Authors: Tim Montrief, MD (@EMinMiami) and Nathan Boin, MD (University of Miami/Jackson Memorial Hospital Emergency Medicine Residency Program) // Edited by Alex Koyfman, MD (@EMHighAK) and Brit Long, MD (@long_brit)
Case: A 42-year-old woman with a history of obesity and alcohol abuse presents two days after an outpatient ERCP for choledocholithiasis. She complains of persistent vomiting which started the morning after her procedure, along with diffuse abdominal pain. Her heart rate is 104 beats per minute, but all other vital signs are normal. Her abdominal exam is notable for diffuse moderate tenderness with mild guarding but no evidence of peritoneal signs. What is on your differential for this patient? What are your next steps?
Background
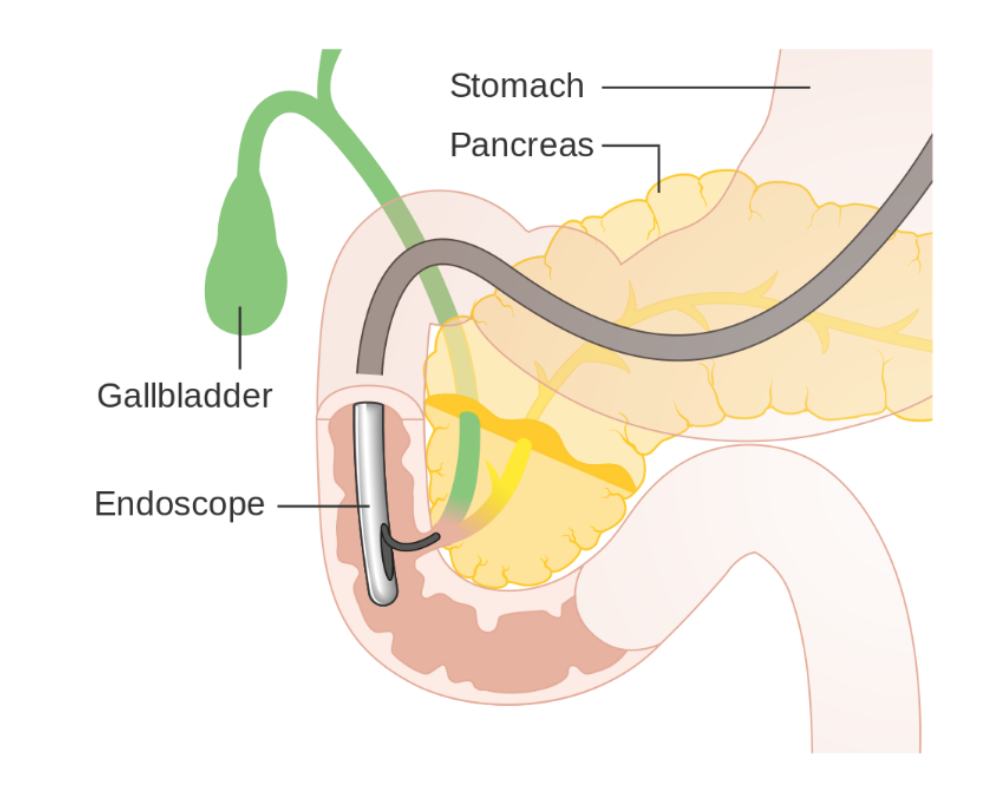
Over 700,000 endoscopic retrograde cholangiopancreatographies (ERCP) are performed annually in the U.S.1,2 Common indications include choledocholithiasis, biliary stone removal, biliary stenting, the diagnosis and management of biliary and pancreatic neoplasms, and management of sphincter dysfunction.3,4 ERCP is performed by a gastroenterologist who passes an endoscope through the oropharynx, esophagus, and stomach into the duodenum, insufflating the gastrointestinal tract. After visualizing the common opening to the ducts from the biliary tree and pancreas (the major duodenal papilla), the gastroenterologist passes a catheter through the endoscope and into the ducts (Figure 1). Contrast may be injected into the pancreatic and/or biliary ducts, or instrumentation (sphincterotomy, balloon dilation, electrocautery, etc.) may be performed.
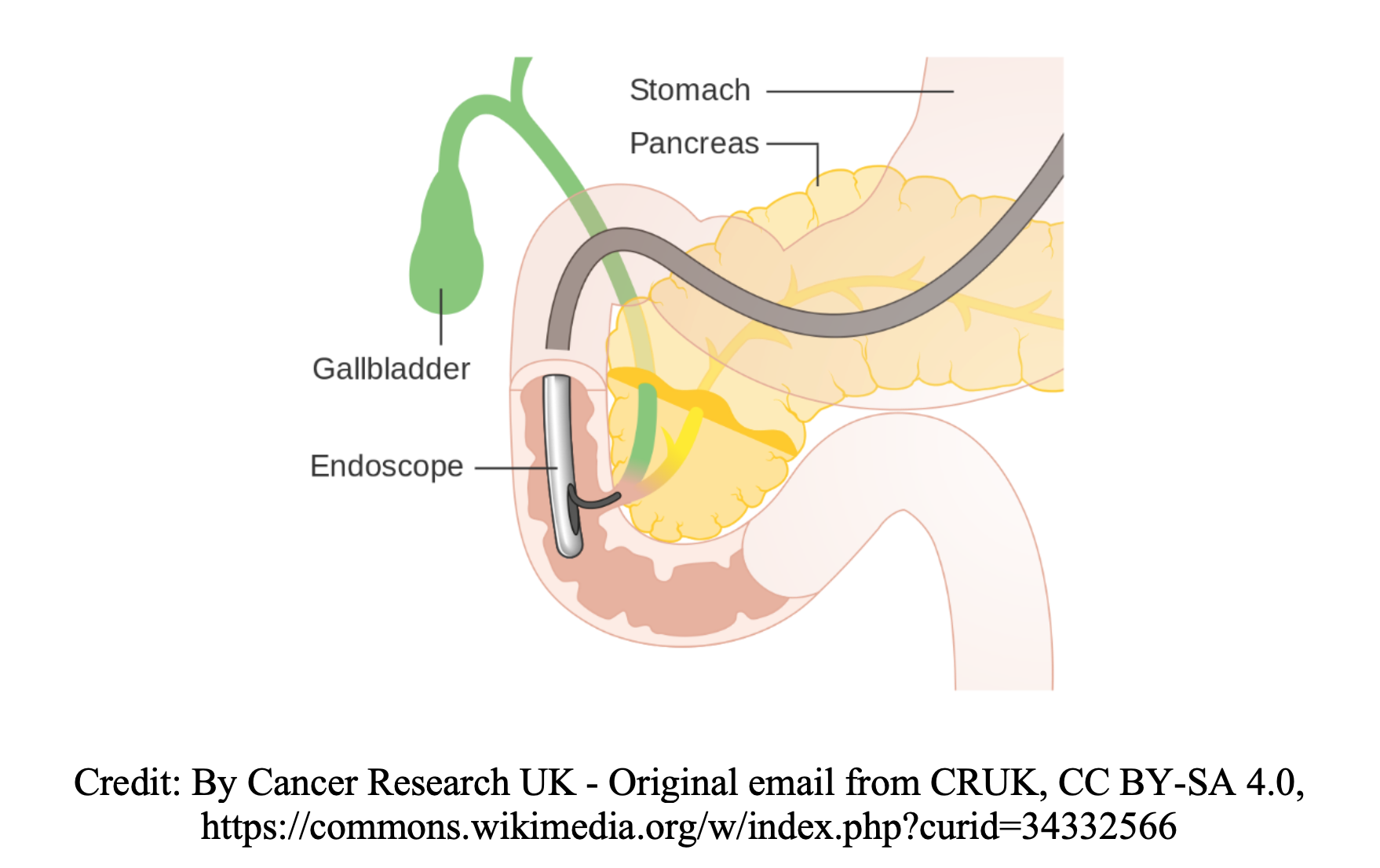
ERCP has recently increasingly become an outpatient procedure.5,6 Post-ERCP complication rates may reach 9.7%, with a mortality rate of 0.7%.7-9 Acute pancreatitis, gastrointestinal bleeding, viscous perforation, and biliary infections remain the most common complications, followed by aspiration, air gas embolism, pneumomediastium, formation of liver abscesses, and gallstone ileus. This post will focus on the four most common complications:
- Post-ERCP pancreatitis (PEP)
- Viscous perforation
- Gastrointestinal bleeding
- Biliary infections (acute cholangitis and cholecystitis)
Risk factors for post-ERCP complications are provided in Table 1 below.

Post-ERCP Pancreatitis
Acute pancreatitis is the most common complication of ERCP, with an estimated incidence between 3.5-9.7% and a mortality rate of 0.7%.7,9In high risk populations, the incidence can reach 67%.10,11The pathophysiology of PEP is poorly understood, but local inflammation plays a key role. It is also thought that mechanical obstruction of the pancreatic sphincter, biliary tree edema, hydrostatic injection, and pancreatic irritation from contrast causes sphincter of Oddi dysfunction.12
The clinical manifestations of PEP are the same as those seen in patients with acute pancreatitis due to other causes. Patients will present with sudden-onset, persistent epigastric pain, usually associated with vomiting (up to 80% of cases).13 The pain might radiate to the back, most commonly the lower thoracic spine.14 The abdominal exam will often reveal epigastric tenderness with guarding, and in cases of severe pancreatitis which has progressed to hemorrhage, Grey Turners sign (flank ecchymosis) or Cullen’s sign (periumbilical ecchymosis) may be found.14 It’s important to note that differentiating between PEP and post-ERCP viscous perforation is challenging, as there are no reliable clinical signs or symptoms distinguishing the two.14
Lab testing in the initial evaluation of PEP should be guided by the clinical presentation, but may include a complete blood count, electrolyte panel, serum lipase and amylase levels, and liver function studies.14,15 While amylase is rarely utilized in the ED, most studies evaluating ERCP patients evaluate amylase and lipase. A transient increase in serum pancreatic enzymes is found in up to 75% of patients, regardless of symptoms.10 This increase does not necessarily constitute pancreatitis.16 But, serum pancreatic enzyme may be used to predict patients who are unlikely to develop PEP.17 In one study involving 231 patients following ERCP, values of amylase and lipase below 276 unit/L and 1000 unit/L, respectively, at two hours after ERCP were highly predictive of the absence of pancreatitis, even in patients experiencing abdominal pain (negative predictive values of 97% and 98%, respectively).18 Amylase and lipase start to rise a few hours after the onset of pancreatitis, so blood tests drawn immediately after ERCP may be falsely-reassuring.If the clinical suspicion PEP remains high, repeat tests four hours after ERCP should be obtained.
Imaging is not necessary to diagnose PEP.15,19 However, if the diagnosis of PEP remains in doubt, contrast-enhanced computed tomography (CT) of the abdomen can confirm the diagnosis while also excluding other etiologies for the presentation.15,20 A normal CT does not exclude mild pancreatitis and is falsely-negative in up to 27% of the time.15,21,22 While right upper quadrant (RUQ) ultrasound is currently recommended in all patients with pancreatitis, it has not been evaluated in PEP specifically.23 RUQ ultrasound may show evidence of cholecystitis, common bile duct dilation, or, in the case of intra-abdominal hemorrhage, free fluid in the abdomen.24 Direct pancreatic ultrasound has limited diagnostic or prognostic value in PEP, as the study is frequently inadequate and, given the availability of advanced imaging modalities, it does not add additional information.21,22
Management of PEP is similar to other causes of pancreatitis. The initial management of a patient with PEP centers on supportive care, fluid resuscitation, pain control, and early nutritional support.15 Acute pancreatitis typically causes intravascular volume loss form release of vascular mediators and increased vascular permeability with nausea and vomiting.24 While inadequate fluid resuscitation has been associated with increased rates of pancreatic necrosis, large volume fluid resuscitation may not be necessary for these patients.24,25 This has been recognized by the American Gastroenterological Association Institute’s 2018 guidelines which recommend “judicious goal-directed fluid therapy versus other methods,” as goal-directed fluid resuscitation does not improve outcomes.24,26 Current recommendations from the American Gastroenterological Association also include transfusion of packed red blood cells when the hematocrit falls below 25% and albumin if the serum albumin level drops below 2 g/dL.27
As pain is often exacerbated with oral intake early in the disease process, patients should be kept NPO during the acute phase.24 Patients with mild disease may be started on oral acetaminophen or immediate-release morphine in the ED. For patients requiring parenteral analgesia, a multi-modal approach is ideal. Potential agents include intravenous acetaminophen, subdissociative dose ketamine, fentanyl, morphine, and hydromorphone.24 Additionally, regional anesthesia may provide opioid-sparing analgesia in these patients. The transversus abdominis plane block may be effective and is feasible for emergency physicians to perform.28 Routine administration of antibiotics in patients with PEP is not currently recommended.29
Gastrointestinal Bleeding
Bleeding is a serious complication of ERCP and is most commonly associated with endoscopic biliary and/or pancreatic sphincterotomy. The incidence of postsphincterotomy bleeding is estimated to be 0.3% to 2%.16,30,31 Gastrointestinal bleeding is associated with a mortality rate of 3.54%.7 Most ERCP-associated bleeding is intraluminal, although intraductal bleeding, intra-abdominal bleeding, and hematomas (hepatic, splenic, and intra-abdominal) can occur.16,32-36 Post-ERCP bleeding is classified into either immediate (during or immediately after the procedure) or delayed (hours to weeks after the procedure).16 Approximately half of bleeding complications occur immediately following ERCP.37-41
Patients present with hematemesis, coffee-ground emesis, and/or melena. Hematochezia may occasionally be the initial presentation in a briskly bleeding patient. Other presentations which can accompany post-ERCP hemorrhage include abdominal pain, hemodynamic instability, and symptoms of anemia such as fatigue, syncope and angina.42 Laboratory investigations are similar as those for an upper GI bleed- CBC, CMP, Type and screen, and Coagulation panel. Options for initial evaluation of post-ERCP hemorrhage include point of care ultrasound (POCUS), upper endoscopy, CT angiogram, and interventional radiology-directed catheter angiography. While intra-abdominal hemorrhage following ERCP is rare, a FAST exam should be performed to evaluate for free fluid within the peritoneum. POCUS is highly sensitive and specific (>96% and >95% respectively).43 For most patients, the initial diagnostic evaluation of choice is an urgent upper endoscopy.44 CT angiogram or catheter angiography remains an option for patients with high-volume bleeding as it may be impossible to adequately visualize the source of hemorrhage with endoscopy.45
Management of post-ERCP GI bleeding is similar to that of any upper GI bleed. The majority of post-ERCP bleeding is self-limited and not clinically significant.46 Patients in hemorrhagic shock require emergent resuscitation, including large-bore intravenous access and typed and cross-matched blood, with the consideration of massive transfusion protocols.47 Patients should be aggressively resuscitated prior to intubation if needed, and induction agent doses should be reduced to minimize peri-procedural hypotension or arrest.48,49 Blood transfusion, reversal of coagulopathy, and medication administration (Ceftriaxone, PPI, Octreotide, etc) is similar to the treatment algorithm for other causes of GI bleeding.44 Early consultation with interventional radiology, general surgery, and gastroenterology is recommended for control of clinically significant bleeding. The majority of post-ERCP bleeding can be managed with medical treatment and emergent endoscopic therapy alone.37,50 In patients with refractory bleeding, both angiographic embolization and surgery are effective therapies.37,39,50,51 Angiographic embolization has been shown to control bleeding in 83% to 100% of patients and may be considered before surgery.52-54
Viscous Perforation
Viscous perforation during ERCP remains a rare entity, with an incidence between 0.08% and 0.6%.37,55-57 Prompt diagnosis and management are vital, as delayed therapy may lead to sepsis, with an 8-23% mortality. Perforations may occur anywhere within the gastrointestinal tract, including the thoracic esophagus, stomach, duodenum, pancreatic duct, and biliary duct. Perforation may occur secondary to several mechanisms:
- Luminal perforation by the endoscope, typically resulting in intraperitoneal perforation,
- Extramural passage of guidewires or migration of stents, or
- Extension of a sphincterotomy incision beyond the intramural portion of the bile or pancreatic duct with retroperitoneal leakage.16,58
ERCP-related perforations are classified according to location or mechanism of injury, which dictate appropriate management (Table 2).16,59

Clinical presentation depends on location of the perforation. While more than 70% of patients with esophageal perforation complain of chest pain, other symptoms and signs are variable and nonspecific in many cases.60 The pain associated with esophageal perforation is classically described as acute in onset, with radiation to the back or to the left shoulder. In approximately 25% of patients, chest pain is followed by shortness of breath and vomiting.60 This triad of shortness of breath, vomiting, and chest pain is known as the Mackler Triad.60 In those patients with cervical esophageal perforation, neck pain, dysphonia, hoarseness, dysphagia, and subcutaneous emphysema may be seen. Patients with perforation commonly present with severe epigastric or back pain and epigastric tenderness progressing to generalized abdominal wall rigidity with peritonitis.61 Subcutaneous emphysema, fever, and tachycardia may also be present. Signs of peritonitis develop 4-6 hours after the ERCP, when duodenal contents extravasate into the peritoneal cavity.
Once perforation is suspected, neck, abdominal, and chest radiographs should be obtained. Signs of thoracic esophageal perforation include pneumomediatinum, hydrothorax, pleural effusion, and subcutaneous emphysema.60 Pneumothorax is present in up to 77% of cases.62,63 If the patient is able to swallow, a chest radiograph with water soluble contrast medium is highly specific for the detection of perforation.64 However, the sensitivity remains poor (60-70%). A contrast-enhanced CT scan of the chest should be performed in the case of negative contrast radiograph despite high clinical suspicion or in order to rule out alternative diagnoses.65,66
POCUS is a bedside alternative to plain radiographs in appropriate patients.67 Both diagnostic modalities demonstrate high positive predictive value (95% in ultrasound compared to 94% in radiography) and similar specificity (53%).67,68 Ultrasound has been shown to have a higher sensitivity (92% vs. 78%), and negative predictive value (39% vs. 20%) compared to plain radiographs.67 The main sonographic sign indicating a perforated viscous is an increased echogenicity of a peritoneal stripe associated with multiple reflection artifacts and characteristic comet-tail appearance.67 This is best detected using a linear probe in the right upper quadrant.67 Thickened bowel loops, localized fluid collections containing air, thickened bowel or gallbladder walls, and decreased bowel motility may be indirect signs of a perforation.69,70
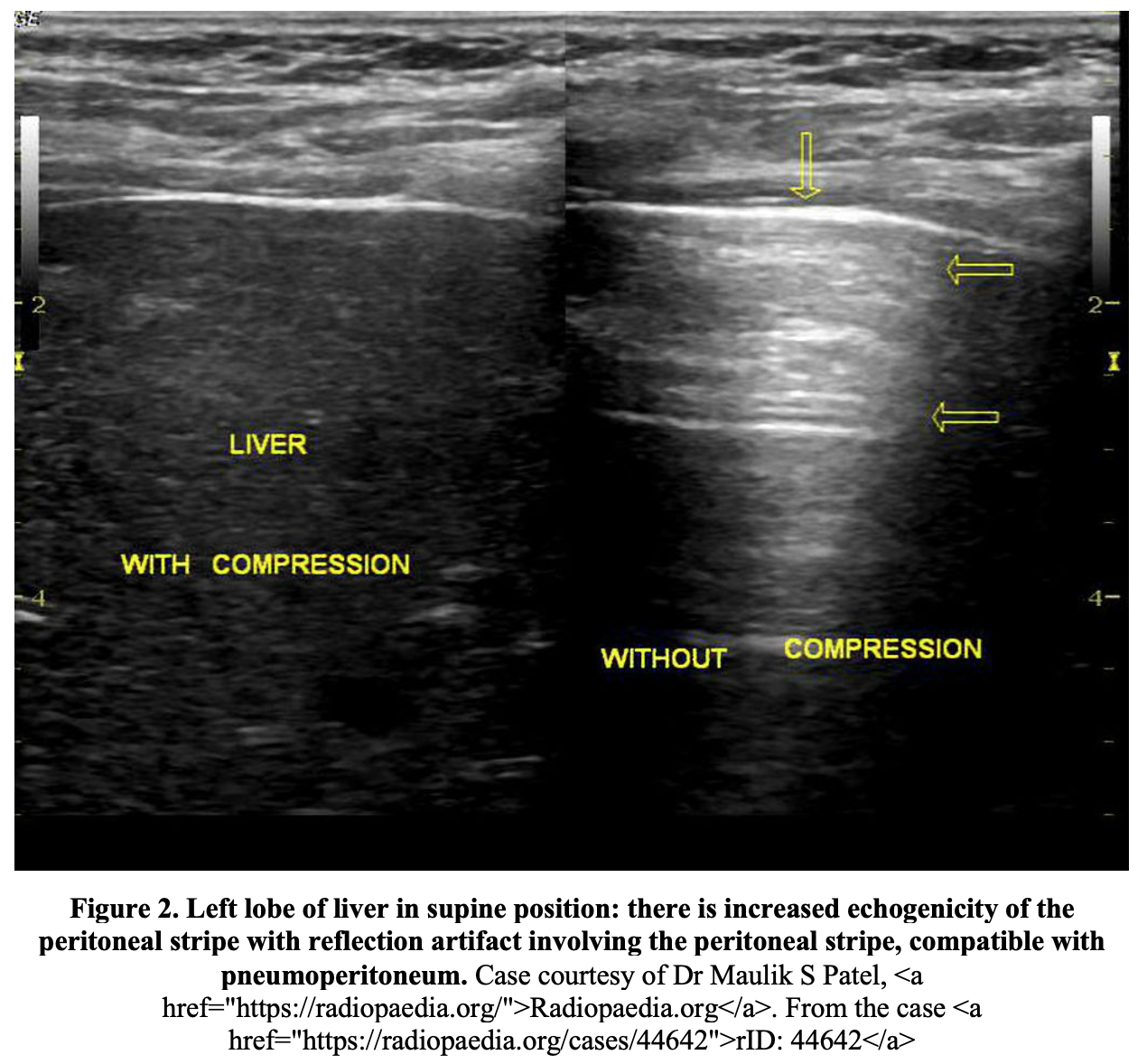
A CT scan is the definitive imaging choice.71,72 CT is the most sensitive test, as it is capable of assessing for both retroperitoneal and intraperitoneal air.71,73 The amount of air does not correlate with the severity of the perforation, but rather reflects the degree of endoscopic insufflation after the perforation occurred.74 Up to 29% of asymptomatic patients have evidence of retroperitoneal air on CT scan performed 24 hours after ERCP.75 This is primarily related to air insufflation during endoscopy, suggesting that the presence of retroperitoneal air in an asymptomatic patient is not indicative of perforation; in such cases only in-hospital observation is required.73 Pneumomediastinum may be seen in addition to the retroperitoneal air, but in and of itself, is not an indication for surgery.73
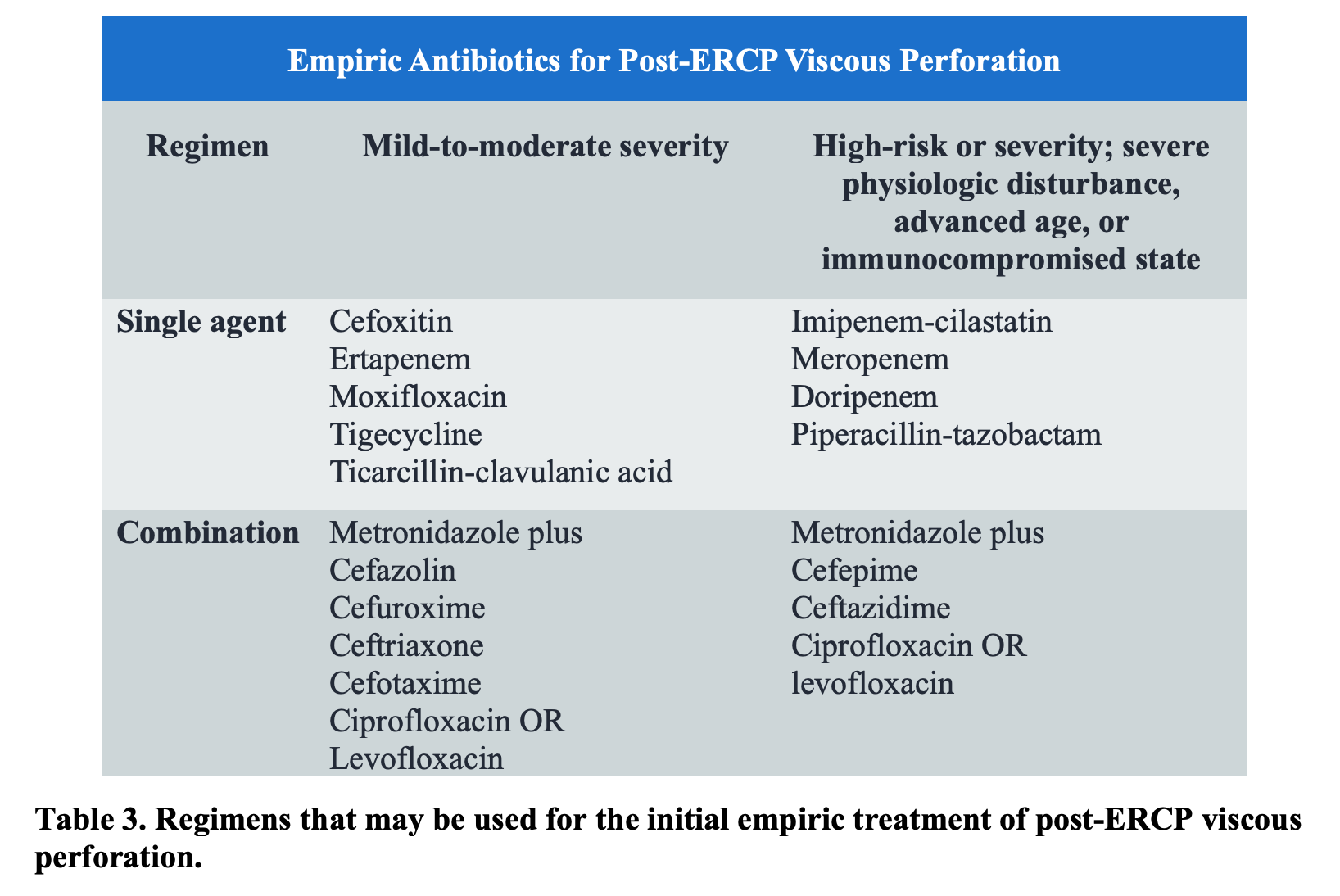
Treatment of post-ERCP perforation varies. Patients in extremis with diffuse peritonitis require rapid intravenous fluid resuscitation, initiation of empiric antibiotics, and emergency surgery.61,76 Hemodynamically stable patients are approached similarly but with a focus on preoperative optimization with intravenous fluid resuscitation as well as correction of any coagulopathy, acidosis, and electrolyte disturbances. Many of these patients will require gastric decompression via naso- or oro-gastric tube, strict fasting, and empiric antibiotics. Antimicrobial therapy for presumed intra-abdominal infection should target enteric gram-negative aerobic and facultative bacilli and enteric gram-positive streptococci (Table 3).77 When using this table, providers can use either single or combination therapy. Patients with post-ERCP perforation require hospitalization. Early consultation with general surgery and gastroenterology is recommended for source control.
Biliary Tract Infections
Urgent biliary decompression, often by ERCP, is essential in the management of acute obstructive cholangitis.78 However, infection is also a complication of ERCP, including acute cholangitis, cholecystitis, liver abscess formation, and infections of pancreatic pseudocysts.16 The incidence of post-ERCP cholangitis ranges from 0.4-10%, carrying a mortality rate of 0.1%.7,16 This is similar to the incidence of acute cholecystitis, which ranges from 0.5-13.5% in some populations, although the associated mortality rate is difficult to differentiate from post-ERCP cholangitis.16,79
In cholangitis, bacteria may enter the biliary tree hematogenously or retrograde during endoscopy with direct instrumentation.80 Post ERCP cholecystitis is a closely related entity, thought to be related to gallbladder contamination from contrast material in the setting of duct obstruction or biliary dyskinesia.16 These biliary infections are typically unimicrobial; the most common organisms implicated are Enterobacteriaceae (Escherichia coli and Klebsiella species), Staphylococcus epidermidis, alpha hemolytic streptococci, Enterococcus, and Pseudomonas aeruginosa.81,82
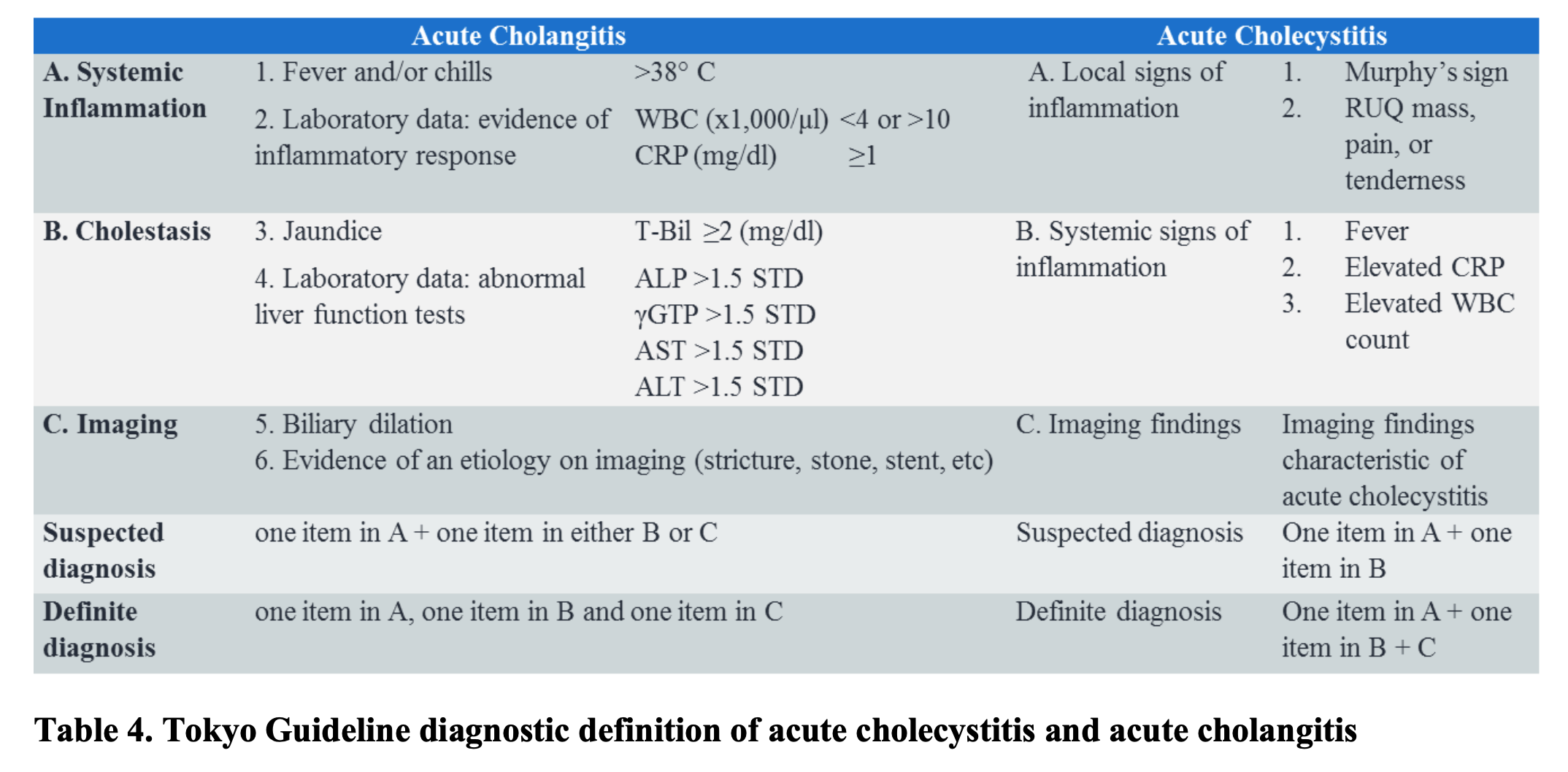
Acute cholecystitis and cholangitis should be suspected in those patients presenting with a combination of fever and right upper quadrant pain. Charcot’s Triad (fever, right upper quadrant pain, and jaundice) occurs in only 50-75% of patients with acute cholangitis, and carries a poor sensitivity (21.2-26.4%). The development of confusion and hypotension may occur in patients with suppurative cholangitis, or Reynolds pentad, which is reported in 3.5–7.7% of the patients.83 Diagnosis is most commonly based on the Tokyo Guidelines. The Tokyo Guidelines are a set of clinical and radiologic diagnostic criteria for acute cholecystitis and cholangitis created to address the controversy regarding the optimal criteria for clinical diagnosis (Table 4).83,84
Initial lab studies in the evaluation of post-ERCP biliary tract infections include a CBC, CMP, urine pregnancy test, coagulation studies, venous blood gas, and lactic acid.85 Blood cultures help guide antibiotic selection for hospitalized patients.86 Patients typically have an elevated white blood cell count with predominance of neutrophils, as well as elevations in the serum alkaline phosphatase, gamma-glutamyl transpeptidase, and bilirubin (predominantly conjugated).86 Acute hepatocyte necrosis occur with micro abscess formation in the liver, indicated by aminotransferases elevations as high as 2000 IU/L.87 Imaging for the diagnosis and evaluation of biliary tract infections include RUQ ultrasound, CT, magnetic resonance cholangiopancreatography (MRCP), and ERCP.87 ERCP and MRCP are highly sensitive in assessing the biliary tree (reported at 100%), but may have limited availability in the ED setting.88 RUQ ultrasound is the initial imaging study of choice, as it has a high specificity for bile duct dilation and bile duct stones (94 to 100%).89 Abdominal CT without contrast can identify bile duct dilatation as well as stenosis with high sensitivity and specificity (65 to 88% and 73 to 97%, respectively).83 An abdominal CT should be performed in patients with suspected acute cholangitis who have a normal RUQ ultrasound, or when the diagnosis is in doubt.
Mild-to-moderate acute cholecystitis or cholangitis may be managed with a carbapenem, fluoroquinolone, penicillin with beta-lactamase inhibitor, or fourth-generation cephalosporin.90 Ampicillin/sulbactam has poor antimicrobial activity against Escherichia coli and has been removed from the North American guidelines.90 Severe disease or in suspected health care−associated disease, vancomycin should be added to the monotherapy to cover Enterococcus species. Linezolid or daptomycin is recommended if the patient is colonized by vancomycin‐resistant Enterococcus (VRE).
Many less-severe infections respond to medical therapy alone. However, early recognition of patients requiring immediate procedural intervention is crucial, as delayed biliary decompression after failure of medical therapy carries a mortality rate up to 80%.91 Although there is no high‐quality evidence in making patients with acute cholangitis/cholecystitis NPO, in principle patients should be fasted in case of emergent procedures.85 It is important to involve gastroenterology, general surgery, and interventional radiology in the management of these patients early as they may require immediate biliary decompression or cholecystectomy.85,92
Case Conclusion: Your patient is presenting within the expected timeframe for post-ERCP pancreatitis and has multiple risk factors including age, female sex, and obesity. Her labs reveal elevations in her lipase and amylase 3x the upper limit of normal, with no evidence of biliary obstruction on formal imaging. You give her intravenous fluids, parenteral pain medications, and admit her for continued management.
Take Home Points:
- The most common post-ERCP complication is acute pancreatitis, followed by gastrointestinal bleeding, viscous perforation, and biliary tract infections.
- Risk factors for post-ERCP complications include patient factors (aberrant anatomy, renal failure, presence of coagulopathy), provider factors (experience and appropriate endoscope disinfection), and procedure factors (multiple cannulation attempts, contrast injection, intraoperative gallbladder opacification).
- A transient increase in serum pancreatic enzymes is common following ERCP, found in up to 75% of patients.
- When evaluating a post-ERCP viscous perforation, the amount of intraperitoneal air does not correlate with the severity of the perforation, but rather reflects the degree of endoscopic insufflation after the perforation occurred.
- Up to 29% of asymptomatic patients have evidence of retroperitoneal air on CT scan performed 24 hours after ERCP.
- Most post-ERCP gastrointestinal bleeding is mild, however, severe intra-abdominal hemorrhage can occur.
- Post-ERCP biliary infections are typically unimicrobial; the most common organisms implicated are Enterobacteriaceae (Escherichia coli and Klebsiella species), Staphylococcus epidermidis, alpha hemolytic streptococci, Enterococcus, and Pseudomonas aeruginosa.
References/Further Reading:
- Ahmed M, Kanotra R, Savani GT, et al. Utilization trends in inpatient endoscopic retrograde cholangiopancreatography (ERCP): A cross-sectional US experience. Endosc Int Open. 2017;5(4):E261-E271.
- Peery AF, Dellon ES, Lund J, et al. Burden of gastrointestinal disease in the United States: 2012 update. Gastroenterology.2012;143(5):1179-1187 e1173.
- Committee ASoP, Maple JT, Ben-Menachem T, et al. The role of endoscopy in the evaluation of suspected choledocholithiasis. Gastrointest Endosc.2010;71(1):1-9.
- Baron TH, Mallery JS, Hirota WK, et al. The role of endoscopy in the evaluation and treatment of patients with pancreaticobiliary malignancy. Gastrointest Endosc. 2003;58(5):643-649.
- Jeurnink SM, Poley JW, Steyerberg EW, Kuipers EJ, Siersema PD. ERCP as an outpatient treatment: a review. Gastrointest Endosc. 2008;68(1):118-123.
- Rabago L, Guerra I, Moran M, et al. Is outpatient ERCP suitable, feasible, and safe? The experience of a Spanish community hospital. Surg Endosc. 2010;24(7):1701-1706.
- Andriulli A, Loperfido S, Napolitano G, et al. Incidence rates of post-ERCP complications: a systematic survey of prospective studies. Am J Gastroenterol. 2007;102(8):1781-1788.
- Williams EJ, Taylor S, Fairclough P, et al. Risk factors for complication following ERCP; results of a large-scale, prospective multicenter study. Endoscopy.2007;39(9):793-801.
- Kochar B, Akshintala VS, Afghani E, et al. Incidence, severity, and mortality of post-ERCP pancreatitis: a systematic review by using randomized, controlled trials. Gastrointest Endosc. 2015;81(1):143-149 e149.
- Freeman ML, Guda NM. Prevention of post-ERCP pancreatitis: a comprehensive review. Gastrointest Endosc. 2004;59(7):845-864.
- Freeman ML, Overby C, Qi D. Pancreatic stent insertion: consequences of failure and results of a modified technique to maximize success. Gastrointest Endosc. 2004;59(1):8-14.
- Thaker AM, Mosko JD, Berzin TM. Post-endoscopic retrograde cholangiopancreatography pancreatitis. Gastroenterol Rep (Oxf). 2015;3(1):32-40.
- Johnson CD, Besselink MG, Carter R. Acute pancreatitis. BMJ. 2014;349:g4859.
- Gapp J, Chandra S. Acute Pancreatitis. In: StatPearls.Treasure Island (FL)2019.
- Parekh PJ, Majithia R, Sikka SK, Baron TH. The “Scope” of Post-ERCP Pancreatitis. Mayo Clin Proc. 2017;92(3):434-448.
- Committee ASoP, Chandrasekhara V, Khashab MA, et al. Adverse events associated with ERCP. Gastrointest Endosc. 2017;85(1):32-47.
- Thomas PR, Sengupta S. Prediction of pancreatitis following endoscopic retrograde cholangiopancreatography by the 4-h post procedure amylase level. J Gastroenterol Hepatol. 2001;16(8):923-926.
- Gottlieb K, Sherman S, Pezzi J, Esber E, Lehman GA. Early recognition of post-ERCP pancreatitis by clinical assessment and serum pancreatic enzymes. Am J Gastroenterol. 1996;91(8):1553-1557.
- Phillip V, Steiner JM, Algul H. Early phase of acute pancreatitis: Assessment and management. World J Gastrointest Pathophysiol. 2014;5(3):158-168.
- Bollen TL, Singh VK, Maurer R, et al. A comparative evaluation of radiologic and clinical scoring systems in the early prediction of severity in acute pancreatitis. Am J Gastroenterol. 2012;107(4):612-619.
- Koo BC, Chinogureyi A, Shaw AS. Imaging acute pancreatitis. Br J Radiol. 2010;83(986):104-112.
- Silverstein W, Isikoff MB, Hill MC, Barkin J. Diagnostic imaging of acute pancreatitis: prospective study using CT and sonography. AJR Am J Roentgenol. 1981;137(3):497-502.
- Tenner S, Baillie J, DeWitt J, Vege SS, American College of G. American College of Gastroenterology guideline: management of acute pancreatitis. Am J Gastroenterol. 2013;108(9):1400-1415; 1416.
- Waller A, Long B, Koyfman A, Gottlieb M. Acute Pancreatitis: Updates for Emergency Clinicians. J Emerg Med. 2018;55(6):769-779.
- Brown A, Baillargeon JD, Hughes MD, Banks PA. Can fluid resuscitation prevent pancreatic necrosis in severe acute pancreatitis? Pancreatology. 2002;2(2):104-107.
- Crockett SD, Wani S, Gardner TB, Falck-Ytter Y, Barkun AN, American Gastroenterological Association Institute Clinical Guidelines C. American Gastroenterological Association Institute Guideline on Initial Management of Acute Pancreatitis. Gastroenterology. 2018;154(4):1096-1101.
- Forsmark CE, Baillie J, Practice AGAIC, Economics C, Board AGAIG. AGA Institute technical review on acute pancreatitis.Gastroenterology. 2007;132(5):2022-2044.
- Smith DI, Hoang K, Gelbard W. Treatment of acute flares of chronic pancreatitis pain with ultrasound guided transversus abdominis plane block: a novel application of a pain management technique in the acute care setting. Case Rep Emerg Med. 2014;2014:759508.
- Working Group IAPAPAAPG. IAP/APA evidence-based guidelines for the management of acute pancreatitis. Pancreatology. 2013;13(4 Suppl 2):e1-15.
- Cotton PB, Garrow DA, Gallagher J, Romagnuolo J. Risk factors for complications after ERCP: a multivariate analysis of 11,497 procedures over 12 years. Gastrointest Endosc. 2009;70(1):80-88.
- Rustagi T, Jamidar PA. Endoscopic retrograde cholangiopancreatography-related adverse events: general overview. Gastrointest Endosc Clin N Am. 2015;25(1):97-106.
- Cho CL, Yuen KK, Yuen CH, Chong LC, Chu RW. Splenic laceration after endoscopic retrograde cholangiopancreatography. Hong Kong Med J. 2008;14(2):145-147.
- Zizzo M, Lanaia A, Barbieri I, Zaghi C, Bonilauri S. Subcapsular Hepatic Hematoma After Endoscopic Retrograde Cholangiopancreatography: A Case Report and Review of Literature. Medicine (Baltimore). 2015;94(26):e1041.
- Cardenas A, Crespo G, Balderramo D, Bordas JP, Sendino O, Llach J. Subcapsular liver hematoma after Endoscopic Retrograde Cholangiopancreatography in a liver transplant recipient. Ann Hepatol. 2008;7(4):386-388.
- Wolters F, Ryan B, Beets-Tan R, Dejong C. Delayed massive hemobilia after biliary stenting. Endoscopy. 2003;35(11):976-977.
- Kingsley DD, Schermer CR, Jamal MM. Rare complications of endoscopic retrograde cholangiopancreatography: two case reports. JSLS. 2001;5(2):171-173.
- Freeman ML, Nelson DB, Sherman S, et al. Complications of endoscopic biliary sphincterotomy. N Engl J Med. 1996;335(13):909-918.
- Finnie IA, Tobin MV, Morris AI, Gilmore IT. Late bleeding after endoscopic sphincterotomy for bile duct calculi. BMJ. 1991;302(6785):1144.
- Boujaoude J, Pelletier G, Fritsch J, et al. Management of clinically relevant bleeding following endoscopic sphincterotomy. Endoscopy. 1994;26(2):217-221.
- Gholson CF, Favrot D, Vickers B, Dies D, Wilder W. Delayed hemorrhage following endoscopic retrograde sphincterotomy for choledocholithiasis. Dig Dis Sci. 1996;41(5):831-834.
- Ferreira LE, Fatima J, Baron TH. Clinically significant delayed postsphincterotomy bleeding: a twelve year single center experience. Minerva Gastroenterol Dietol. 2007;53(3):215-223.
- Kim BS, Li BT, Engel A, et al. Diagnosis of gastrointestinal bleeding: A practical guide for clinicians. World J Gastrointest Pathophysiol. 2014;5(4):467-478.
- Savatmongkorngul S, Wongwaisayawan S, Kaewlai R. Focused assessment with sonography for trauma: current perspectives.Open Access Emerg Med. 2017;9:57-62.
- Barkun AN, Bardou M, Kuipers EJ, et al. International consensus recommendations on the management of patients with nonvariceal upper gastrointestinal bleeding. Ann Intern Med. 2010;152(2):101-113.
- Wortman JR, Landman W, Fulwadhva UP, Viscomi SG, Sodickson AD. CT angiography for acute gastrointestinal bleeding: what the radiologist needs to know. Br J Radiol. 2017;90(1075):20170076.
- Talukdar R. Complications of ERCP. Best Pract Res Clin Gastroenterol. 2016;30(5):793-805.
- Osman D, Djibre M, Da Silva D, Goulenok C, group of e. Management by the intensivist of gastrointestinal bleeding in adults and children. Ann Intensive Care. 2012;2(1):46.
- Hayat U, Lee PJ, Ullah H, Sarvepalli S, Lopez R, Vargo JJ. Association of prophylactic endotracheal intubation in critically ill patients with upper GI bleeding and cardiopulmonary unplanned events. Gastrointest Endosc. 2017;86(3):500-509 e501.
- Heffner AC, Swords D, Kline JA, Jones AE. The frequency and significance of postintubation hypotension during emergency airway management. J Crit Care. 2012;27(4):417 e419-413.
- Rabenstein T, Schneider HT, Hahn EG, Ell C. 25 years of endoscopic sphincterotomy in Erlangen: assessment of the experience in 3498 patients. Endoscopy. 1998;30(9):A194-201.
- Saeed M, Kadir S, Kaufman SL, Murray RR, Milligan F, Cotton PB. Bleeding following endoscopic sphincterotomy: angiographic management by transcatheter embolization. Gastrointest Endosc. 1989;35(4):300-303.
- Millward SF. ACR Appropriateness Criteria on treatment of acute nonvariceal gastrointestinal tract bleeding. J Am Coll Radiol. 2008;5(4):550-554.
- So YH, Choi YH, Chung JW, Jae HJ, Song SY, Park JH. Selective embolization for post-endoscopic sphincterotomy bleeding: technical aspects and clinical efficacy. Korean J Radiol. 2012;13(1):73-81.
- Dunne R, McCarthy E, Joyce E, et al. Post-endoscopic biliary sphincterotomy bleeding: an interventional radiology approach. Acta Radiol. 2013;54(10):1159-1164.
- Masci E, Toti G, Mariani A, et al. Complications of diagnostic and therapeutic ERCP: a prospective multicenter study. Am J Gastroenterol. 2001;96(2):417-423.
- Loperfido S, Angelini G, Benedetti G, et al. Major early complications from diagnostic and therapeutic ERCP: a prospective multicenter study. Gastrointest Endosc. 1998;48(1):1-10.
- Colton JB, Curran CC. Quality indicators, including complications, of ERCP in a community setting: a prospective study. Gastrointest Endosc. 2009;70(3):457-467.
- Howard TJ, Tan T, Lehman GA, et al. Classification and management of perforations complicating endoscopic sphincterotomy. Surgery. 1999;126(4):658-663; discussion 664-655.
- Stapfer M, Selby RR, Stain SC, et al. Management of duodenal perforation after endoscopic retrograde cholangiopancreatography and sphincterotomy. Ann Surg. 2000;232(2):191-198.
- Soreide JA, Viste A. Esophageal perforation: diagnostic work-up and clinical decision-making in the first 24 hours. Scand J Trauma Resusc Emerg Med. 2011;19:66.
- Langell JT, Mulvihill SJ. Gastrointestinal perforation and the acute abdomen. Med Clin North Am. 2008;92(3):599-625, viii-ix.
- Okten I, Cangir AK, Ozdemir N, Kavukcu S, Akay H, Yavuzer S. Management of esophageal perforation. Surg Today. 2001;31(1):36-39.
- Rubesin SE, Levine MS. Radiologic diagnosis of gastrointestinal perforation. Radiol Clin North Am. 2003;41(6):1095-1115, v.
- Kaman L, Iqbal J, Kundil B, Kochhar R. Management of Esophageal Perforation in Adults. Gastroenterology Res. 2010;3(6):235-244.
- Maniatis V, Chryssikopoulos H, Roussakis A, et al. Perforation of the alimentary tract: evaluation with computed tomography. Abdom Imaging. 2000;25(4):373-379.
- Backer CL, LoCicero J, 3rd, Hartz RS, Donaldson JS, Shields T. Computed tomography in patients with esophageal perforation. Chest. 1990;98(5):1078-1080.
- Coppolino F, Gatta G, Di Grezia G, et al. Gastrointestinal perforation: ultrasonographic diagnosis. Crit Ultrasound J. 2013;5 Suppl 1:S4.
- Gans SL, Stoker J, Boermeester MA. Plain abdominal radiography in acute abdominal pain; past, present, and future. Int J Gen Med. 2012;5:525-533.
- Chao A, Gharahbaghian L, Perera P. Diagnosis of pneumoperitoneum with bedside ultrasound. West J Emerg Med. 2015;16(2):302.
- Abu-Zidan FM, Cevik AA. Diagnostic point-of-care ultrasound (POCUS) for gastrointestinal pathology: state of the art from basics to advanced. World J Emerg Surg. 2018;13:47.
- Zissin R, Shapiro-Feinberg M, Oscadchy A, Pomeranz I, Leichtmann G, Novis B. Retroperitoneal perforation during endoscopic sphincterotomy: imaging findings. Abdom Imaging. 2000;25(3):279-282.
- Borofsky S, Taffel M, Khati N, Zeman R, Hill M. The emergency room diagnosis of gastrointestinal tract perforation: the role of CT. Emerg Radiol. 2015;22(3):315-327.
- Wu HM, Dixon E, May GR, Sutherland FR. Management of perforation after endoscopic retrograde cholangiopancreatography (ERCP): a population-based review. HPB (Oxford). 2006;8(5):393-399.
- Ruiz-Tovar J, Lobo E, Sanjuanbenito A, Martinez-Molina E. Case series: pneumoretroperitoneum secondary to duodenal perforation after endoscopic retrograde cholangiopancreatography. Can J Surg. 2009;52(1):68-69.
- Genzlinger JL, McPhee MS, Fisher JK, Jacob KM, Helzberg JH. Significance of retroperitoneal air after endoscopic retrograde cholangiopancreatography with sphincterotomy. Am J Gastroenterol. 1999;94(5):1267-1270.
- Booth FV, Doerr RJ, Khalafi RS, Luchette FA, Flint LM, Jr. Surgical management of complications of endoscopic sphincterotomy with precut papillotomy. Am J Surg. 1990;159(1):132-135; discussion 135-136.
- Solomkin JS, Mazuski JE, Bradley JS, et al. Diagnosis and management of complicated intra-abdominal infection in adults and children: guidelines by the Surgical Infection Society and the Infectious Diseases Society of America. Clin Infect Dis. 2010;50(2):133-164.
- Khashab MA, Tariq A, Tariq U, et al. Delayed and unsuccessful endoscopic retrograde cholangiopancreatography are associated with worse outcomes in patients with acute cholangitis. Clin Gastroenterol Hepatol. 2012;10(10):1157-1161.
- Nelson DB. Infectious disease complications of GI endoscopy: part II, exogenous infections. Gastrointest Endosc. 2003;57(6):695-711.
- Szary NM, Al-Kawas FH. Complications of endoscopic retrograde cholangiopancreatography: how to avoid and manage them. Gastroenterol Hepatol (N Y). 2013;9(8):496-504.
- Kullman E, Borch K, Lindstrom E, Ansehn S, Ihse I, Anderberg B. Bacteremia following diagnostic and therapeutic ERCP. Gastrointest Endosc. 1992;38(4):444-449.
- Subhani JM, Kibbler C, Dooley JS. Review article: antibiotic prophylaxis for endoscopic retrograde cholangiopancreatography (ERCP). Aliment Pharmacol Ther. 1999;13(2):103-116.
- Kiriyama S, Kozaka K, Takada T, et al. Tokyo Guidelines 2018: diagnostic criteria and severity grading of acute cholangitis (with videos). J Hepatobiliary Pancreat Sci. 2018;25(1):17-30.
- Yokoe M, Hata J, Takada T, et al. Tokyo Guidelines 2018: diagnostic criteria and severity grading of acute cholecystitis (with videos). J Hepatobiliary Pancreat Sci. 2018;25(1):41-54.
- Miura F, Okamoto K, Takada T, et al. Tokyo Guidelines 2018: initial management of acute biliary infection and flowchart for acute cholangitis. J Hepatobiliary Pancreat Sci. 2018;25(1):31-40.
- Ahmed M. Acute cholangitis – an update. World J Gastrointest Pathophysiol. 2018;9(1):1-7.
- Attasaranya S, Fogel EL, Lehman GA. Choledocholithiasis, ascending cholangitis, and gallstone pancreatitis. Med Clin North Am. 2008;92(4):925-960, x.
- de Ledinghen V, Lecesne R, Raymond JM, et al. Diagnosis of choledocholithiasis: EUS or magnetic resonance cholangiography? A prospective controlled study. Gastrointest Endosc. 1999;49(1):26-31.
- Pasanen PA, Partanen KP, Pikkarainen PH, Alhava EM, Janatuinen EK, Pirinen AE. A comparison of ultrasound, computed tomography and endoscopic retrograde cholangiopancreatography in the differential diagnosis of benign and malignant jaundice and cholestasis. Eur J Surg. 1993;159(1):23-29.
- Gomi H, Solomkin JS, Schlossberg D, et al. Tokyo Guidelines 2018: antimicrobial therapy for acute cholangitis and cholecystitis. J Hepatobiliary Pancreat Sci. 2018;25(1):3-16.
- Salek J, Livote E, Sideridis K, Bank S. Analysis of risk factors predictive of early mortality and urgent ERCP in acute cholangitis. J Clin Gastroenterol. 2009;43(2):171-175.
- Okamoto K, Suzuki K, Takada T, et al. Tokyo Guidelines 2018: flowchart for the management of acute cholecystitis. J Hepatobiliary Pancreat Sci. 2018;25(1):55-72.






1 thought on “Complications of ERCP: ED presentations, evaluation, and management”
Pingback: Quiz 36, May 24th 2019