Written by: Sean Hickey, MD (@seanhickey92 – Resident Physician, Icahn School of Medicine at Mount Sinai) and Stephen Alerhand, MD (@SAlerhand – Assistant Professor, Rutgers New Jersey Medical School) // Edited by: Manpreet Singh MD (@MPrizzleER – Assistant Professor of Emergency Medicine – Harbor-UCLA Medical Center) and Brit Long, MD (@long_brit)
This write up has been adapted from the above authors’ review in JEM: (PMID 31745658)
Pulmonary Embolism Classification
Half of pulmonary embolisms (PE’s) are diagnosed in the emergency setting [1, 2]. They can be categorized into three groups (Table 1) [3, 4]:
Table 1: Classification of Pulmonary Embolism [5] 
* Hemodynamic instability:
- Sustained hypotension with systolic blood pressure (SBP) < 90 mm Hg for at least 15 min or requiring inotropic support that is not due to another cause other than PE
- Pulselessness
- Persistent profound bradycardia (heart rate < 40 bpm) with signs or symptoms of shock
** Right ventricular (RV) strain:
- Bedside echocardiography
- Elevated laboratory markers (BNP > 500 pg/mL, Troponin I > 0.4 ng/mL or troponin T > 0.1 ng/mL)
- New ECG changes (incomplete or complete right bundle branch block, anteroseptal ST elevation or depression, anterolateral T wave inversion)
Background on Pulmonary Embolism Management
- In brief, evidence supports the use of thrombolytics for massive PE, as long as there are no contraindications [3, 6-9].
- Non-massive PE’s do not warrant thrombolytics, and some may even be safely treated at home [10, 11].
- It is for the sub-massive PE category that the evidence is less clear [4, 12-16]. This decision may depend on the patient’s clinical course and warrants discussion with the patient and consultants. A detailed synopsis can be found HERE.
Right Ventricular Structure and Dynamics in Pulmonary Embolism
- The RV has an unusual geometric shape which precludes the straight-forward assessment of volume and ejection fraction.
- It appears triangular-shaped when viewed from the side and crescent-shaped when viewed in cross-section [17, 18]. Its three parts are the inlet, highly trabeculated body, and smooth outlet portion (known as the conus or infundibulum) [19].
- The contraction of the RV is a complex process.
- It begins at the proximal portion and ends at the infundibulum and outflow tract. The inlet and outlet portions contract perpendicularly to each other.
- The RV carries superficial muscle fibers for circumferential contraction and deep subendocardial fibers for longitudinal contraction [20, 21], the latter of which contributes 80% of the cardiac output [10, 22].
- The RV and pulmonary arterial vasculature constitute a low-resistance, high-capacitance system.
- The RV is thus better able to handle increased preload than increased afterload [23].
- In the setting of a PE, there is an abrupt increase in pulmonary vascular resistance and RV afterload.
- The RV cannot overcome the increased pressure needed to overcome the large clot burden and associated mechanical obstruction.
- Even an obstruction of >25-30% of the pulmonary arteries is associated with an increase in pulmonary pressures [24] and 30% reduction in RV stroke volume [25]. Outright RV failure may be seen when 50-75% of the pulmonary vasculature is obstructed by thrombi [24]. This strain on the RV will first lead to chamber dilation and regional wall stress, followed by systolic dysfunction and septal deviation [26]. In this display of ventricular interdependence, the decrease in preload will lead to compromise of the cardiac output [27]. Hypotension, decreased coronary perfusion, and ischemia will follow.
- The RV cannot overcome the increased pressure needed to overcome the large clot burden and associated mechanical obstruction.
- European and American guidelines have stated that RV dysfunction and cardiac biomarker elevations are more relevant for risk stratification than the anatomic burden and distribution of pulmonary artery clots [5, 28].
- RV dysfunction can help prognosticate outcomes in normotensive patients with PE.
- In the short-term, this finding on bedside echocardiography can portend development of hypotension, cardiorespiratory deterioration, or death despite the initiation of anticoagulation.
- In the long-term, patients treated with standard anticoagulation and not thrombolytics may end up with persistent RV strain and functional limitation [29, 30]. It is these patients who ought to be considered for systemic or catheter-based thrombolytics, or at least more intensive clinical monitoring and disposition. In contrast, those without any signs of RV dysfunction may be considered for extended observation or even treated as an outpatient [11, 31].
Role of Echocardiography After the Diagnosis of Pulmonary Embolism
- The goal of echocardiography in cases of established PE is to identify high-risk patients (and thus direct care) before they may decompensate.
- Making matters difficult, the RV is difficult to evaluate using bedside echocardiography due to the complex geometric shape and contractile pattern described above.
- No single echocardiographic parameter (i.e. RV:LV ratio, D-shaped intraventricular septum, McConnell’s sign, tricuspid regurgitation) provides a clear-cut assessment of RV function.
TAPSE for Evaluation of Right Ventricular Dysfunction
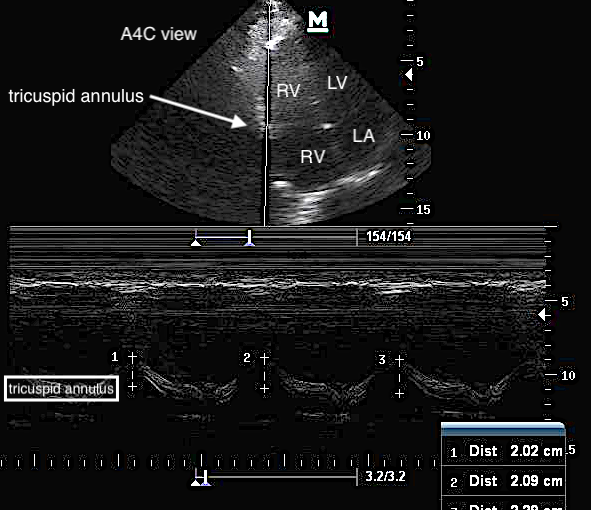
Tricuspid annular plane systolic excursion (TAPSE) has gained traction as a risk-stratification and prognostic tool through its assessment of global RV function and ejection fraction. In the apical 4-chamber view, this one-dimensional measure of RV systolic function is obtained by measuring the vertical movement of the tricuspid annulus between the end of diastole and end of systole in M-mode. This reflects the longitudinal contraction of the RV.
- Approximates RV ejection fraction with 80% sensitivity and 75% specificity [32].
- Used by cardiologists to reliably assess RV dysfunction and ejection fraction correlated with cardiac MRI and right heart angiography [32-34].
- The mid-esophageal 4-chamber view of transesophageal echocardiography (TEE) was validated as a good correlate to RV systolic function as reflected by RV fractional area change [35].
- Validated against the RV ejection fraction calculated from the biplanar Simpson’s rule [36].
- Can assess for RV dysfunction in patients with heart failure with preserved ejection fraction [37-39].
TAPSE as a Prognostic Tool in Patients with Pulmonary Embolism
- The numerical value of TAPSE is decreased in patients with acute PE [40, 41].
- Abnormal TAPSE is independently predictive of increased short-term mortality [42] and increased length-of-stay in the intensive care unit (ICU) [43, 44].
- In those who are normotensive, it reflects RV function and independently predicts survival [42], while demonstrating superiority for risk stratification compared to RV/LV ratio [45].
- TAPSE measurements can even be used to predict acute decompensation in patients undergoing acute pulmonary artery embolectomy [46].
- TAPSE may recover incompletely after 3 months [40], and is independently associated with a decreased long-term survival [47].
Why makes TAPSE so attractive as a measurement tool?
- Unlike the other qualitative RV parameters of RV dysfunction, TAPSE provides a quantitative value that corresponds to RV systolic function. This allows an easy determination of normal compared to abnormal.
- TAPSE can be reproduced by physicians with high interobserver reliability [48-50].
- It is less dependent on optimal image quality.
What are the cut-off measurements of TAPSE?
There is no consensus measurement or cut-off for TAPSE values in prognostication [42, 43, 45, 46, 51].

Limitations to TAPSE
- TAPSE measures the longitudinal excursion of the tricuspid annulus in one dimension. It does not incorporate the transverse contribution to ejection fraction of the RV free wall and septum. Therefore, TAPSE may be less accurate in patients with regional differences in RV function.
- The numerical value may be affected by the angle of the M-mode cursor over the tricuspid annulus.
- The precise cut-off value of numerical estimation has not been established.
- A decreased TAPSE is not specific to PE. It may also result from pulmonary hypertension and congestive heart failure [48].
How to Obtain TAPSE
Obtain an apical 4-chamber view of the heart.

Place the M-mode cursor over the lateral aspect of the tricuspid annulus. Measure the distance of the maximal longitudinal displacement. Calculate the average of three consecutive cardiac cycles.

Cases
Case 1
In a patient with diagnosed PE who has normal vitals, ECG, and biomarkers, you put the US probe on this patient and find that the TAPSE is 13 mm. He has a sub-massibve PE, so you decide to admit this patient to Stepdown instead of just Telemetry.
Case 2
In a patient with diagnosed PE who has normal vitals, ECG, and biomarkers, you put the US probe on this patient and find that the TAPSE is 14 mm. Given the association between abnormal TAPSE and worse prognosis, you discuss this finding with the patient and convince him that his condition requires admission and closer monitoring, rather than discharge on apixaban and close outpatient follow-up with his Cardiologist.
Bottom Line
TAPSE provides a quantitative value that serves as a surrogate for RV function. It is easy to obtain and reproducible. In patients diagnosed with PE, it can serve as a risk-stratification tool by guiding the decision of whether to administer thrombolytics, and also determining which type of hospital disposition the patient’s condition warrants.
References
- Jones, A.E. and J.A. Kline, Availability of technology to evaluate for pulmonary embolism in academic emergency departments in the United States.J Thromb Haemost, 2003. 1(10): p. 2240-2.
- Pollack, C.V., et al., Clinical characteristics, management, and outcomes of patients diagnosed with acute pulmonary embolism in the emergency department: initial report of EMPEROR (Multicenter Emergency Medicine Pulmonary Embolism in the Real World Registry).J Am Coll Cardiol, 2011. 57(6): p. 700-6.
- Konstantinides, S.V., 2014 ESC Guidelines on the diagnosis and management of acute pulmonary embolism.Eur Heart J, 2014. 35(45): p. 3145-6.
- Jaff, M.R., et al., Management of massive and submassive pulmonary embolism, iliofemoral deep vein thrombosis, and chronic thromboembolic pulmonary hypertension: a scientific statement from the American Heart Association.Circulation, 2011. 123(16): p. 1788-830.
- Torbicki, A., et al., Guidelines on the diagnosis and management of acute pulmonary embolism: the Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology (ESC).Eur Heart J, 2008. 29(18): p. 2276-315.
- Wadhera, R.K. and G. Piazza, Treatment Options in Massive and Submassive Pulmonary Embolism.Cardiol Rev, 2016. 24(1): p. 19-25.
- Wan, S., et al., Thrombolysis compared with heparin for the initial treatment of pulmonary embolism: a meta-analysis of the randomized controlled trials.Circulation, 2004. 110(6): p. 744-9.
- Sharifi, M., et al., Pulseless electrical activity in pulmonary embolism treated with thrombolysis (from the “PEAPETT” study).Am J Emerg Med, 2016. 34(10): p. 1963-1967.
- Kearon, C., et al., Antithrombotic therapy for VTE disease: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines.Chest, 2012. 141(2 Suppl): p. e419S-e496S.
- Ho, S.Y. and P. Nihoyannopoulos, Anatomy, echocardiography, and normal right ventricular dimensions.Heart, 2006. 92 Suppl 1: p. i2-13.
- Aujesky, D., et al., Outpatient versus inpatient treatment for patients with acute pulmonary embolism: an international, open-label, randomised, non-inferiority trial.Lancet, 2011. 378(9785): p. 41-8.
- Goldhaber, S.Z., L. Visani, and M. De Rosa, Acute pulmonary embolism: clinical outcomes in the International Cooperative Pulmonary Embolism Registry (ICOPER).Lancet, 1999. 353(9162): p. 1386-9.
- Meyer, G., et al., Fibrinolysis for patients with intermediate-risk pulmonary embolism.N Engl J Med, 2014. 370(15): p. 1402-11.
- Chatterjee, S., et al., Thrombolysis for pulmonary embolism and risk of all-cause mortality, major bleeding, and intracranial hemorrhage: a meta-analysis.JAMA, 2014. 311(23): p. 2414-21.
- Konstantinides, S., et al., Heparin plus alteplase compared with heparin alone in patients with submassive pulmonary embolism.N Engl J Med, 2002. 347(15): p. 1143-50.
- Konstantinides, S.V., et al., Impact of Thrombolytic Therapy on the Long-Term Outcome of Intermediate-Risk Pulmonary Embolism.J Am Coll Cardiol, 2017. 69(12): p. 1536-1544.
- James, T.N., Anatomy of the crista supraventricularis: its importance for understanding right ventricular function, right ventricular infarction and related conditions.J Am Coll Cardiol, 1985. 6(5): p. 1083-95.
- Haddad, F., et al., Right ventricular function in cardiovascular disease, part I: Anatomy, physiology, aging, and functional assessment of the right ventricle.Circulation, 2008. 117(11): p. 1436-48.
- Lee, J.Z., et al., Comparison of tricuspid annular plane systolic excursion with fractional area change for the evaluation of right ventricular systolic function: a meta-analysis.Open Heart, 2018. 5(1): p. e000667.
- Dahhan, T., et al., Echocardiography in the Risk Assessment of Acute Pulmonary Embolism.Semin Respir Crit Care Med, 2017. 38(1): p. 18-28.
- Bystrom, B., P. Lindqvist, and M. Henein, The right ventricle: knowing what is right.Int J Cardiovasc Imaging, 2008. 24(7): p. 701-2.
- Carlsson, M., et al., The quantitative relationship between longitudinal and radial function in left, right, and total heart pumping in humans.Am J Physiol Heart Circ Physiol, 2007. 293(1): p. H636-44.
- Haddad, F., et al., Right ventricular function in cardiovascular disease, part II: pathophysiology, clinical importance, and management of right ventricular failure.Circulation, 2008. 117(13): p. 1717-31.
- McIntyre, K.M. and A.A. Sasahara, The hemodynamic response to pulmonary embolism in patients without prior cardiopulmonary disease.Am J Cardiol, 1971. 28(3): p. 288-94.
- Guyton, A.C., A.W. Lindsey, and J.J. Gilluly, The limits of right ventricular compensation following acute increase in pulmonary circulatory resistance.Circ Res, 1954. 2(4): p. 326-32.
- Weekes, A.J., et al., Diagnostic Accuracy of Right Ventricular Dysfunction Markers in Normotensive Emergency Department Patients With Acute Pulmonary Embolism.Ann Emerg Med, 2016. 68(3): p. 277-91.
- Wilcox, S.R., C. Kabrhel, and R.N. Channick, Pulmonary Hypertension and Right Ventricular Failure in Emergency Medicine.Ann Emerg Med, 2015. 66(6): p. 619-28.
- Kearon, C., et al., Antithrombotic therapy for venous thromboembolic disease: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition).Chest, 2008. 133(6 Suppl): p. 454S-545S.
- Kline, J.A., et al., Prospective evaluation of right ventricular function and functional status 6 months after acute submassive pulmonary embolism: frequency of persistent or subsequent elevation in estimated pulmonary artery pressure.Chest, 2009. 136(5): p. 1202-1210.
- Kasper, W., et al., Prognostic significance of right ventricular afterload stress detected by echocardiography in patients with clinically suspected pulmonary embolism.Heart, 1997. 77(4): p. 346-9.
- Zondag, W., et al., Comparison of two methods for selection of out of hospital treatment in patients with acute pulmonary embolism.Thromb Haemost, 2013. 109(1): p. 47-52.
- Ueti, O.M., et al., Assessment of right ventricular function with Doppler echocardiographic indices derived from tricuspid annular motion: comparison with radionuclide angiography.Heart, 2002. 88(3): p. 244-8.
- Park, J.R., et al., Evaluation of right ventricular dysfunction and prediction of clinical outcomes in acute pulmonary embolism by chest computed tomography: comparisons with echocardiography.Int J Cardiovasc Imaging, 2012. 28(4): p. 979-87.
- Kjaergaard, J., et al., Evaluation of right ventricular volume and function by 2D and 3D echocardiography compared to MRI.Eur J Echocardiogr, 2006. 7(6): p. 430-8.
- Morita, Y., K. Nomoto, and G.W. Fischer, Modified Tricuspid Annular Plane Systolic Excursion Using Transesophageal Echocardiography for Assessment of Right Ventricular Function.J Cardiothorac Vasc Anesth, 2016. 30(1): p. 122-6.
- Miller, D., et al., The relation between quantitative right ventricular ejection fraction and indices of tricuspid annular motion and myocardial performance.J Am Soc Echocardiogr, 2004. 17(5): p. 443-7.
- Puwanant, S., et al., Right ventricular function in patients with preserved and reduced ejection fraction heart failure.Eur J Echocardiogr, 2009. 10(6): p. 733-7.
- Morris, D.A., et al., Right ventricular myocardial systolic and diastolic dysfunction in heart failure with normal left ventricular ejection fraction.J Am Soc Echocardiogr, 2011. 24(8): p. 886-97.
- Burke, M.A., et al., Prognostic importance of pathophysiologic markers in patients with heart failure and preserved ejection fraction.Circ Heart Fail, 2014. 7(2): p. 288-99.
- Rydman, R., et al., Echocardiographic evaluation of right ventricular function in patients with acute pulmonary embolism: a study using tricuspid annular motion.Echocardiography, 2010. 27(3): p. 286-93.
- Vitarelli, A., et al., Right ventricular function in acute pulmonary embolism: a combined assessment by three-dimensional and speckle-tracking echocardiography.J Am Soc Echocardiogr, 2014. 27(3): p. 329-38.
- Lobo, J.L., et al., Prognostic significance of tricuspid annular displacement in normotensive patients with acute symptomatic pulmonary embolism.J Thromb Haemost, 2014. 12(7): p. 1020-7.
- Pruszczyk, P., et al., Prognostic value of echocardiography in normotensive patients with acute pulmonary embolism.JACC Cardiovasc Imaging, 2014. 7(6): p. 553-60.
- Zanobetti, M., et al., Prognostic value of emergency physician performed echocardiography in patients with acute pulmonary thromboembolism.West J Emerg Med, 2013. 14(5): p. 509-17.
- Paczynska, M., et al., Tricuspid annulus plane systolic excursion (TAPSE) has superior predictive value compared to right ventricular to left ventricular ratio in normotensive patients with acute pulmonary embolism.Arch Med Sci, 2016. 12(5): p. 1008-1014.
- Schmid, E., et al., Tricuspid annular plane systolic excursion (TAPSE) predicts poor outcome in patients undergoing acute pulmonary embolectomy.Heart Lung Vessel, 2015. 7(2): p. 151-158.
- Khemasuwan, D., et al., Right ventricular echocardiographic parameters are associated with mortality after acute pulmonary embolism.J Am Soc Echocardiogr, 2015. 28(3): p. 355-62.
- Daley, J., et al., Emergency physician performed tricuspid annular plane systolic excursion in the evaluation of suspected pulmonary embolism.Am J Emerg Med, 2017. 35(1): p. 106-111.
- Kopecna, D., et al., Interobserver reliability of echocardiography for prognostication of normotensive patients with pulmonary embolism.Cardiovasc Ultrasound, 2014. 12: p. 29.
- Hammarstrom, E., et al., Tricuspid annular motion.J Am Soc Echocardiogr, 1991. 4(2): p. 131-9.
- Ciurzynski, M., et al., Tricuspid Regurgitation Peak Gradient (TRPG)/Tricuspid Annulus Plane Systolic Excursion (TAPSE)- A Novel Parameter for Stepwise Echocardiographic Risk Stratification in Normotensive Patients With Acute Pulmonary Embolism.Circ J, 2018. 82(4): p. 1179-1185.





5 thoughts on “US Probe: TAPSE”
Pingback: Nursing FOAMed Review #2 (Aug 9th - Aug 20th) - Code Blue LLC
Pingback: LITFL Review 344 | Edwin M. Thames
Pingback: LITFL Review 344 • LITFL Medical Blog • FOAM Review
Pingback: TAPSE za procjenu desnog ventrikula - Prijenosni ultrazvuk
Pingback: What’s the Diagnosis? By Dr. Sean Coulson -