Authors: Steven Chapman, DO, MPH (EM Resident Physician at the University of Kentucky); Diana Labrada, MD (EM Attending Physician at the University of Kentucky) // Reviewed by: Jessica Pelletier, DO (EM Education Fellow at Washington University School of Medicine in St. Louis); Marina Boushra, MD (EM-CCM Attending at the Cleveland Clinic Foundation); Brit Long, MD (@long_brit)
Case
A 42-year-old male with a history of intravenous (IV) drug use, diabetes, and hypertension presents to the emergency department (ED) with a complaint of lower back pain. He reports that the pain has been worsening for one week, is located along the lower back, and radiates into the right leg. The patient currently resides at a group living facility where he tested positive for COVID today. He notes subjective fever but denies nausea, vomiting, chills, and cough. What are the next steps? Is an emergent magnetic resonance imaging (MRI) study indicated in this patient?
Introduction
As technologies evolve and imaging becomes easier to obtain, MRI use in the ED is on the rise. It is often thought that this testing modality has been underutilized due to limited availability.1 Recent data suggest that the United States has the highest utilization of MRI scans in the world and has the second highest number of MRI scanners per capita, after Japan.1 With expected continued improvement in MRI technology, it is essential to understand when the usage of MRI within the ED is appropriate.
Brain MRI
Stroke/TIA
When choosing an imaging modality in the setting of a suspected transient ischemic attack (TIA) or an ischemic stroke, the old adage “time is brain” should determine which imaging modality is selected. The mean sensitivity of MRI for detecting an acute ischemic injury is 93% while computed tomography (CT) sensitivity within 12 hours of onset is 73-87%.2,3 Non-contrast head CT (NCCT) has a sensitivity of 12% for ischemic strokes less than three hours old.4 CTA has a sensitivity of 83.2% and specificity of 95% in evaluating acute ischemic stroke.4 Regarding acute ischemic stroke, CT perfusion (CTP) has a sensitivity of 82% and specificity of 96% with no significant difference between patients who received CTP within six hours of symptom onset compared with those who received the study after six hours of onset.5 While the sensitivity of MRI for stroke is higher than that of CTP, the wider availability and rapidity of obtaining CTP make it an invaluable tool in stroke workup. When deciding between these imaging modalities in a time-sensitive scenario, multiple factors should guide decision-making, including ease of access and the turnaround time for imaging results. In most cases, the MRI for a stroke patient is likely to be completed on an inpatient basis and should not delay ED management and intervention.
Cerebral Venous Sinus Thrombosis (CVST)
CVST occurs with a thrombus obstructing blood flow of cerebral veins and/or the dural sinus. This lack of blood flow can lead to cerebral infarction.6 Dural sinus occlusion can result in a decrease in cerebrospinal fluid absorption and hence increased intracranial pressure.6 When there is a concern for CVST, combined utilization of MRI and magnetic resonance venography (MRV) is the preferred method of assessment, which has replaced more invasive endovascular arteriography and venous digital subtraction angiography.7 While MRV is the imaging modality of choice, it is not universally available.8 CT venography (CTV) is more readily available and produces comparable results with a sensitivity of 95-100%; however, it should be noted that the data are based on studies with small sample sizes.9 The sensitivity for MRV with T2-weighted imaging is 90%.10 The choice of imaging modality for CVST will likely come down to the availability of both diagnostic equipment and time.
Spine MRI
Non-Infectious Spinal Cord Compression
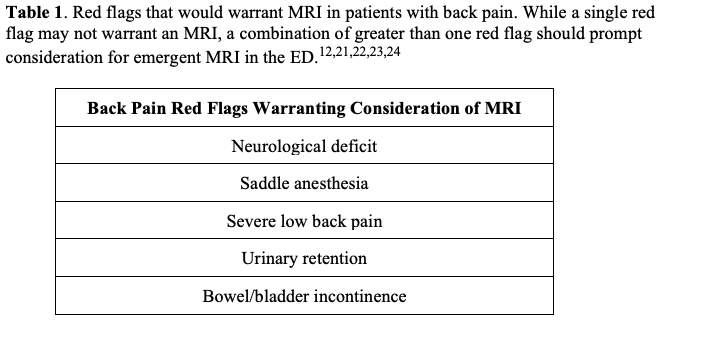
Spinal cord compression can present in various ways, ranging from traumatic injuries with bone fragment compression to compression by tumors or hematomas. When there is a concern for neoplastic compression of the spine, MRI may identify spinal metastasis in patients for whom radiographs, CT scans, and bone scans have been unrevealing.11 Symptoms that can indicate the need for MRI include acute-onset back pain or acute worsening of chronic back pain combined with “red flags,” including extremity weakness, Lhermitte sign, hyperreflexia, and bowel or bladder incontinence (Table 1).12 The gold standard imaging for spondylotic myelopathy and metastatic spinal cord compression is gadolinium contrast-enhanced MRI, which has a sensitivity of 93% and specificity of 97%.12,13 MRI will also need to include contrast should an infectious process be considered as the source of compression (please see discussion of Epidural Abscess below).12
Spinal Trauma
NCCT is the initial study of choice in the setting of suspected bony injuries to the spinal column. Clinical decision rules such as Canadian C-spine Rule or the National Emergency X-Radiography Utilization Study (NEXUS) can help inform the decision to obtain imaging of the cervical spine.14,15 If imaging in the setting of trauma is clinically indicated, NCCT is the first-line modality, as it is highly sensitive for identifying bony injury.14,15 CT of the cervical spine has a sensitivity of 90.9% and a specificity of 100%; there is an overall sensitivity of 98% and specificity of 97% when applying CT to the thoracolumbar spine.16,17 MRI is less sensitive and specific for bony injury in acute trauma, with a sensitivity of 76% and specificity of 95% for acute fracture lines. The time-consuming nature of MRI likewise precludes its use in acute traumatic settings.
The utility of MRI in spinal trauma is in the evaluation of soft tissue injury that is not well assessed with CT imaging (Table 2). MRI can be useful to visualize soft tissue changes such as ligamentous injury, epidural hematomas, and disc protrusions.11,20 In evaluating for soft tissue injuries, MRI with contrast has a sensitivity of 100% compared to 33% sensitivity for non-contrast MRI.18 This is significantly more sensitive than CT, which has a sensitivity of 32% and a specificity of 100% for ligamentous injuries of the cervical spine. 19 MRI is also a valuable tool for patients with neurological deficits that are not explained by plain film or CT findings. Spinal cord injury without radiographic abnormality (SCIWORA) refers to patients with clinical signs (i.e. pain, numbness, paresthesia) of posttraumatic spinal cord injury without radiographic evidence of fracture or misalignment on x-ray or CT.25 SCIWORA is more common in pediatric patients due to ligamentous laxity but is also seen in adults.25 Cervical spinal involvement is most common, but the thoracolumbar spine may also be affected.25 In patients in which there is a concern for traumatic spinal injury who have negative x-ray and CT imaging, MRI is a valuable tool for further evaluation.25

Cauda Equina Syndrome (CES)
CES occurs due to sacral and lumbar nerve root compression within the lower vertebral canal, leading to ischemia or infarction of nervous tissue. This condition commonly presents with bowel or bladder dysfunction and saddle anesthesia.27 CES is a clinical diagnosis; however, emergent MRI with contrast (the diagnostic test of choice) should be pursued to assess for potential underlying causes of CES as this is a surgical emergency.28 If the clinical concern is high, evaluation by a spinal surgeon should not be delayed for imaging. Ultimately, if obtaining the MRI would result in a delay of care, it is prudent to discuss the treatment plan with neurosurgery while awaiting imaging. In assessing spinal stenosis, a potential cause of CES, MRI sensitivity is 81-97% and myelography sensitivity is 67-78%.29 If the patient is unable to undergo MRI (due to poor tolerance, implanted metallic devices, etc.), CT myelography is an alternative (see further discussion below).27 If there is concern that MRI or CT myelography will result in a delay of care, CT imaging may be considered (with the knowledge that this modality has limited sensitivity and specificity for identifying underlying causes of CES). CT has a sensitivity of 77% and a specificity of 75% for identifying disc herniation and a sensitivity of 87% and a specificity of 75% for spinal canal stenosis.30,31 While not the ideal test modality, an expedited CT image may provide the consulting neurosurgeon with vital information while awaiting a more definitive modality.
CT myelography is typically performed by a radiologist and involves the injection of contrast material into the spinal canal during CT imaging.32 This will likely be done as an inpatient study as it is an examination requiring coordination between specialized personnel and fluoroscopy. Again, if imaging will result in a delay of care, evaluation by a spinal surgeon should be obtained. As MRI is becoming more available and is less invasive, CT myelography is not often ordered in the ED, it is prudent to briefly discuss the contraindications of the exam, including coagulopathies, increased intracranial pressure, sepsis, contrast allergy, history of seizures, skin infection overlying the necessary puncture site, and pregnancy. In addition, medications known to decrease the seizure threshold such as tricyclic antidepressants (TCAs), monoamine oxidase inhibitors (MAOIs), and selective serotonin reuptake inhibitors (SSRIs) should be held for 24-72 hours prior to myelography.33
Epidural Abscess
An infection within the epidural space is a life-threatening infection as it is able to spread to surrounding tissues and progress to sepsis; approximately 5% of patients with spinal epidural abscesses die from sepsis.34 Patients with a history of IV drug use, diabetes, alcoholism, and end-stage renal disease are at increased risk of developing a spinal epidural abscess.35 Often a secondary complication of hematogenous spread via septic thrombosis of epidural veins, epidural abscess requires rapid diagnosis, as delay in treatment can result in neurologic deficits and death.36 The gold standard for diagnosis of a spinal epidural abscess is MRI with gadolinium contrast, which has a sensitivity and specificity of 90%.37 CT with contrast has a low sensitivity of 6% in the identification of epidural abscess.38 While MRI is pending, a contrasted CT can be obtained to attempt visualization of the abscess and begin treatment. CT myelography has a sensitivity of 90% but is invasive and should be considered when MRI is unavailable, delayed, or contraindicated, such as in patients with MRI-incompatible implants.39 While CT myelography would be more definitive than CT with contrast, it is more time-consuming and, as discussed, has many contraindications.
Bone MRI
Osteomyelitis
Patients with a history of bacteremia, IV drug use, open fractures, endocarditis, and risk factors for poor wound healing are at an increased risk of developing osteomyelitis.40 When these patients present with back pain and associated signs and symptoms of acute infection such as fever, chills, local signs of inflammation, and elevated inflammatory markers, a high degree of suspicion should be present for osteomyelitis.41 Laboratory workup may be nonspecific for osteomyelitis, making radiographic imaging key to diagnosis. MRI has the highest sensitivity of the imaging modalities, with a sensitivity of 90% and specificity of 80%.42 For patients unable to undergo MRI, bone scan is an alternative imaging modality that could be done in an inpatient setting. Abnormalities in bone scans reflect the rate of new bone formation, but this is not specific to infection.43 Bone scintigraphy has a sensitivity of 100% and a specificity of up to 100%; however the specificity is decreased in the setting of prior bone trauma, orthopedic hardware, or underlying bone disease.43 X-ray imaging has a low sensitivity and specificity of 43-75% and 75-83%, respectively.44,45 As many as 80% of patients with osteomyelitis have normal radiographs in the first two weeks after onset of infection.44,45 The sensitivity and specificity of CT with contrast for osteomyelitis are 67% and 50%, respectively.45
MRI Limitations
While MRI is more costly than CT, a recent study found that the use of rapid MRI protocols for acute ischemic stroke within the ED resulted in an 18.7% reduction in total direct cost and 17% reduction in length of stay within the hospital.46 Logistic considerations to weigh when determining whether to obtain an MRI in the ED include the time involved to obtain the study and the fact that MRI may require the patient to leave the department, which may not be advisable in unstable patients and may stress department staffing limitations.
Patient ability to tolerate the study must also be considered. Claustrophobia can increase patient anxiety, making imaging more difficult.47 Patients may require sedation, analgesia, anxiolysis, or even anesthesia while undergoing MRI.47 This is often not practical in the ED setting. Patient body habitus is another consideration, as the MRI opening bore size typically ranges from 60-80 cm wide with maximum patient weight for the MRI table typically ranging from 250-300 kg.48
Implants such as artificial joints, insulin pumps, or neurostimulators may preclude the use of MRI. Of note, implants manufactured within the last three decades are made with non-ferromagnetic material and are typically labeled “MRI-safe” or “MRI-conditional.”49 This means that specific exam conditions must be met for the imaging to be obtained, most often due to the presence of a weakly ferromagnetic implant component.49 These scenarios require coordination with the radiology department, consultants, and manufacturers.49 Despite MRI-safe or MRI-conditional labels, heating of the metal during MRI can still cause injuries, including burns and pain.50 In patients with an altered level of consciousness or who are unresponsive, it is advised to obtain a plain radiograph to exclude the presence of metallic foreign bodies, implants, or other devices prior to MRI.47
Current practice guidelines for non-contrast MRI recommend no special considerations during pregnancy as no current literature has demonstrated negative effects on fetal development. It is recommended, however, that MRI only be performed in pregnancy in the following situations: the information needed from the MRI cannot be gained by ultrasound, the study results can affect care of the patient or fetus during pregnancy, and the referring physician does not feel it is appropriate to wait until after pregnancy to perform the study.50 MRI with gadolinium contrast during pregnancy has been associated with increased risk of rheumatological, inflammatory, or infiltrative skin conditions in the neonate, as well as stillbirth and neonatal death.51 In comparison, CT imaging during pregnancy results in an increased risk of cancer for the fetus due to the teratogenic effects of ionizing radiation.52 Use of these modalities in pregnancy requires a discussion of the risks and benefits with the patient, which can be aided by obstetric consultants. Imaging in pregnant patients with suspected life-threatening pathology should not be delayed or withheld due to their pregnancy status.
MRI is the most sensitive testing modality for the identification of metallic foreign bodies; however, depending on the material, the MRI runs the risk of heating and dislodging the material during the scan.53 MRI should not be performed if there is a known or suspected metallic foreign body, especially if the metallic composition is unknown; this is especially true if the foreign body is close to critical structures including the eyes, brain, spinal cord, nerves, or large vessels.54 If the risk of heating and migration during the scan is considered to be low, the clinical benefit may outweigh the risk.55 In the specific evaluation of a foreign body, MRI should not be the initial exam.54 Conventional x-ray imaging is widely available and provides high sensitivity, depicting up to 80% of all foreign bodies and up to 98% of radiopaque bodies, making the x-ray an excellent first-line evaluation.54 In the event of negative x-rays and high clinical suspicion for foreign body, CT or ultrasound should be considered. CT is 15 times more sensitive than x-ray and is the best modality for visualizing plastic, glass, and stone; however, CT is less sensitive than ultrasound in evaluating small superficial foreign bodies.54 Ultrasonography has an overall sensitivity of 72% and specificity of 92% for foreign bodies and is the best modality for visualization of wooden foreign bodies, with a sensitivity of 90% and specificity of 96.7%.54
Pearls and Pitfalls
- The use of MRI in the ED is evolving, but current practice should focus on when MRI will provide results that faster and more accessible modalities cannot obtain.
- CTA, CT with CTA, CTP, and MRI with MRA are available options for brain imaging in stroke patients. However, given the time involved in obtaining MRI, CT modalities are likely better suited for acute evaluation at this time.
- The current preferred method of assessment of CVST is the combined utilization of MRI and MRV; however, given the lack of universal availability, CTV may be utilized with comparable results.
- MRI of the spine with contrast is indicated for suspected spinal cord compression, trauma, infection, or CES.
- In regard to imaging modalities, MRI has the highest sensitivity and specificity when there is a concern for osteomyelitis.
- Close coordination between the ED physician, radiology staff, and manufacturers should occur in cases involving foreign bodies, implants, and medical devices.
References
- Chung M, Dahabreh IJ, Hadar N, et al. Emerging MRI Technologies for Imaging Musculoskeletal Disorders Under Loading Stress [Internet]. Rockville (MD): Agency for Healthcare Research and Quality (US); 2011 Nov. (Comparative Effectiveness Technical Briefs, No. 7.) Available from: https://www.ncbi.nlm.nih.gov/books/NBK82287/
- Krieger DA, Dehkharghani S. Magnetic Resonance Imaging in Ischemic Stroke and Cerebral Venous Thrombosis. Top Magn Reson Imaging. 2015 Dec;24(6):331-52. doi: 10.1097/RMR.0000000000000067. PMID: 26636639.
- Lev MH. CT versus MR for acute stroke imaging: is the “obvious” choice necessarily the correct one? AJNR Am J Neuroradiol. 2003 Nov-Dec;24(10):1930-1. PMID: 14625210; PMCID: PMC8148937
- Seltzer J, Neill L. (2020, Jan 6). Emergency Guide to Stroke Neuroimaging. [NUEM Blog. Expert Commentary by Jahromi B]. Retrieved from http://www.nuemblog.com/blog/2018/4/20/stroke-neuroimaging
- Shen J, Li X, Li Y, Wu B. Comparative accuracy of CT perfusion in diagnosing acute ischemic stroke: A systematic review of 27 trials. PLoS One. 2017 May 17;12(5):e0176622. doi: 10.1371/journal.pone.0176622. PMID: 28520753; PMCID: PMC5435168.
- Stam J. Thrombosis of the cerebral veins and sinuses. N Engl J Med. 2005 Apr 28;352(17):1791-8. doi: 10.1056/NEJMra042354. PMID: 15858188.
- Quint DJ. Indications for emergent MRI of the central nervous system. JAMA. 2000 Feb 16;283(7):853-5. doi: 10.1001/jama.283.7.853. PMID: 10685696.
- Digge P, Prakashini K, Bharath KV. Plain CT vs MR venography in acute cerebral venous sinus thrombosis: Triumphant dark horse. Indian J Radiol Imaging. 2018 Jul-Sep;28(3):280-284. doi: 10.4103/ijri.IJRI_328_17. PMID: 30319203; PMCID: PMC6176664.
- Lisette F. van Dam, Marianne A.A. van Walderveen, Lucia J.M. Kroft, Nyika D. Kruyt, Marieke J.H. Wermer, Matthias J.P. van Osch, Menno V. Huisman, Frederikus A. Klok, Current imaging Modalities for diagnosing cerebral vein thrombosis – A critical review, Thrombosis Research, Volume 189, 2020, Pages 132-139,
- Idbaih A, Boukobza M, Crassard I, Porcher R, Bousser MG, Chabriat H. MRI of clot in cerebral venous thrombosis: high diagnostic value of susceptibility-weighted images. Stroke 2006;37(4):991–995.
- Arce D, Sass P, Abul-Khoudoud H. Recognizing spinal cord emergencies. Am Fam Physician. 2001 Aug 15;64(4):631-8. Erratum in: Am Fam Physician 2002 May 1;65(9):1751. PMID: 11529262.
- Singleton JM, Hefner M. Spinal Cord Compression. [Updated 2022 May 22]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK557604/
- Rammohan G, Meyers M, Marco C. Spinal Cord Compression: Evaluation and Management. Emergency Medicine Reports. Relias Media. 2021 Sept 1.
- Stiell IG, Clement CM, Grimshaw J, Brison RJ, Rowe BH, Schull MJ, Lee JS, Brehaut J, McKnight RD, Eisenhauer MA, Dreyer J, Letovsky E, Rutledge T, MacPhail I, Ross S, Shah A, Perry JJ, Holroyd BR, Ip U, Lesiuk H, Wells GA. Implementation of the Canadian C-Spine Rule: prospective 12 centre cluster randomised trial. BMJ. 2009 Oct 29;339:b4146. doi: 10.1136/bmj.b4146. PMID: 19875425; PMCID: PMC2770593.
- Stiell IG, Clement CM, McKnight RD, Brison R, Schull MJ, Rowe BH, Worthington JR, Eisenhauer MA, Cass D, Greenberg G, MacPhail I, Dreyer J, Lee JS, Bandiera G, Reardon M, Holroyd B, Lesiuk H, Wells GA. The Canadian C-spine rule versus the NEXUS low-risk criteria in patients with trauma. N Engl J Med. 2003 Dec 25;349(26):2510-8. doi: 10.1056/NEJMoa031375. PMID: 14695411.
- Resnick S, Inaba K, Karamanos E, et al. Clinical Relevance of Magnetic Resonance Imaging in Cervical Spine Clearance: A Prospective Study. JAMA Surg. 2014;149(9):934–939. doi:10.1001/jamasurg.2014.867
- Sixta S, Moore FO, Ditillo MF, Fox AD, Garcia AJ, Holena D, Joseph B, Tyrie L, Cotton B; Eastern Association for the Surgery of Trauma. Screening for thoracolumbar spinal injuries in blunt trauma: an Eastern Association for the Surgery of Trauma practice management guideline. J Trauma Acute Care Surg. 2012 Nov;73(5 Suppl 4):S326-32. doi: 10.1097/TA.0b013e31827559b8. PMID: 23114489.
- Ghasemi A, Haddadi K, Shad AA. Comparison of Diagnostic Accuracy of MRI with and Without Contrast in Diagnosis of Traumatic Spinal Cord Injuries. Medicine (Baltimore). 2015 Oct;94(43):e1942. doi: 10.1097/MD.0000000000001942. Erratum in: Medicine (Baltimore). 2015 Dec;94(48):1. PMID: 26512624; PMCID: PMC4985437.
- Diaz JJ Jr, Aulino JM, Collier B, Roman C, May AK, Miller RS, Guillamondegui O, Morris JA Jr. The early work-up for isolated ligamentous injury of the cervical spine: does computed tomography scan have a role? J Trauma. 2005 Oct;59(4):897-903; discussion 903-4. doi: 10.1097/01.ta.0000188012.84356.dc. PMID: 16374279.
- Cavallaro, M., D’Angelo, T., Albrecht, M.H. et al. Comprehensive comparison of dual-energy computed tomography and magnetic resonance imaging for the assessment of bone marrow edema and fracture lines in acute vertebral fractures. Eur Radiol 32, 561–571 (2022). https://doi.org/10.1007/s00330-021-08081-8
- Bennett J, M Das J, Emmady PD. Spinal Cord Injuries. [Updated 2022 May 11]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK560721/
- McMordie JH, Viswanathan VK, Gillis CC. Cervical Spine Fractures Overview. [Updated 2022 May 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK448129/
- Fernández-de Thomas RJ, De Jesus O. Thoracolumbar Spine Fracture. [Updated 2022 Aug 22]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK562204/
- Cowley P. Neuroimaging of Spinal Canal Stenosis. Magn Reson Imaging Clin N Am. 2016 Aug;24(3):523-39. doi: 10.1016/j.mric.2016.04.009. PMID: 27417399.
- Atesok K, Tanaka N, O’Brien A, Robinson Y, Pang D, Deinlein D, Manoharan SR, Pittman J, Theiss S. Posttraumatic Spinal Cord Injury without Radiographic Abnormality. Adv Orthop. 2018 Jan 4;2018:7060654. doi: 10.1155/2018/7060654. PMID: 29535875; PMCID: PMC5817293.
- Mohamed MA, Majeske KD, Sachwani-Daswani G, Coffey D, Elghawy KM, Pham A, Scholten D, Wilson KL, Mercer L, McCann ML. Impact of MRI on changing management of the cervical spine in blunt trauma patients with a ‘negative’ CT scan. Trauma Surg Acute Care Open. 2016 Oct 19;1(1):e000016. doi: 10.1136/tsaco-2016-000016. Erratum in: Trauma Surg Acute Care Open. 2016 Nov 9;1(1): PMID: 29766060; PMCID: PMC5891701.
- McNamee J, Flynn P, O’Leary S, Love M, Kelly B. Imaging in cauda equina syndrome–a pictorial review. Ulster Med J. 2013 May;82(2):100-8. PMID: 24082289; PMCID: PMC3756868.
- Lavy C, Marks P, Dangas K, Todd N. Cauda equina syndrome-a practical guide to definition and classification. Int Orthop. 2022 Feb;46(2):165-169. doi: 10.1007/s00264-021-05273-1. Epub 2021 Dec 4. PMID: 34862914; PMCID: PMC8782783.
- Rider LS, Marra EM. Cauda Equina And Conus Medullaris Syndromes. [Updated 2022 Aug 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK537200/
- Ma B, Wu H, Jia LS, Yuan W, Shi GD, Shi JG. Cauda equina syndrome: a review of clinical progress. Chin Med J (Engl). 2009 May 20;122(10):1214-22. PMID: 19493474.
- Bischoff RJ, Rodriguez RP, Gupta K, Righi A, Dalton JE, Whitecloud TS. A comparison of computed tomography-myelography, magnetic resonance imaging, and myelography in the diagnosis of herniated nucleus pulposus and spinal stenosis. J Spinal Disord. 1993 Aug;6(4):289-95. doi: 10.1097/00002517-199306040-00002. PMID: 8219542.
- Patel DM, Weinberg BD, Hoch MJCT. CT Myelography: Clinical Indications and Imaging Findings. RadioGraphics 2020;40(2):470-484.
- “ACR–ASNR PRACTICE GUIDELINE FOR THE PERFORMANCE OF MYELOGRAPHY AND CISTERNOGRAPHY.” American Society of Neuroradiology, https://www.asnr.org/wp-content/uploads/2016/12/Myelography.pdf.
- Ameer MA, Knorr TL, Munakomi S, et al. Spinal Epidural Abscess. [Updated 2022 Nov 17]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK441890/
- Babic M, Simpfendorfer CS, Berbari EF. Update on spinal epidural abscess. Curr Opin Infect Dis. 2019 Jun;32(3):265-271. doi: 10.1097/QCO.0000000000000544. PMID: 31021957.
- Lener S, Hartmann S, Barbagallo GMV, Certo F, Thomé C, Tschugg A. Management of spinal infection: a review of the literature. Acta Neurochir (Wien). 2018 Mar;160(3):487-496. doi: 10.1007/s00701-018-3467-2. Epub 2018 Jan 22. PMID: 29356895; PMCID: PMC5807463.
- Yang X, Guo R, Lv X, Lai Q, Xie B, Jiang X, Dai M, Zhang B. Challenges in diagnosis of spinal epidural abscess: A case report. Medicine (Baltimore). 2019 Feb;98(5):e14196. doi: 10.1097/MD.0000000000014196. PMID: 30702572; PMCID: PMC6380696.
- Rausch VH, Bannas P, Schoen G, Froelich A, Well L, Regier M, Adam G, Henes FO. Diagnostic Yield of Multidetector Computed Tomography in Patients with Acute Spondylodiscitis. Rofo. 2017 Apr;189(4):339-346. English. doi: 10.1055/s-0043-101864. Epub 2017 Mar 1. PMID: 28249310.
- Moore, Derek W. “Spinal Epidural Abscess.” Orthobullets, 24 June 2021, https://www.orthobullets.com/spine/2026/spinal-epidural-abscess.
- Momodu II, Savaliya V. Osteomyelitis. [Updated 2022 May 12]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK532250/
- Fritz JM, McDonald JR. Osteomyelitis: approach to diagnosis and treatment. Phys Sportsmed. 2008 Dec;36(1):nihpa116823. doi: 10.3810/psm.2008.12.11. PMID: 19652694; PMCID: PMC2696389.
- Gaillard F, Yap J, Rasuli B, et al. Osteomyelitis. Reference article, Radiopaedia.org. https://doi.org/10.53347/rID-7662
- Love C, Palestro CJ. Nuclear medicine imaging of bone infections. Clin Radiol. 2016 Jul;71(7):632-46. doi: 10.1016/j.crad.2016.01.003. Epub 2016 Feb 17. PMID: 26897336.
- Lee YJ, Sadigh S, Mankad K, Kapse N, Rajeswaran G. The imaging of osteomyelitis. Quant Imaging Med Surg. 2016 Apr;6(2):184-98. doi: 10.21037/qims.2016.04.01. PMID: 27190771; PMCID: PMC4858469.
- Pineda C, Espinosa R, Pena A. Radiographic imaging in osteomyelitis: the role of plain radiography, computed tomography, ultrasonography, magnetic resonance imaging, and scintigraphy. Semin Plast Surg. 2009 May;23(2):80-9. doi: 10.1055/s-0029-1214160. PMID: 20567730; PMCID: PMC2884903.
- de Havenon A, Orlando C, Delic A, et al. Direct cost analysis of rapid MRI in the emergency department evaluation of patients suspected of having acute ischemic stroke. The Neuroradiology Journal. 2022;0(0). doi:10.1177/19714009221108681
- Ruhani Doda Khera MD, MBA, Joshua A. Hirsch MD, Karen Buch MD and Sanjay Saini MD, MBA Magnetic Resonance Imaging Clinics of North America, 2022-08-01, Volume 30, Issue 3, Pages 553-563, Copyright @ 2022 Elsevier Inc.
- Nabasenja C, Barry K, Nelson T, Chandler A, Hewis J. Imaging individuals with obesity. J Med Imaging Radiat Sci. 2022 Jun;53(2):291-304. doi: 10.1016/j.jmir.2022.02.003. Epub 2022 Feb 25. PMID: 35227632.
- Poh, P.G., Liew, C., Yeo, C. et al. Cardiovascular implantable electronic devices: a review of the dangers and difficulties in MR scanning and attempts to improve safety. Insights Imaging 8, 405–418 (2017). https://doi.org/10.1007/s13244-017-0556-3
- Sammet S. Magnetic resonance safety. Abdom Radiol (NY). 2016 Mar;41(3):444-51. doi: 10.1007/s00261-016-0680-4. PMID: 26940331; PMCID: PMC4848040.
- Kallmes D.F., Watson R.E.: Gadolinium administration in undetected pregnancy: cause for alarm?. Radiology 2019; 293: pp. 201-202.
- Sadro Claudia T, MD, Dubinsky Theodore J. CT in Pregnancy: Risks and Benefits. FABR, FRCPC. Applied Radiology. 2013 Oct;42(10):6–16.
- Campbell EA, Wilbert CD. Foreign Body Imaging. [Updated 2022 May 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK470294/
- Carneiro BC, Cruz IAN, Chemin RN, Rizzetto TA, Guimarães JB, Silva FD, Junior CY, Pastore D, Ormond Filho AG, Nico MAC. Multimodality Imaging of Foreign Bodies: New Insights into Old Challenges. Radiographics. 2020 Nov-Dec;40(7):1965-1986. doi: 10.1148/rg.2020200061. PMID: 33136481.
- https://www.mriphysics.scot.nhs.uk/implant-safety-policies/metal-objects-and-foreign-bodies/





