Welcome back to the “52 in 52” series. This collection of posts features recently published must-know articles. Post #21 looks at targeted temperature management after cardiac arrest with non-shockable rhythm, or the Hyperion trial.

Author: Brannon Inman (Chief Resident, Emergency Medicine Physician, San Antonio, TX) // Reviewed by: Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit)
Targeted Temperature Management for Cardiac Arrest with Non-shockable Rhythm
AKA: The “Hyperion” Trial
Question:
Does moderate hypothermia improve neurologic outcomes in cardiac arrest patients with non-shockable rhythm?
Design: A multicenter open-label randomized controlled trial
PICO
Population:
- Enrolled patients in 25 ICUs (11 academic, 14 community)
- Inclusion criteria
- Age ≥ 18
- Enrolled in and out of hospital cardiac arrest patients resuscitated from non-shockable cardiac arrest with any cause
- GCS ≤ 8
- Exclusion criteria
- A no-flow time (from collapse to initiation of cardiopulmonary resuscitation [CPR]) of more than 10 minutes
- A low-flow time (from initiation of CPR to return of spontaneous circulation) of more than 60 minutes
- Major hemodynamic instability
- Continuous epinephrine or norepinephrine infusion >1 μg per kilogram of body weight per minute
- Time from cardiac arrest to screening of more than 300 minutes
- Moribund (terminal/ near fatal) condition
- Child-Pugh class C cirrhosis of the liver (severe hepatic dysfunction)
- Pregnancy or breast-feeding
- Status of being under guardianship
- Status of being an inmate at a correctional facility
- Previous inclusion in another randomized, controlled trial involving patients with cardiac arrest in which the neurologic outcome at 90 days was assessed as the primary endpoint
- Lack of health insurance
- A decision by the next of kin for the patient not to participate
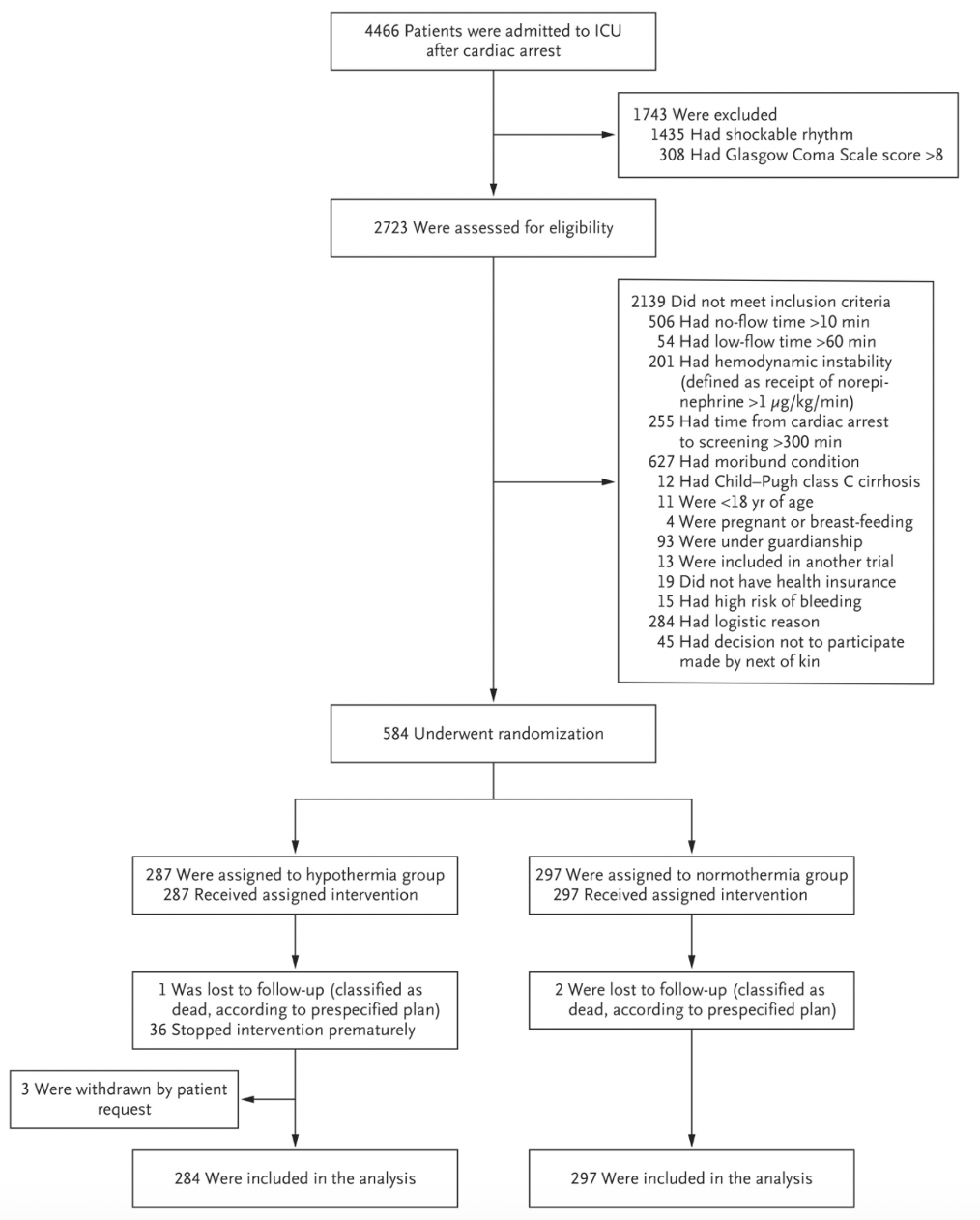
Randomization:
- Normothermia group with slightly more arrest at home, asystole as initial rhythm, and post-ROSC shock state
- Slightly longer median time to randomization in the hypothermia group
- Roughly the same temperature at randomization between the groups

Intervention:
- 24 hours of hypothermia at 33ºC following each ICU’s own standardized protocol
- Treatment began a median of 16 minutes post-randomization
- Treatment was stopped prematurely in 12.7% of patients
- Patients were warmed at a rate of 0.25-0.5ºC/hour

Comparator:
- 48 hours of normothermia 36.5-37.5 ºC following each ICU’s own standardized protocol
- Over 5 % of patients were unable to be maintained at 36.5-37.5 ºC, and had documented fever (≥38ºC)
Outcome:
- Primary outcome survival with a good neurologic outcome (CPC score of 1 or 2 at 90 days)
- 2% of hypothermia patients
- 7% of normothermia patients
- Fragility index of 1
- A fragility index of 1 indicates that if 1 patient in the control group were “converted” from NOT having the primary endpoint to HAVING the primary endpoint, the study would lose statistical significance (p > 0.05). The higher the fragility index, the more robust the results of a study.


Take Aways:
- Positive study
- There is limited external generalizability due to one country being included and the practice patterns of the aforementioned country (stopping sedation in the intervention group in patients with suspected brain death during the trial period).
- There was a slightly longer time to randomization in the hypothermia group. If therapeutic cooling works, it probably is time sensitive. This could skew the data toward the null.
- Quite a few patients in the normothermia group actually had a fever. It’s not explicitly stated in the text whether these patients were included in the analysis or not. This may have biased the data/outcomes.
- The failure to maintain normothermia in the control group questions the intensity of nursing and support staff care provided in the control group vs. the intervention group (therapeutic cooling is known to become labor intensive, with more nursing care etc.). Hyperthermia is what we try to avoid post arrest.
- There are a lot of differences between the two groups:
- Longer duration of therapeutic cool in the intervention group (up to 64 hours) vs the control (48 hours)
- The intervention group sedated to a RASS of -5, vs RASS of 0 in the control
- Longer time to randomization in the intervention group
My Take:
- This is extremely fragile data supporting the use of therapeutic cooling patients with non-shockable rhythms with coma following ROSC.
- In light of other data suggesting no benefit, these fragile data have not meaningfully convinced me of the benefit of therapeutic cooling. This study has limited applications due to the reasons mentioned above, and issues with randomization.
References:
- Bagci Z, Arslan A, Neselioglu S. Pediatric Carbon Monoxide Poisoning: Effects of Hyperbaric Oxygen Therapy on Thiol/Disulfide Balance. Pediatr Emerg Care. 2022;38(3):104-107. Doi: 10.1097/PEC.0000000000002619






