
- Start the flow of fluid through the Morgan lens before placing it in the patient’s eye. This will allow it to float above the surface of the eye.
- Have the patient look down, then slide the lens under the top lid. Have them look up, then pull the lower lid over the bottom edge of the lens.
- Do not allow the IVF flow to run dry with the Morgan lens in place. Use a Y-connector to ensure that a second liter bag is ready to start as soon as the first runs dry.
- Briefly pause the flow before the end of each bag so that you can use a pH strip to measure the pH at the medial canthus. Your goal pH is 7.0.
- If the pH is 7.0, restart the IV fluids and then remove the Morgan lens. Do not attempt to remove the Morgan lens without fluid running through it, or you risk further traumatizing the eye due to a suction cup effect.
- For strong acids or strong bases, 2 hours of irrigation is recommended to ensure anterior chamber neutralization, even if surface pH reaches 7.0.4
- Children as young as six months can use the one-size-fits-all Morgan lens, and are known to tolerate them. 5
- Alternately, you can grab a nasal cannula and use the plastic sump connector from an NG tube to connect it to a bag of IV fluids. The prongs from the nasal cannula should be placed across the bridge of the nose so that fluid runs out of them into the eyes. 6
- Patients may find this more tolerable than the Morgan lens, and it avoids the risk of inducing further trauma to the surface of the eye from direct contact with the Morgan lens if the bag of IVF runs dry.
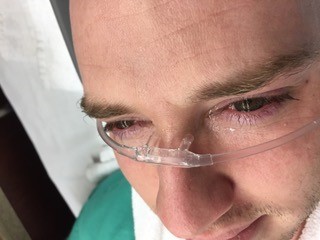
Image used with permission. Credit: Kathryn Kopec, DO, Emergency Medicine Attending and Medical Toxicologist, Atrium Health’s Carolinas Medical Center.
- Keep your patient dry and comfortable by placing their head in a basin with a Yankauer suction laying in the bottom to drain the irrigation fluid. 7
- When irrigation is complete, check IOP – alkali ocular injuries can result in secondary glaucoma due to ciliary body injury.
- Get ophthalmology and Poison Control on board early.
- Ophthalmology may recommend antibiotic coverage with ophthalmic erythromycin, inflammation control with ophthalmic prednisolone acetate, pain control through cycloplegia with ophthalmic cyclopentolate, and preservative-free artificial tears. 8
- Poison Control (1-800-222-1222) can identify if there are any nuances to a treatment based on the specific exposure.
Grading and Prognosis
- Do a thorough ophthalmologic exam. In addition to determining visual acuity, IOP, and pH, carefully evaluate the limbus, cornea and iris.
- The limbus is the area immediately surrounding the outer edges of the iris, containing all of the stem cells responsible for regenerating corneal epithelium. Ischemia of this area looks white, while the surrounding tissue is likely red and inflamed.
- If the limbus is ischemic and the stem cells die, the bulbar conjunctiva will grow over the cornea in an attempt to heal the injured area. Bulbar conjunctiva is opaque and rich in vasculature, which will severely limit the patient’s vision.
- You can use the Roper Hall classification scheme to prognosticate based on degree of corneal involvement as well as limbal ischemia. 9 However, limbal ischemia is highly subjective, with wide inter-rater variation among even cornea specialists. 10
- Grade I: No limbal ischemia, but some corneal epithelial damage is present on fluorescein exam. Good prognosis.
- Grade II: Limbal ischemia is limited to 1/3 the circumference of the iris. Corneal haze may be visible to the naked eye, but iris details should be discernible. Prognosis is still good.
- Grade III: Limbal ischemia from 1/3 to 1/2 of the circumference of the iris. The cornea has obvious total epithelial loss with haze that obscures iris details. Prognosis is guarded. Corneal epithelium likely will not regenerate – Rather, opacified conjunctiva will grow over this area, decreasing visual acuity.
- Grade IV: Greater than half of the limbus is ischemic. The cornea may be opaque with obscuring of the iris and pupil, or melting may be present. This carries a poor prognosis even with prompt medical intervention and vision loss is nearly guaranteed.
- A new classification scheme has been proposed, rating severity of injury from 0 to 3 in 3 categories: Degree of opacification, degree of vascularization, and degree of symblepharon (growth of scar tissue that extends from the palpebral conjunctiva to the bulbar conjunctiva, tethering the two to each other). These features are not present on initial evaluation but may begin to form as early as one week following the injury. 11
- Generally speaking, with household bleach and prompt attention, corneal injuries are generally mild, superficial, and the corneal epithelium recovers within 2 days. 12 Higher concentrations of bleach found in industrial settings penetrate easily through the cornea and cause severe injury by denaturing the proteins of the eye. 13
What about the potential for ingestion or inhalation injury in this patient?
- Alkali ingestions can be serious, with larger ingestions leading to liquefactive necrosis of the esophagus, stomach, and even duodenum. 14
- Acid ingestions classically cause coagulation necrosis. 15
- Getting a good airway exam, even if it means breaking out the flexible laryngoscope, is crucial.
- All patients with intentional ingestions of caustic agents will need an EGD between 4 and 24 hours after ingestion.
- In patients with unintentional ingestions, watch out for persistent drooling and vomiting, refusal to take anything by mouth, or respiratory distress, as those patients will need a scope as well. 16
- Corticosteroids and empiric antibiotics have not been shown to change outcomes. 17
Our patient likely did not ingest or inhale a significant amount during his brief exposure. If he continues to have a normal lung exam and has no significant stridor, coughing, or persistent drooling and vomiting, and if he can tolerate PO, he can be discharged home with ophthalmology follow-up.
Main points:
- Alkali-based injuries, such as those caused by bleach exposure, cause liquefactive necrosis.
- Acid-based injuries cause coagulation necrosis.
- Tissues of the eye are particularly sensitive and need thorough irrigation either with nasal cannula tubing or with the Morgan lens, with good ophthalmology follow-up.
- Tetracaine is safe and will improve the tolerability of the irrigation.
- Evaluate for injuries to the cornea, iris, and limbus, and check IOP to ensure that damage to the ciliary bodies has not caused secondary glaucoma.
- Always think “Worst First”, but when it comes to household bleach, with brief exposures, and with prompt treatment, patients tend to do well. 18
Bonus fun skunk fact:
Skunk spray is alkaline, which is why home remedies are acidic. However, the idea that neutralizing the pH will fix the smell is a myth, and vinegar or tomato juice are likely not as effective as you think. The components of the spray that smell so bad are sulfur-containing thiols, which can be broken down to the non-malodorous sulfonic acid by oxidation using a mixture of hydrogen peroxide, baking soda, and liquid detergent — Not that you’d want to pour this into your eyes, either. 19
References:
- Richards, A. Beware traumatic conjunctivitis: airbags can cause severe alkali eye injuries. Clin. Experiment. Ophthalmol. 44, 732–734 (2016).
- Ernst, A. A., Thomson, T., Haynes, M. & Weiss, S. J. Warmed versus room temperature saline solution for ocular irrigation: a randomized clinical trial. Ann. Emerg. Med. 32, 676–679 (1998).
- Jones, J. B., Schoenleber, D. B. & Gillen, J. P. The tolerability of lactated Ringer’s solution and BSS plus for ocular irrigation with and without the Morgan therapeutic lens. Acad. Emerg. Med. 5, 1150–1156 (1998).
- MorTan, Inc. The Morgan Lens – Ocular Chemical Burns. (2018).
- Pediatric Use of the Morgan Lens | The Morgan Lens | MorTan Inc. The Morgan Lens | MorTan Inc. https://www.morganlens.com/resource-library/pediatric-use-of-the-morgan-lens.
- Hoffman, L. Solutions for Difficult Problems:Eye Irrigation — Morgan Lens No More! Part 2. Emergency Medicine News (2016).
- Lin, M. Trick of the trade: Eye irrigation setup. ALiEM https://www.aliem.com/2010/04/trick-of-trade-eye-irrigation-setup/ (2010).
- Treating Acute Chemical Injuries of the Cornea. American Academy of Ophthalmology https://www.aao.org/eyenet/article/treating-acute-chemical-injuries-of-cornea (2012).
- Roper-Hall, M. J. Thermal and chemical burns. Trans. Ophthalmol. Soc. U. K. 85, 631–653 (1965).
- Kam, K. W., Patel, C. N., Nikpoor, N., Yu, M. & Basu, S. Limbal ischemia: Reliability of clinical assessment and implications in the management of ocular burns. Indian J. Ophthalmol. 67, 32–36 (2019).
- Wang, F. et al. Correlation analysis of the clinical features and prognosis of acute ocular burns-exploration of a new classification scheme. Graefes Arch. Clin. Exp. Ophthalmol. (2019) doi:10.1007/s00417-019-04525-6.
- Slaughter, R. J., Watts, M., Vale, J. A., Grieve, J. R. & Schep, L. J. The clinical toxicology of sodium hypochlorite. Clin. Toxicol. 57, 303–311 (2019).
- Benzoni, T. & Hatcher, J. D. Bleach Toxicity. in StatPearls (StatPearls Publishing, 2019).
- ATSDR – Medical Management Guidelines (MMGs): Carbon Disulfide. https://www.atsdr.cdc.gov/MMG/MMG.asp?id=927&tid=84.
- Chibishev, A., Pereska, Z., Chibisheva, V. & Simonovska, N. Corrosive poisonings in adults. Mater Sociomed 24, 125–130 (2012).
- Christesen, H. B. T. & Thybo Christesen, H. B. Prediction of complications following caustic ingestion in adults. Clinical Otolaryngology vol. 20 272–278 (1995).
- Harley, E. H. & Collins, M. D. Liquid household bleach ingestion in children: a retrospective review. Laryngoscope 107, 122–125 (1997).
- Toxicological Profile for Chlorine. (U.S. Department of Health and Human Services, Public Health Service, Agency for Toxic Substances and Disease Registry, 2010).
- The Chemistry of Skunk Spray — NOVA | PBS. https://www.pbs.org/wgbh/nova/nature/chemistry-skunk.html.






