Authors: Brit Long, MD (@long_brit, EM Attending Physician, San Antonio, TX) and Courtney M. Lee, EM PA (Joint Base Elmendorf Richardson Medical Center, Emergency Medicine) // Edited by: Alex Koyfman, MD (@EMHighAK, EM Attending Physician, UT Southwestern Medical Center / Parkland Memorial Hospital)
Welcome back to emDocs Cases! Today we have case-based discussion on a core EM topic, with a look at some controversy and cutting edge treatments.
———————————————————————————
End stage renal disease (ESRD) is all too common in the U.S., defined by glomerular filtration rate (GFR) less than 15 mL/min.1-5 These patients most commonly die from cardiovascular disease, and sepsis is the most common cause of hospitalization.2-7 There are several types of renal failure, including failure to excrete waste products, failure to synthesize hormones, and failure to regulate physiologic feedback systems.1-5 In order to replace renal function in ESRD, patients typically undergo transplant or dialysis (hemodialysis (HDS) or peritoneal (PD)).
A 42-year-old male with history of renal disease on dialysis, hypertension, HIV, and diabetes presents to the ED, brought in by a friend. He received HDS yesterday. VS include HR 92, BP 100/70, RR 18, Room air oxygen 98%, T 98oF. We are going to take this patient through a variety of symptoms and conditions, but before we start a more focused history and exam, let’s discuss HDS and the initial patient assessment.
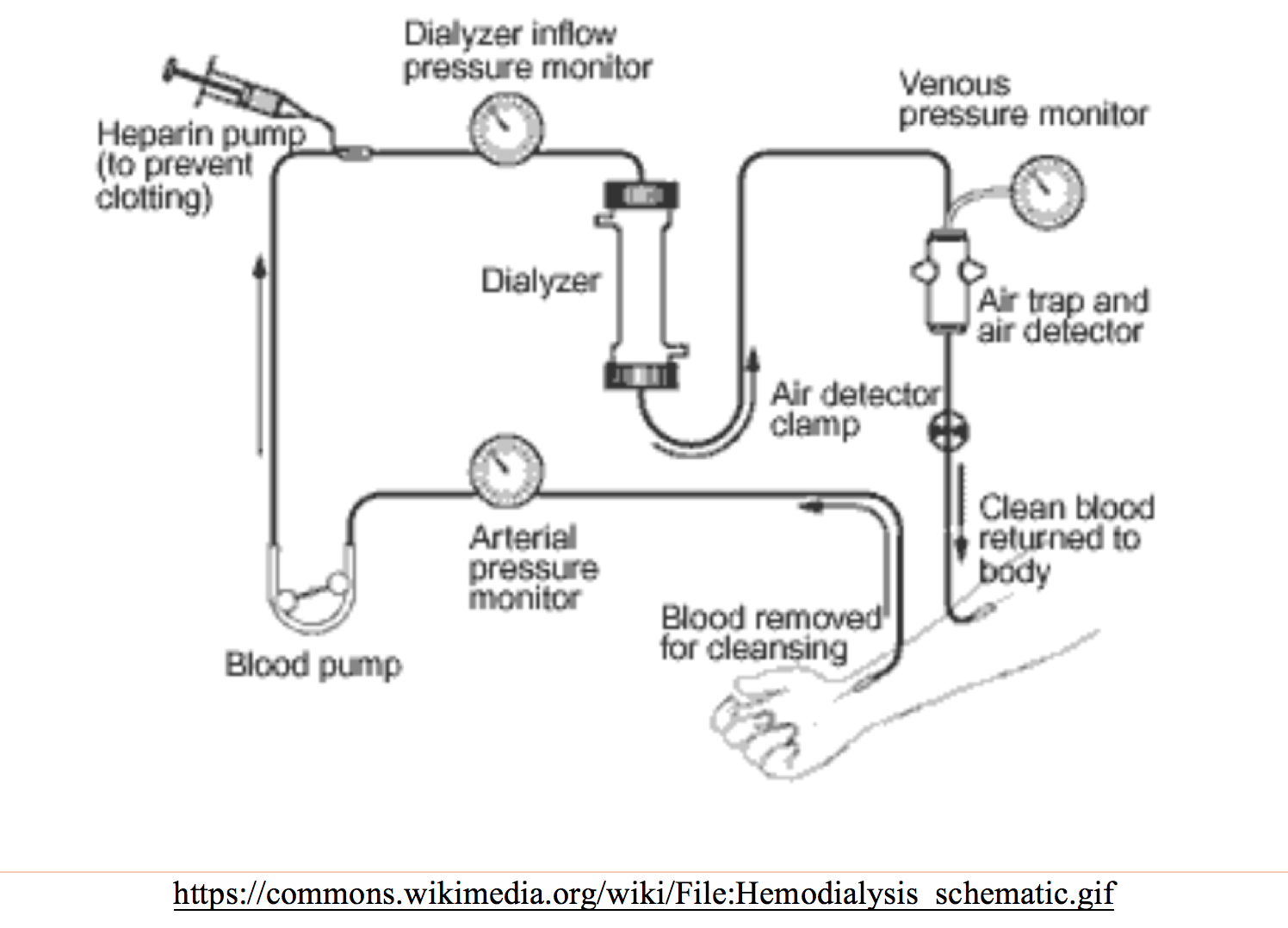
Hemodialysis: Involves a filter to produce plasma ultrafiltrate, with access site including arteriovenous (AV) fistula (lowest complication rate), graft, or catheter. Fluid removal is controlled by pressure gradient across filter, with clearance dependent on filter pore size, ultrafiltration, and concentration gradient.1-6 Heparin is often used during HDS for the access site, with sessions lasting approximately 4 hours (shorter times are associated with worse outcomes).1-4
History: Etiology of ESRD, dialysis schedule, recently missed dialysis sessions, time for each session, patient dry weight, average interdialytic weight gain, uremic symptoms, prior complications, urine production, and present of native kidneys.4-9
Exam: Vital signs, cardiopulmonary (heart failure signs, murmurs, lungs for wheezes/rales, distant heart sounds, peripheral edema), vascular access site (bruit, thrill, erythema, warmth, discharge, bleeding, swelling), and neurologic assessment (mental status, focal neurologic deficit, peripheral reflexes, sensation, and asterixis).4-9
———————————————————————————
Let’s say the patient is altered and confused after missing one dialysis session. You find asterixis on exam. D-stick is 122.

Neurologic: A variety of neurologic complications are common. One of the most common is uremic encephalopathy. A specific level of BUN or uremia is not reliable for diagnosis, and uremic encephalopathy is a diagnosis of exclusion. A variety of other conditions must be considered before latching onto this diagnosis. For the altered patient, these conditions include intracerebral hemorrhage (ICH) or ischemic stroke. Rapid blood glucose assessment, BMP, CBC, TSH, ECG, and Head CT noncontrast should be obtained.4,5,9,10
- Uremic encephalopathy – Due to over 70 toxins and neurotransmitter imbalance, with increase in those missing dialysis. Primarily affects those with GFR < 15 mL/min. Older patients and those with comorbidities typically demonstrate more severe symptoms. Asterixis may be seen. Early signs consist of mood swings, weakness, and irritability, transitioning into tremor, asterixis, lethargy, and even coma. Neurologic deficits may change during the disease course. Treatment includes dialysis, though neurologic changes may persist for several days after dialysis.9-13
- Dialysis Disequilibrium Syndrome (DDS) – Due to serum osmolality changes in HDS, resulting in diffusion of water into the CNS. Risks include first HDS treatment, BUN > 60 mmol/L (175 mg/dL), CKD, acidosis. Treatment includes slowing or stopping HDS. Symptoms usually resolve after several hours. Symptomatic treatment warranted for nausea and vomiting. If seizing, provide a hypertonic solution (mannitol or 5ml 10-20% NaCl).9,10,14,15
- ICH/ischemic stroke – ESRD patients are at high risk for stroke, and if stroke occurs, ESRD is a poor prognostic marker. Subdural hematoma occurs 10-20X more frequently. Altered mental status is more common in patients with ICH, and patients may have focal deficit.10,16-20 Ischemic stroke is also more common in this population, with worse outcomes. CT head, ECG, coagulation panel, CBC, RFP are needed. Thrombolytics can be provided for ischemic stroke if contraindications are not present.9,19,22 If ICH is present, reversal of anticoagulation may be needed, along with neurosurgical consultation.7,9,19-21
———————————————————————————
Instead of altered mental status, let’s switch gears. The patient presents with chest pain and shortness of breath. What conditions do you need to consider?
Cardiopulmonary: These patients are at significant risk for cardiopulmonary complications, with cardiovascular disease accounting for over 40% of mortality.7-9,23-25 Electrolyte abnormalities, LVH, hypercholesterolemia, and atherosclerosis predispose to increased risk of ACS, and ESRD patients are also at increased risk for severe arrhythmias and heart failure, with many patients receiving an implantable cardiac device.7-9,26-30
- Pericarditis/Effusion/Tamponade – Pericarditis classically presents with fever, sharp chest pain that radiates to the trapezius, chest pain that worsens in supine position, and pericardial friction rub (more common in uremic pericarditis) on exam. However, this rub is not always present. Patients may present with dyspnea, and pericardial effusion may occur in 20% of patients. Fluid from uremic pericardial effusion is typically sterile with fibrin. BUN is often greater than 60 mg/dL, and the ECG may not reveal the normal stages of pericarditis. Chest X-ray may show enlarged cardiac size if the effusion is longstanding. Ultrasound (US) is an important measure for these patients to evaluate for evidence of tamponade. Poor prognostic findings warranting admission include hemodynamic instability, WBC > 13x 109/L, fever, effusion > 2 cm, poor social situation, and poor compliance with dialysis. Patients who are hypotensive require immediate assessment for tamponade, with IV fluid bolus to enhance preload. Pericardiocentesis with IV fluid is required in the peri-arrest state. For patients with pericarditis who may be appropriate for discharge with no effusion, acetaminophen should be considered, with dialysis.5-9,31,32
- ACS – This accounts for the highest percentage of deaths in these patients. Patients may not present typically with chest pain, but nausea, shortness of breath, and weakness, and the ECG at baseline may demonstrate LVH or other abnormalities. Troponin may be elevated at baseline, but a change from baseline or elevation (by 20%) on repeat assessment is suggestive of NSTEMI.4,7,9,33-38 These patients should be given aspirin and standard ACS medications. No dose adjustment is needed for aspirin, clopidogrel, unfractionated heparin, or thrombolytics, but lovenox dosing does require adjustment. If STEMI is present, PCI or thrombolytics is needed.9,34,35
- Pulmonary Edema – Fluid overload is the most frequent cause of dyspnea is ESRD and is most commonly due to missed HDS session. However, ACS or CHF may also result in overload. Rapid IV access, ECG, monitor, and oxygen are required. US of the heart and lungs can be beneficial (cardiac function, presence of B lines, IVC assessment). Immediate resuscitation is needed, with nitroglycerin (SL or IV) and noninvasive positive pressure ventilation if in respiratory distress. HDS is required, and phlebotomy can function as a bridge to HDS if other measures fail.9,39-42
- Air Embolism – Venous air embolism is the most common form and may occur during access placement, removal, or during HDS session. Air typically travels to the right side of the heart, lodging in the pulmonary circulation, but arterial embolism may occur in the setting of R to L shunt. These patients present with sudden respiratory distress, chest pain, hypotension, neurologic abnormalities, livedo reticularis, crepitus over involved vessels, and a mill wheel murmur (churning sound heard throughout the entire cardiac cycle).5,7,9,43-47 ECG, CK, CBC, renal function, VBG, US, and chest X-ray are needed. Thrombocytopenia and elevated CK may be found, and chest X-ray may show focal atelectasis, oligemia, or pulmonary artery enlargement. Rapid assessment of the ABCs with high flow oxygen is needed. The patient should be placed in the left lateral decubitus or Trendelenburg position for venous air embolism or supine position for arterial embolism. The catheter should be clamped and HDS discontinued. Aspiration of air from the RV is present in case report literature. Hyperbaric oxygen therapy can be used for end organ dysfunction.5,7,9,43-47
———————————————————————————
The patient presents with redness and pain along the access site, as well as fever, tachycardia, and generalized weakness. He meets criteria for sepsis on your evaluation of vital signs with suspected source.

Infectious: Patients with severe renal disease are at high risk for infection, with catheter based infections the most common cause of bacteremia and sepsis (followed by lower respiratory tract infection).5,9,48-53 As renal function decreases, the risk of infection drastically rises due to decreased T-cell activation and decreased leukocyte function.5,9,48-53 The typical infections like pneumonia and cellulitis may occur, but interestingly these patients are at high risk for Clostridium difficile infection, recurrence, and mortality.53 UTI may also develop, even without urine production. Catheter-related infections possess an incidence of 5.5 episodes per 1000 catheter days. Up to 1/3 are due to Gram-positive cocci, with 20% polymicrobial.52-58 The risk of infection due to MRSA is 100 times greater in ESRD patients on HDS, with catheter at higher risk than fistula (8X). Patients with sepsis from suspected access site infection warrant broad-spectrum coverage: vancomycin at normal loading dose (20 mg/kg IV) AND third generation cephalosporin such as ceftazidime 2 g IV or gentamicin 1-2 mg/kg IV (not exceeding 100mg).7,52-58 Cultures should be obtained from the suspected site and a separate peripheral site. Infections in other body locations from an access site are considered metastatic infections, such as endocarditis.57,58
What about the hypotensive septic patient? How much fluid should you provide? The Surviving Sepsis Campaign recommends 30 mL/kg in sepsis.59 Fluid assessment in ESRD patients is not easy, but these patients in sepsis are intravascularly depleted. Unfortunately, literature suggests patients with ESRD and dialysis dependency do not receive the recommended 30 mL/kg (commonly less than 1L total in 6 hours).60 Patients who do receive 30 mL/kg or greater do not demonstrate adverse events associated with IV fluid resuscitation. These patients should receive IV fluids with repeat assessments.5,9,59,60
———————————————————————————
Time for another alteration in patient case…The patient’s access site has not stopped bleeding since his dialysis session this morning. He is tachycardic, and EMS placed a large dressing over the site. What should you consider in management of his bleeding?
Vascular Access: In the U.S., close to 400,000 patients with chronic kidney disease use HDS, and approximately 25% of these patients are hospitalized annually due to access site complications, dependent of the type access.61-63 If HDS is to be used, a fistula is the optimal form for access.64 ESRD patients are at high risk for bleeding due to uremia (vWF factor disruption) and normocytic normochromic anemia. Coagulation studies are usually normal, and stage of ESRD does not correlate with rates of hemorrhage.64-70 Physicians should assess access site color, swelling, warmth, bruit, and thrill, as well as distal extremity neurovascular status.
- Bleeding – Hemorrhage is multifactorial, with risks including platelet function (uremia) and anticoagulation (heparin used during HDS).64-70 Other risks include infection, stenosis, and aneurysm. Heparin’s half-life approximates 1.5-2 hours. These patients require immediate assessment of hemodynamic status, with rapid control of the bleeding site. IV access, CBC, type and screen, renal function panel, and coagulation panel are recommended.9,63-70 Start with direct pressure to the site for 5-10 minutes. A single suture is useful for linear tear across the access site. Topical hemostatic agents are useful in combination with direct, focal pressure (rather than a large, bulky dressing). To improve platelet function, desmopressin 0.3 mcg/kg IV should be used, which can decrease hemorrhage in 50% of patients. If anticoagulation from heparin is a concern, 1 mg of protamine IV per 100 units heparin is recommended. HDS corrects uremic platelet dysfunction in up to 85% of patients. Further strategies include cryoprecipitate (to replace fibrinogen), and estrogen 25 mg IV. If these measures fail, a tourniquet can be applied.63-70
- Aneurysm/Pseudoaneurysm – These commonly occur due to frequent cannulation, which weakens the vessel wall. Incidence ranges from 5-6%, and stenosis drastically increases the risk. Aneurysms present with pain, motor/sensory dysfunction, skin erosion, and hemorrhage, while pseudoaneurysm is more likely to present with infection or bleeding.5,9,71-74 Doppler US and vascular surgery consultation are needed, with operative repair required.
- Thrombosis/Stenosis – This complication is the most common problem in HDS access sites, with over 75% of patients with grafts experiencing this complication within the first year of placement. Thrombosis/stenosis also increases the risk of aneurysm and pseudoaneurysm. Patients may have extremity edema, increased vein formation, and change in the bruit/thrill. Pain and absence of bruit/thrill are common with thrombosis.5,9,71-73 Doppler US and vascular surgery consultation are needed. Treatment (thrombolysis versus surgical thrombectomy with or without angioplasty) is not required emergently, but should occur within 24-48 hours.71-74
- Infection – We covered access site infection briefly above, but this section will give you a little more of a deep dive. Infection is most common within the first 6 months of placement and is more common in central venous access sites compared to AV fistula (2-5% rate over fistula lifetime).4,5,9,48-50,54,55,75 Close to 10% of grafts will undergo infection, with infection most commonly from Staph aureus, Staph epidermidis, and gram-negative bacteria.54-58 This is the most common cause of graft loss. Evaluate for erythema, induration, and warmth of the access site; however, due to altered immune function, patients may not demonstrate these findings but rather systemic symptoms such as myalgias and fever.9,48-50,55-58 Close to 10% of patients will experience an infection at another site (endocarditis, osteomyelitis, epidural abscess, etc.).48-50,54-57 Cultures should be obtained from the site and a separate peripheral site, with the catheter blood culture often showing a 4-fold higher colony count. Doppler US of the graft, CBC, lactate, renal function, and cultures are required with admission. Vancomycin (20mg/kg) and gentamycin (100 mg) or ceftazidime (2 g) IV are recommended.52,54,58,75
———————————————————————————
The next patient is a 48-year-old female with peritoneal dialysis presenting with cloudy infiltrate and generalized abdominal pain. She is hemodynamically stable, but her abdomen is tender to palpation. Fluid obtained from the peritoneal cavity reveals 242 leukocytes/mm3 with 78% neutrophils. What is this condition, and what is the likely cause?

Peritoneal Dialysis Background: The peritoneal membrane serves as blood-dialysate interface. Dialysate glucose creates an osmotic pressure difference. Exchanges occur most commonly four times per day, with 2L infused into the peritoneal cavity. If completed 4 times per day, this adds to 8L total infused, with typically 10L drained over 24 hours.5,9,76-78
History: Cause of ESRD, compliance with dialysis, type of dialysis, PD parameters (concentration, exchanges per day, time of exchanges), other complications of PD, baseline weight and laboratory values, uremic symptoms, presence of native kidneys, and ability to produce urine should be assessed.5,9,76-78
Exam: Evaluate hemodynamic status. For focused complaints related to peritoneal dialysis, the abdomen should be examined for hernia, tenderness, and catheter site appearance.
Peritonitis: The most common complication of PD, which occurs on average once every 15-18 months with mortality reaching 12.5% per episode. Presentation includes fever, abdominal pain, and potentially rebound tenderness. Cloudy fluid is suggestive.79-84
– Diagnosis includes peritoneal fluid aspiration, with greater than 100 leukocytes/mm3 and > 50% neutrophils. Dwell time of the fluid is important in ensuring appropriate sample collection. Gram stain cannot be relied on for diagnosis, as it is positive in 10-40% of patients. Peritoneal fluid providing a positive urine dipstick is suggestive for peritonitis, but a negative reading cannot rule out the diagnosis.
– Organisms include Staphylococcus epidermidis (30-40%), Staph aureus (10%), Streptococcus (20%), and gram-negative species in 20%.9,79-84
– Treatment includes antibiotics, in consultation with nephrology. These should be provided by rapid intraperitoneal exchanges. This route of treatment is more efficacious than IV. Doses include vancomycin 30 mg/kg, with ceftazidime 1g or gentamycin 0.6 mg/kg IP daily.82-85
Other Complications: Infection around the PD catheter presents with pain, redness, and discharge. S. aureus and Pseudomonas are the most common organisms. If the patient appears toxic, IV antibiotics and admission are required. Otherwise, oral antibiotics with close follow up are needed.84,85 Abdominal hernias occur in close to 15% of patients, and surgical consultation is needed due to high risk of incarceration and strangulation.9,84,85
Key Points
– ESRD is increasing in the U.S., with greater all-cause mortality, cardiovascular events, and hospitalization rates when compared to patients with normal renal function.
– Replacement therapy includes HDS or PD, with each type possessing unique features. A focused history and exam are needed based on the type of dialysis.
– Other organ systems are affected, resulting in neurologic complications, cardiopulmonary pathology, higher rates of infection, and access site complications.
– Neurologic complications include uremic encephalopathy, cerebrovascular pathology, and several others.
– Cardiopulmonary conditions include pericarditis, pericardial effusion/tamponade, acute coronary syndrome, sudden cardiac death, electrolyte abnormalities, pulmonary edema, and air embolism.
– ESRD patients have higher risk of infections, though patients often present atypically.
– Access site complications include bleeding, aneurysm/pseudoaneurysm, thrombosis/stenosis, and infection.
– A common complication of PD is peritonitis. Antibiotics through the intraperitoneal route are the best means of treatment.
*Note: This does not discuss all of the conditions that may result in the specific patient presentations. Keep your differential broad!
References/Further Reading:
- United States Renal Data System (USRDS) Annual Data Report. Epidemiology of kidney disease in the United States. 2015. Available from: https://www.usrds.org/adr.aspx National Kidney and Urologic Diseases Information Clearinghouse (NKUDIC). Kidney Disease Statistics for the United States. US Department of Health and Human Services. Available at http://kidney.niddk.nih.gov/kudiseases/pubs/kustats/#7. Accessed July 4, 2017.
- Chapter 1: Definition and classification of CKD. Kidney Int Suppl 2013; 3:19. http://www.kdigo.org/clinical_practice_guidelines/pdf/CKD/KDIGO_2012_CKD_GL.pdf. Accessed July 4, 2017.
- Go AS,Chertow GM, Fan D, McCulloch CE, Hsu CY. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med. 2004 Sep 23;351(13):1296-305.
- Arulkumaran N, Montero RM, Singer M. Management of the dialysis patient in general intensive care. Br J Anaesth 2012;108(2):183-192.
- Foley M, Mehta N, Sinert R. Chapter 90: End-Stage Renal Disease. Tintinalli’s Emergency Medicine: A Comprehensive Study Guide, 8e. http://accessemergencymedicine.mhmedical.com/content.aspx?bookid=1658§ionid=109433467#1121508429. Accessed 15 July 2017.
- Bradbury BD, Fissell RB, Albert JM, et al. Predictors of early mortality among incident US hemodialysis patients in the Dialysis Outcomes and Practice Patterns Study (DOPPS). Clin J Am Soc Nephrol 2007; 2:89.
- Hodde L, Sandroni S. Emergency department evaluation and management of dialysis patient complications. J Emerg Med. 1992; 10:317-334.
- Flythe JE, Curhan GC, Brunelli SM. Shorter length dialysis sessions are associated with increased mortality, independent of body weight. Kidney international. 2013;83(1):104-113.
- Long B, Koyfman A, Lee M. Emergency Medicine Evaluation and Management of the End Stage Renal Disease Patient. American Journal of Emergency Medicine. 2017; Article in Press.
- Baumgaertel MW, Kraemer M, Berlit P. Neurologic complications of acute and chronic renal disease. Handbook of Clinical Neurology. 2014;119 (3): 383-93.
- Bucurescu M. Neurological manifestations of uremic encephalopathy. Medscape. Available at: http://emedicine.medscape.com/article/1135651-overview. Accessed July 4, 2017.
- Lohr JW. Uremic encephalopathy. Medscape. Available at: http://emedicine.medscape.com/article/239191-overview Accessed July 4, 2017.
- Kennedy AC, Linton AL, Eaton JC. Urea levels in cerebrospinal fluid after haemodialysis. Lancet 1962;1: 410–411.
- Silver SM, Sterns RH, Halperin ML. Brain swelling after dialysis: old urea or new osmoles? Am J Kidney Dis 1996;28:1–13.
- Arieff AI. Dialysis disequilibrium syndrome: current concepts on pathogenesis and prevention. Kidney Int 1994;45:629–635.
- Holzmann MJ, Aastveit A, Hammar N, et al. Renal dysfunction increases the risk of ischemic and hemorrhagic stroke in the general population. Ann Med 2012;44:607–615.
- Power A, Hamady M, Singh S, et al. High but stable incidence of subdural haematoma in haemodialysis – a single-centre study. Nephrol Dial Transplant 2010;25:2272–2275.
- Hsieh CY, Chen CH, Wu AB, et al. Comparative outcomes between hemo- and peritoneal dialysis patients with acute intracerebral hemorrhage. Am J Nephrol 2010;32:31–37.
- Wu C-L, Tsai C-C, Kor C-T, et al. Stroke and Risks of Development and Progression of Kidney Diseases and End-Stage Renal Disease: A Nationwide Population-Based Cohort Study. Burdmann EA, ed. PLoS ONE. 2016;11(6):e0158533.
- Hemphill JC 3rd, Greenberg SM, Anderson CS, et al. Guidelines for the Management of Spontaneous Intracerebral Hemorrhage: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke 2015; 46:2032.
- Connolly ES Jr, Rabinstein AA, Carhuapoma JR, et al. Guidelines for the management of aneurysmal subarachnoid hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2012; 43:1711.
- Jauch EC, Saver JL, Adams HP Jr, et al. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2013; 44:870.
- Hajhosseiny R, Khavandi K, Goldsmith DJ. Cardiovascular disease in chronic kidney disease: untying the Gordian knot. Int J Clin Pract 2013;67:14.
- Kanbay M,Afsar B, Goldsmith D, et al. Sudden death in hemodialysis: an update. Blood Purif. 2010;30(2):135-45
- Wang AY,Lam CW, Chan IH, et al. Sudden cardiac death in end-stage renal disease patients: a 5-year prospective analysis. 2010 Aug;56(2):210-6.
- Alfonzo A, Simpson K, Deighan C. Modifications to advanced life support in renal failure. Resuscitation. 2007;73:12-28.
- Bozbas H,Atar I, Yildirir A, et al. Prevalence and predictors of arrhythmia in end stage renal disease patients on hemodialysis. Ren Fail. 2007;29(3):331-9.
- Malhis M,Al-Bitar S, Farhood S, et al. Changes in QT intervals in patients with end-stage renal disease before and after hemodialysis. Saudi J Kidney Dis Transpl. 2010 May;21(3):460-5.
- Pun PH,Lehrich RW, Honeycutt EF, et al. Modifiable risk factors associated with sudden cardiac arrest within hemodialysis clinics. Kidney Int. 2011 Jan;79(2):218-27.
- Charytan DM,Patrick AR, Liu J, et al. Trends in the use and outcomes of implantable cardioverter-defibrillators in patients undergoing dialysis in the United States. Am J Kidney Dis. 2011 Sep;58(3):409-17.
- Maisch B, Seferovic PM, Ristic AD, et al. Guidelines on the diagnosis and management of pericardial diseases executive summary: the task force on the diagnosis and management of pericardial diseases of the European Society of Cardiology. Eur Heart J. 2004;25:587-610.
- Alpert MA, Ravenscraft MD. Pericardial involvement in end-stage renal disease. Am J Med Sci. 2003;325:228-236.
- Stacy SR, Suarez-Cuervo C, Berger Z, et al. Role of troponin in patients with chronic kidney disease and suspected acute coronary syndrome: a systematic review. Ann Intern Med 2014; 161:502.
- Thygesen K, Alpert JS, Jaffe AS, et al. Third universal definition of myocardial infarction. J Am Coll Cardiol 2012; 60:1581.
- Newby LK, Jesse RL, Babb JD, et al. ACCF 2012 expert consensus document on practical clinical considerations in the interpretation of troponin elevations: a report of the American College of Cardiology Foundation task force on Clinical Expert Consensus Documents. J Am Coll Cardiol 2012; 60:2427.
- Korkmaz H,Saşak G, Celik A, et al. The comparison of cardiac biomarkers positivities in hemodialysis patients without acute coronary syndrome. Ren Fail. 2011;33(6):578-81.
- Sommerer C,Beimler J, Schwenger V, et al. Cardiac biomarkers and survival in haemodialysis patients. Eur J Clin Invest. 2007 May;37(5):350-6.
- Sommerer C,Giannitsis E, Schwenger V, et al. Cardiac biomarkers in haemodialysis patients: the prognostic value of amino-terminal pro-B-type natriuretic peptide and cardiac troponin T. Nephron Clin Pract. 2007;107(3):c77-81.
- Zoccali C,Mallamaci F, Tripepi G. Hypertension as a cardiovascular risk factor in end-stage renal failure. Curr Hypertens Rep. 2002 Oct;4(5):381-6.
- Leypoldt JK1,Cheung AK, Delmez JA, et al. Relationship between volume status and blood pressure during chronic hemodialysis. Kidney Int. 2002 Jan;61(1):266-75.
- B Charra, C Chazot. Volume control, blood pressure and cardiovascular function. Nephron Physiology 2003;93:94-101.
- Halle MP,Hertig A, Kengne AP, et al. Acute pulmonary oedema in chronic dialysis patients admitted into an intensive care unit. Nephrol Dial Transplant. 2012 Feb;27(2):603-7.
- McCarthy C, Behravesh S, Naidu S, Oklu R. Air embolism: practical tips for prevention and treatment. J Clin Med. 2016; 5(11):93.
- Blanc P, Boussuges A, Henriette K, et al. Iatrogenic cerebral air embolism: importance of an early hyperbaric oxygenation. Intensive Care Med. 2002; 28(5):559-563.
- Dudney TM, Elliott CG. Pulmonary embolism from amniotic fluid, fat, and air. Prog Cardiovasc Dis 1994;36:447.
- Orebaugh SL. Venous air embolism: clinical and experimental considerations. Crit Care Med 1992;20:1169.
- Yu AS, Levy E. Paradoxical cerebral air embolism from a hemodialysis catheter. Am J Kidney Dis 1997;29:453.
- Dagher GA, Harmouche E, Jabbour E, et al. Sepsis in hemodialysis patients. BMC Emergency Medicine 2015;15:30.
- James MT, Quan H, Tonelli M, et al. CKD and risk of hospitalization and death with pneumonia. Am J Kidney Dis 2009;54:24.
- Dalrymple LS, Katz R, Kestenbaum B, et al. The risk of infection-related hospitalization with decreased kidney function. Am J Kidney Dis 2012;59:356.
- Wu MY, Hsu YH, Su CL, et al. Risk of herpes zoster in CKD: a matched-cohort study based on administrative data. Am J Kidney Dis 2012; 60:548.
- Naqvi SB, Collins AJ. Infectious complications in chronic kidney disease. Adv Chronic Kidney Dis 2006; 13:199.
- Thongprayoon C, Cheungpasitporn W, Phatharacharukul P, et al. Chronic Kidney Disease and End Stage Renal Disease are Risk Factors for Poor Outcomes of Clostridium Difficile Infection: A Systematic Review and Meta-analysis. International journal of clinical practice. 2015;69(9):998-1006.
- Nassar GM,Ayus JC. Infectious complications of the hemodialysis access. Kidney Int. 2001 Jul;60(1):1-13.
- Duncan ND,Singh S, Cairns TD, et al. Tesio-Caths provide effective and safe long-term vascular access. Nephrol Dial Transplant. 2004 Nov;19(11):2816-22.
- Fluck R,Wilson J, Tomson CR. UK Renal Registry 12th Annual Report (December 2009): chapter 12: epidemiology of methicillin resistant Staphylococcus aureus bacteraemia amongst patients receiving dialysis for established renal failure in England in 2008: a joint report from the UK Renal Registry and the Health Protection Agency. Nephron Clin Pract. 2010;115 Suppl 1:c261-70.
- Lai C-F, Liao C-H, Pai M-F, et al. Nasal Carriage of Methicillin-resistant Staphylococcus aureusIs Associated with Higher All-Cause Mortality in Hemodialysis Patients. Clinical Journal of the American Society of Nephrology: CJASN. 2011;6(1):167-174.
- Allon M. Treatment Guidelines for Dialysis Catheter–Related Bacteremia: An Update. American journal of kidney diseases: the official journal of the National Kidney Foundation. 2009;54(1):13-17.
- Rhodes A, Evans LE, Alhazzani W, et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Critical Care Medicine. 2017;45(3):486-552.
- Ozuzan P, Fried J, Grotts J. Early fluid resuscitation of end stage renal disease patients with severe sepsis and septic shock. Critical Care Medicine. 2014;42(12):pA1582.
- Dhingra R, Young E, Hulbert-Shearon T, et al. Type of vascular access and mortality in US hemodialysis patients. Kidney Int. 2001; 60(4):1443-1451.
- KDOQI Clinical Practice Guidelines and Clinical Practice Recommendations for 2006 Updates: Hemodialysis Adequacy, Peritoneal Dialysis Adequacy and Vascular Access. Am J Kidney Dis. 2004; 48:S1-S322.
- Ball L. Fatal vascular access hemorrhage: reducing the odds. Nephrol Nurs J. 2013; 40(4):297-303.
- Mannucci P, Remuzzi G, Pusineri F, et al. Deamino-8-D-arginine vasopressin shortens the bleeding time in uremia. N. Engl. J. Med. 1983;308:8-12.
- Linthorst GE, Avis HJ, Levi M. Uremic thrombocytopathy is not about urea. J Am Soc Nephrol. 2010;21:753.
- Galbusera M, Remuzzi G, Boccardo P. Treatment of bleeding in dialysis patients. Semin Dial. 2009;22:279-286.
- Gordz S, Mrowietz C, Pindur G, et al. Effect of desmopressin (DDAVP) on platelet membrane glycoprotein expression in patients with von Willebrand’s disease. Clin Hemorheol Microcirc. 2005;32:83-87.
- Zeigler ZR, Megaludis A, Fraley DS. Desmopressin (d-DAVP) effects on platelet rheology and von Willebrand factor activities in uremia. Am J 1992;39:90-95.
- Livio M, Gotti E, Marchesi D, et al. Uraemic bleeding: role of anaemia and beneficial effect of red cell transfusions. Lancet. 1982;2:1013-1015.
- Hedges SJ, Dehoney SB, Hooper JS, et al. Evidence-based treatment recommendations for uremic bleeding. Nat Clin Pract Nephrol. 2007;3:138.
- Pasklinsky G, Meisner R, Labropoulos N, et al. Management of true aneurysms of hemodialysis access fistulas. J Vasc Surg. 2011; 53(5):1291-1297.
- Mudoni A, Cornacchiari M, Gallieni M, et al. Aneurysms and pseudoaneurysms in dialysis access. Clin Kidney J. 2015; 8(4):363-367.
- Padberg F, Calligaro K, Sidawy A. Complications of arteriovenous hemodialysis access: recognition and management. J Vasc Surg. 2008; 48(5 Suppl):55S-80S.
- Siedlecki A, Barker J, Allon M. Aneurysm formation in arteriovenous grafts: associations and clinical significance. Seminars in Dialysis. 2007; 20(1):73-77.
- Saad TF. Bacteremia associated with tunneled, cuffed hemodialysis catheters. Am J Kidney Dis.1999 Dec;34(6):1114-24.
- Teitelbaum I, Burkart J. Peritoneal dialysis. Am J Kidney Dis 2003; 42:1082.
- Dell’Aquila R, Rodighiero MP, Spanó E, et al. Advances in the technology of automated, tidal, and continuous flow peritoneal dialysis. Perit Dial Int 2007; 27 Suppl 2:S130.
- Rabindranath KS, Adams J, Ali TZ, et al. Continuous ambulatory peritoneal dialysis versus automated peritoneal dialysis for end-stage renal disease. Cochrane Database Syst Rev 2007; :CD006515.
- Mujais S. Microbiology and outcomes of peritonitis in North America. Kidney Int Suppl.2006 Nov;(103):S55-62.
- Li PK,Szeto CC, Piraino B, Bernardini J, et al. Peritoneal dialysis-related infections recommendations: 2010 update. Perit Dial Int. 2010 Jul-Aug;30(4):393-423.
- Ballinger AE,Palmer SC, Wiggins KJ, et al. Treatment for peritoneal dialysis-associated peritonitis. Cochrane Database Syst Rev. 2014 Apr 26;(4):CD005284.
- Davenport A. Peritonitis remains the major clinical complication of peritoneal dialysis: the London, UK, peritonitis audit 2002-2003. Perit Dial Int.2009 May-Jun;29(3):297-302.
- Wiggins KJ, Johnson DW, Craig JC, et al. Treatment of peritoneal dialysis-associated peritonitis: a systematic review of randomized controlled trials. Am J Kidney Dis 2007; 50:967.
- Akoh JA. Peritoneal dialysis associated infections: An update on diagnosis and management. World Journal of Nephrology. 2012;1(4):106-122.
- Scalamogna A, Castelnovo C, De Vecchi A, et al. Exit-site and tunnel infections in continuous ambulatory peritoneal dialysis patients. Am J Kidney Dis 1991;18:674.







2 thoughts on “emDocs Cases: The Sick ESRD Patient”
Pingback: End Stage Renal Disease Case – Found Down
Pingback: emDOCs.net – Emergency Medicine EducationQuality Corner – The Bleeding Fistula and Neutropenic Fever - emDOCs.net - Emergency Medicine Education