Author: Joshua Bloom, MD, PhD (EM Resident Physician, Mt. Sinai St. Luke’s West) // Edited by: Anthony DeVivo, DO (@anthony_devivo, EM Chief Resident Physician, Mount Sinai St. Luke’s-West); Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit); and Manpreet Singh, MD (@MprizzleER)
Cases
- A 47-year old male found down in the street with unknown medical history is brought in by ambulance being actively resuscitated with chest compressions and bag-valve mask ventilation. As he is transferred to your resuscitation bay, you instruct your team to place him on the monitor and obtain intravascular access. You prepare to intubate.
- A 3-year old girl is currently being evaluated in your ED for high fevers and neck stiffness. While your team prepares to obtain labs and intravascular access, she rapidly decompensates and becomes obtunded.
- A 2-month male infant is brought to your emergency room by his mother who is in tears, screaming that her child drowned in the bathtub. A rapid evaluation reveals no pulse, no breathing, and no response to stimuli. Your team immediately begins resuscitative measures.
- A 16-year old female is brought in by EMS from a burning building that collapsed. She is unconscious but maintaining her airway. She has diffuse burns over 90% of her body, and both lower extremities were crushed by debris.
Background
Intravascular access is often necessary in emergency care. Not only does it allow rapid and bioavailable administration of drugs, fluids, and other therapies, but it also facilitates access to blood and serum for diagnostic tests. This access is usually obtained intravenously through peripheral veins, which are easily accessible in a majority of patients and can be cannulated rapidly by experienced personnel. However, there are some circumstances where peripheral IV access is not a rapid, viable option, such as in pediatric patients or patients with otherwise poorly visible veins1,2. In cases where peripheral IV access is not possible or can’t be done in a reasonable period of time (eg. rapidly during a resuscitation), intraosseous access is a fast, easy, and completely acceptable alternative3.
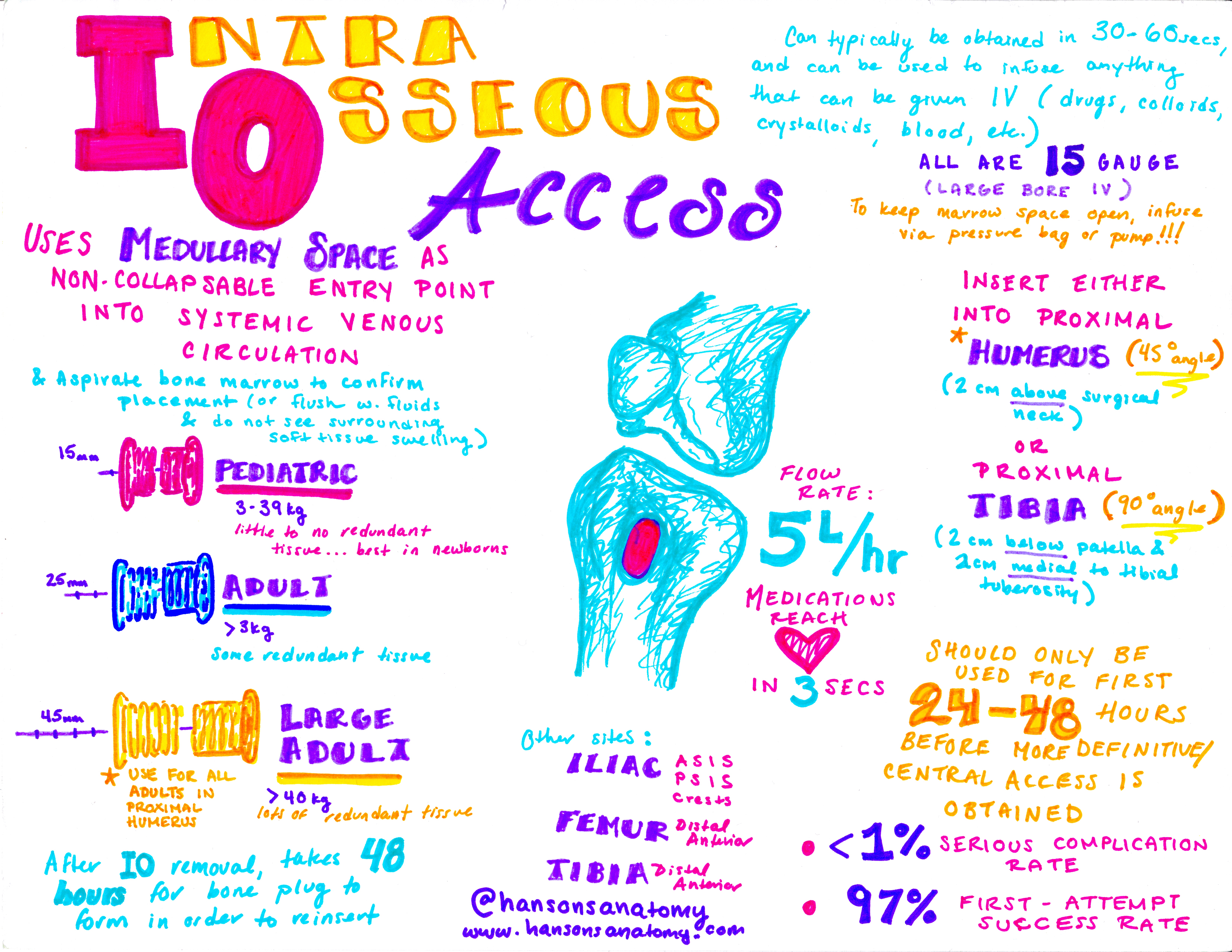
Intraosseous access uses the medullary cavity within bones as a non-collapsible vein. These cavities drain into venous channels that exit the bone into the systemic circulation, much like peripheral veins. Since the medullary cavity is contained in a rigid structure, it will not collapse in a dehydrated patient, and is amenable to administration of vasoactive drugs4-6. Also, since most of our patients have bones, IO access can be obtained in any patients on whom specific bony landmarks can be palpated. In addition, bone marrow aspirates from the cavity can be used for certain diagnostic tests, as will be discussed later.
This article will focus on up-to-date evidence-based indications, contraindications, methods, and tips for performing intraosseous access in the ED.
Indications
- Venous access: for infusion of intravenous fluids and drugs, including vasoactive drugs.
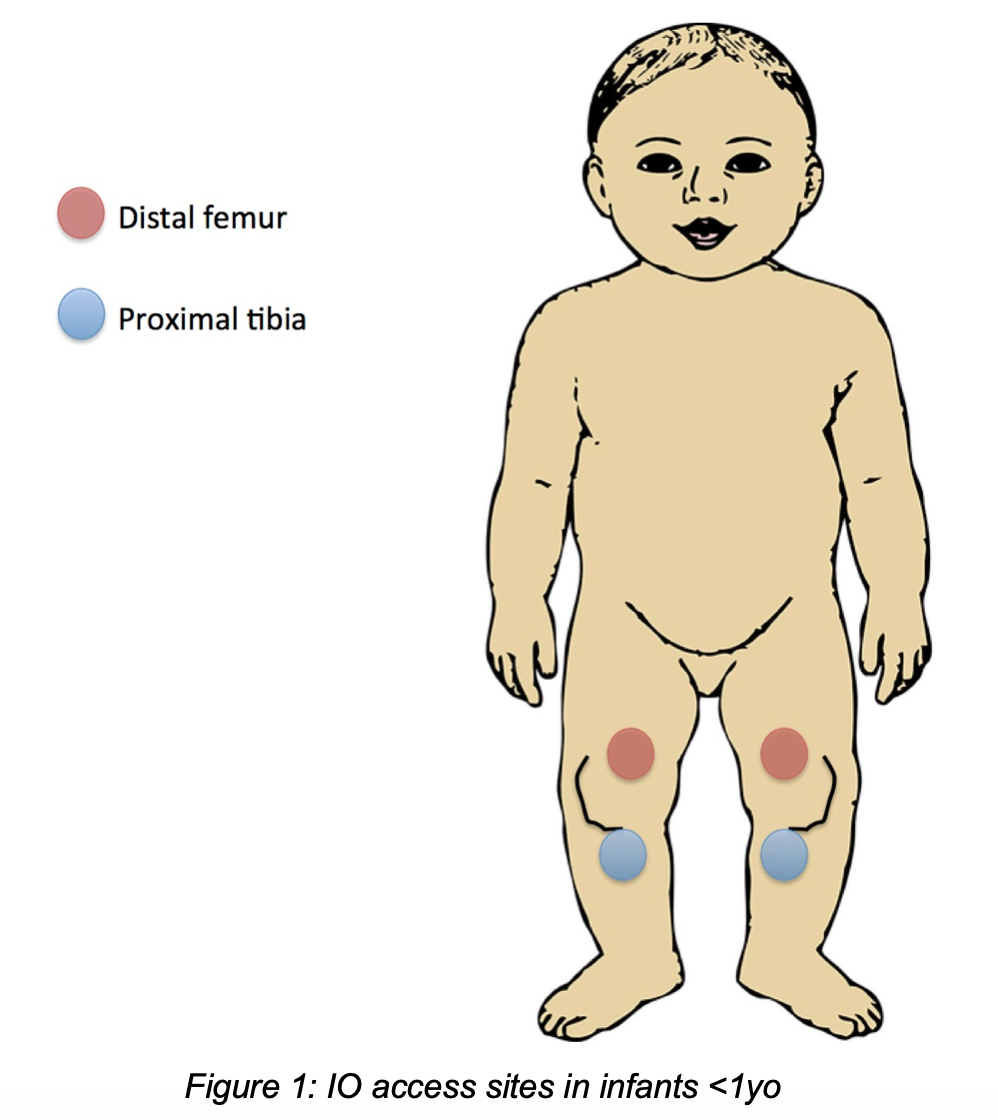
- Infants and children (<12y): in arrest or severe shock, if no easy IV access, place IO in distal femur (<1y), distal tibia/fibula (>1y), or proximal tibia (all) prior to central venous access or venous cutdown.
- Adolescents and adults (>12yo): in arrest, severe shock, or conditions where IV access is difficult or impossible, place IO in proximal humerus (>18yo), proximal tibia, distal tibia/fibula, or sternum (all) prior to central venous access or venous cutdown.
- Diagnostic bone marrow aspiration: frequently performed prior to infusion; rarely indicated in cases where infusion will not be performed.
- Bone marrow aspirates have been shown to provide accurate measures of hemoglobin, Na, Cl, BUN/creatinine, glucose, serum HCO3, pH, pCO2, and serum drug levels. They can also substitute blood for blood cultures and blood type/screen7,8.
- WBC, K, Ca, transaminase, and pO2 are not accurate7. Use in measuring lactic acid is not well established8.
- Aspirates obtained after infusions have been run through the IO line will not be accurate and should not be used.
- It can be difficult to withdraw bone marrow from an IO insertion site, and a lack of aspirate does not necessarily imply the IO is incorrectly placed, as discussed later7.
Contraindications
- Absolute contraindications for IO access:
- Fractured or injured bone at insertion site. Access here would be ineffective, as any administered fluid would infiltrate and not enter venous circulation.
- Vascular compromise proximal to the desired site. Same as above, fluid will infiltrate or otherwise not reach systemic circulation.
- Relative contraindications for IO access:
- Infection of the overlying soft tissue or bone (cellulitis, osteomyelitis).
- Osteogenesis imperfecta, osteopetrosis, or similar bone disorders.
- Presence of right-to-left intracardiac shunt (increased risk of hazardous fat/air/marrow emboli).
Considerations
- Any medications or fluids given IV can be given IO, including vasoactive drugs. A pressure bag may be used to overcome resistance from emissary veins and has been shown to improve infusion rates. A small industry-sponsored study in healthy volunteers demonstrated that a humeral IO can achieve pressure-bag infusion rates of roughly 5L/h (1L/8min)9.
- Intravenous contrast for CT scanning can be delivered via an IO and has been shown to be comparable to IV contrast in small studies and case reports10-12. Given some angiographic studies involve high-pressure contrast delivery, careful monitoring for infiltration is indicated13.
- Although there is evidence of rapid drug delivery from an IO comparable to central venous access, some drugs with a short intravenous half-life may be less effective by this route, and the evidence remains controversial. IO may therefore be nonideal for drugs such as adenosine14,15.
- Awake patients can receive an IO. However, infusion into bone marrow is exquisitely painful, and appropriate analgesia should be given prior to any infusion.
- Younger children and infants may not require battery or impact driven IO cannula insertion.
- Sternal IO access should only be performed with specialized devices to avoid intrathoracic injury.
- Recent prehospital studies have suggested that patients receiving IO access for out-of-hospital cardiac arrest may have worse neurologic outcomes and survival to hospitalization, with conflicting data regarding pre-hospital return of spontaneous circulation (ROSC)16-18. However, these data may have resulted from differences in patient populations more likely to receive an IO access (eg. patients with difficult vascular access).
The Procedure
Materials Needed
- Antiseptic site prep (chlorhexidine or iodine)
- Sterile gloves
- Sterile towels or drape
- IO catheter with stylet and insertion device (mechanical or manual)
- 2% lidocaine with subcutaneous needle and syringes (conscious patients only)
- IO Luer-lock tubing and pressure bag for infusions
- Gauze and tape or specialized dressing to secure device
- It should be noted that many IO insertions in the emergency setting will involve unconscious patients undergoing active resuscitation. In these cases, anesthesia is usually unnecessary, and a sterile insertion can be accomplished without sterile gloves or drapes.
General procedure19
- Position patient and locate the anatomy by palpation; if possible, mark your insertion site.
- Proximal tibia
- Children: 2cm below the tibial tuberosity, 1cm medial
- Adults: 2cm medial to tibial tuberosity, 1cm above.
- Proximal tibia

- Distal tibia/fibula (>1yo only): 1-2cm superior to the medial/lateral malleolus

- Proximal humerus (>18yo only): 2cm below the acromion process or 1cm above the surgical neck (can be palpated directly from below). Patient should be positioned with extremity adducted in internal rotation with hand over umbilicus to minimize exposure of the brachial plexus
- Sternum (>12yo only): upper 1/3 of the sternum (manubrium)
- Distal femur (<1yo only): 1-2cm superior to superior aspect of patella, angled medial or lateral to midline of patella
- Don mask, eye shield, sterile gown/gloves, and apply sterile drape/towels.
- Sterilize site with antiseptic prep solution.
- In awake patients, anesthetize the site with 2% lidocaine into the skin, subcutaneous tissue, and periosteum.
- Brace the extremity or site with non-dominant hand.
- To avoid needlestick injury, do NOT place your hand immediately behind the insertion site.
- Orient your device with needle/catheter perpendicular to the bone surface.
- In skeletally immature patients, angle your needle/catheter slightly away from the joint to spare the growth plate.
- Insert the IO catheter.
- Manual device: apply pressure while twisting back and forth until a decrease in resistance is felt; avoid rocking motions; note significant pressure may be needed in skeletally mature patients.
- Spring or impact driven device: follow device instructions; usually the device is oriented perpendicularly or as appropriate followed by activation of the spring with gentle pressure.
- Battery-driven device: apply manual pressure briefly to penetrate subcutaneous tissue and then press and hold the trigger; the IO will twist and penetrate bone, and the trigger should be released when a decrease in resistance is felt.
- Remove the needle stylet/trocar.
- Confirm placement by physical exam (look for an upright, immobile catheter), and attempt aspiration to aid confirmation and obtain bone marrow for diagnostic tests.
- In awake patients, infuse up to 2% lidocaine (in adults) slowly into the IO catheter for further analgesia; a further 1mL can be given after infusion is begun. Awake children receive smaller doses based on age20.
- Flush with 10mL normal saline and secure with gauze/tape or specialized dressing; IV tubing can now be attached.
- A properly placed IO should stand upright on its own and flush without local infiltration. Although aspiration can be useful in confirming placement, it can be unreliable, and even a properly placed IO catheter may yield no aspirate. Bedside ultrasound with color doppler has been used to confirm IO placement with reasonable accuracy compared to physical exam21,22.
- An IO catheter can be removed by attaching a 10mL Luer-lock syringe and pulling the catheter out with a twisting motion. The site should be cleaned, dressed with sterile gauze, and monitored afterward for signs of infection.
Post-Procedural Complications
- Fracture
- Fractures are a rare complication of IO placement, seen more often in multiple IO attempts on the same bone or patients with osteopathies23,24. Remember, a fractured bone becomes unusable for IO access since infused fluid can easily infiltrate.
- Infiltration and compartment syndrome
- Intraosseous catheters can infiltrate, causing leakage of infusion fluids and drugs with potentially devastating effects. Amputation and limb ischemia have been reported in case studies22,25. As with any vascular access, an IO must be monitored routinely for infiltration.
- Infection
- The development of deep-space infections or osteomyelitis can occur post-IO placement, especially with catheters remaining in place >24 hours26. An IO is a temporary access only. This is especially important when working in an ED receiving a partially resuscitated patient: a provider performing the secondary survey must be familiar with usual IO access sites that may have an indwelling catheter not mentioned in patient handoff. Any indwelling IO no longer needed for vascular access should be removed as soon as possible.
- Bleeding
- As with any invasive procedure, bleeding or soft tissue damage can occur with placement of an IO. Common IO access sites are usually not highly vascular; however, everyone’s anatomy is different, and the proceduralist should be prepared to apply pressure and otherwise treat any complex bleeding or severe tissue damage after IO insertion.
- Intrathoracic injury
- This is a consideration when using a sternal IO. This has been documented most often when long catheters not designed for sternal access were used, especially with impact-driven devices. Specialized sternal catheters exist that are designed to access the manubrium only.
- Fat embolism
- The development of fat or marrow emboli after IO placement is a theoretical risk. Animal models have demonstrated nearly 100% prevalence of fat emboli after IO placement, however the clinical significance of this is unclear27. Acute decompensation or organ ischemia should raise suspicion for this rare event.
- Needle in situ
- Unlike flexible catheters, the IO catheter is rigid and can be deformed by unnecessary force or incorrect technique during placement28. This may impair the function of the catheter, deposit foreign bodies into tissue, or make catheter removal more difficult.
Case Query: Do they need an IO?
- This adult patient undergoing active CPR with intubation in process definitely needs an IO. The IO can be placed quickly in the proximal tibia without interfering with compressions while peripheral/central venous access is obtained by other team members.
- This young female is likely septic and requires rapid access for fluid and medication infusions. Proximal tibia IO access can be obtained simultaneous with efforts at peripheral/central venous access. Remember, if bone marrow can be aspirated, it can be sent for essential diagnostic tests such as pH and blood cultures.
- Access in an infant can be challenging, especially during a resuscitation. Fortunately, this patient can have an IO placed in the distal femur or proximal tibia for immediate administration of fluids and resuscitative drugs.
- Burn patients can have challenging access and need rapid fluid resuscitation. An IO is absolutely indicated in this patient. However, this patient is challenging because the lower extremities have been compromised and the upper extremities are nonideal in a skeletally immature adolescent. A sternal IO can be placed on the upper manubrium with space allowed for chest compressions until a more definitive access can be obtained.
Pearls and Pitfalls
- Patients come in all shapes and sizes. If your patient has excessive subcutaneous or adipose tissue over your preferred site, you may have to choose an alternative site.
- Position for success! Make sure the extremity or site is visible, angled advantageously, and supported. Use bedrolls/sheets or fellow personnel to assist you with this.
- Avoid needlestick injuries! IO catheters and stylets/trocars are still sharps and can injure the proceduralist. Make sure your hand is not directly behind the insertion site, as a misplaced IO can bypass or completely penetrate the extremity and cause injury.
- It is very easy to forget to secure an IO during a busy resuscitation. Although they don’t slip out as easily as an IV, an IO can fall out, and if it does replacement likely needs to be in a different extremity to avoid infiltration. Tape down your IO securely!
- In a resuscitated patient, make sure that any IO access is clearly indicated and discussed with receiving providers during handoff. When receiving a resuscitated patient, examine carefully for the presence of an IO on the extremities or sternum.
- Anecdotally the proximal humerus is favored by many practitioners because of its theoretical faster infusion rates and utility in cases of abdominopelvic or lower extremity compromise9,29. The proximal tibia is frequently easier to palpate and available in patients of all ages. You should feel comfortable inserting an IO in at least two different sites and should be familiar with all of them.
Rapid Procedure Review
- Position patient and site of interest.
- Palpate landmarks to determine insertion point; mark the skin.
- Don sterile gown and gloves, apply sterile drape, and sterilize site with antiseptic.
- (AWAKE ONLY) Anesthetize insertion tract with 2% lidocaine.
- Insert IO until bone resistance decreases suddenly.
- Remove stylet/trocar.
- Examine for correct placement (upright position + flush), attempt to aspirate bone marrow.
- (AWAKE ONLY) Slowly infuse 2% lidocaine for anesthesia.
- Flush IO with saline; palpate for infiltration; connect IV tubing.
- Secure IO with gauze/tape or dressing.
From Dr. Katy Hanson at Hanson’s Anatomy:
References/Further Reading
- Jacobson AF, Winslow EH. Variables influencing intravenous catheter insertion difficulty and failure: an analysis of 339 intravenous catheter insertions. Heart Lung 2005;34(5):345–59.
- Sebbane M, Claret P-G, Lefebvre S, et al. Predicting peripheral venous access difficulty in the emergency department using body mass index and a clinical evaluation of venous accessibility. J Emerg Med 2013;44(2):299–305.
- Fiser DH. Intraosseous infusion. N Engl J Med 1990;322(22):1579–81.
- Hoskins SL, do Nascimento P, Lima RM, Espana-Tenorio JM, Kramer GC. Pharmacokinetics of intraosseous and central venous drug delivery during cardiopulmonary resuscitation. Resuscitation 2012;83(1):107–12.
- Hansen M, Meckler G, Spiro D, Newgard C. Intraosseous line use, complications, and outcomes among a population-based cohort of children presenting to California hospitals. Pediatr Emerg Care 2011;27(10):928–32.
- Burgert JM, Johnson AD, O’Sullivan JC, et al. Pharmacokinetic effects of endotracheal, intraosseous, and intravenous epinephrine in a swine model of traumatic cardiac arrest. Am J Emerg Med 2019;
- Grisham J, Hastings C. Bone marrow aspirate as an accessible and reliable source for critical laboratory studies. Annals of Emergency Medicine 1991;20(10):1121–4.
- Tallman CI, Darracq M, Young M. Analysis of intraosseous blood samples using an EPOC point of care analyzer during resuscitation. Am J Emerg Med 2017;35(3):499–501.
- Miller L, Philbeck T, Montez D, Puga T. 467: A Two-Phase Study of Fluid Administration Measurement During Intraosseous Infusion. YMEM 2010;56(S):S151.
- Ahrens KL, Reeder SB, Keevil JG, Tupesis JP. Successful computed tomography angiogram through tibial intraosseous access: a case report. J Emerg Med 2013;45(2):182–5.
- Budach NM, Niehues SM. CT angiography of the chest and abdomen in an emergency patient via humeral intraosseous access. Emergency Radiology 2017;:1–4.
- Schindler P, Helfen A, Wildgruber M, Heindel W, Schülke C, Masthoff M. Intraosseous contrast administration for emergency computed tomography: A case-control study. PLoS ONE 2019;14(5):e0217629–11.
- Winkler M, Issa M, Lowry C, Chornenkyy Y, Sorrell V. Intraarticular extravasation, an unusual complication of computed tomographic angiography performed with intraosseous needle intravenous access. Cardiovasc Diagn Ther 2018;8(4):516–9.
- Helleman K, Kirpalani A, Lim R. A Novel Method of Intraosseous Infusion of Adenosine for the Treatment of Supraventricular Tachycardia in an Infant. Pediatr Emerg Care 2017;33(1):47–8.
- Goodman IS, Lu CJ. Intraosseous infusion is unreliable for adenosine delivery in the treatment of supraventricular tachycardia. Pediatr Emerg Care 2012;28(1):47–8.
- Clemency B, Tanaka K, May P, et al. Intravenous vs. intraosseous access and return of spontaneous circulation during out of hospital cardiac arrest. Am J Emerg Med 2017;35(2):222–6.
- Feinstein BA, Stubbs BA, Rea T, Kudenchuk PJ. Intraosseous compared to intravenous drug resuscitation in out-of-hospital cardiac arrest. Resuscitation 2017;117:91–6.
- Kawano T, Grunau B, Scheuermeyer FX, et al. Intraosseous Vascular Access Is Associated With Lower Survival and Neurologic Recovery Among Patients With Out-of-Hospital Cardiac Arrest. Annals of Emergency Medicine 2018;71(5):588–96.
- Dev SP, Stefan RA, Saun T, Lee S. Insertion of an Intraosseous Needle in Adults. N Engl J Med 2014;370(24):e35–5.
- Hixson R. Intraosseous Administration of Preservative-Free Lidocaine. www.arrowezio.com.
- Saul T, Siadecki SD, Berkowitz R, Rose G, Matilsky D. The accuracy of sonographic confirmation of intraosseous line placement vs physical examination and syringe aspiration. Am J Emerg Med 2015;33(4):586–8.
- Greenstein YY, Koenig SJ, Mayo PH, Narasimhan M. A Serious Adult Intraosseous Catheter Complication and Review of the Literature. Crit Care Med 2016;44(9):e904–9.
- La Fleche FR, Slepin MJ, emergency JVAO, 1989. Iatrogenic bilateral tibial fractures after intraosseous infusion attempts in a 3-month-old infant. Annals of Emergency Medicine 1989;18(10):1099–101.
- Nutbeam T, Fergusson A. Intraosseous access in osteogenesis imperfecta (IO in OI). Resuscitation 2009;80(12):1442–3.
- Taylor CC, Clarke NMP. Amputation and intraosseous access in infants. BMJ 2011;342(may27 1):d2778–8.
- Rosetti VA, Thompson BM, Miller J, Mateer JR, Aprahamian C. Intraosseous infusion: an alternative route of pediatric intravascular access. Annals of Emergency Medicine 1985;14(9):885–8.
- Orlowski JP, Julius CJ, Petras RE, Porembka DT, Gallagher JM. The safety of intraosseous infusions: risks of fat and bone marrow emboli to the lungs. Annals of Emergency Medicine 1989;18(10):1062–7.
- Krishnan M, Lester K, Johnson A, Bardeloza K, Edemekong P, Berim I. Bent Metal in a Bone: A Rare Complication of an Emergent Procedure or a Deficiency in Skill Set? Case Reports in Critical Care 2016;2016(1):1–5.
- Pasley J, Miller CHT, DuBose JJ, et al. Intraosseous infusion rates under high pressure. Journal of Trauma and Acute Care Surgery 2015;78(2):295–9.






