Authors: Alex Stern, MD, (EM Resident Physician, LAC+USC Medical Center, @alextsternum) // Reviewed by Michael J. Yoo, MD (EM Attending Physician, San Antonio, TX); Alex Koyfman, MD (@EMHighAK); and Brit Long, MD (@long_brit)
Case:
A 44-year-old man with a history of hypertension, diabetes, and severe alcohol use complicated by pancreatitis, presents complaining of approximately three weeks of nausea, vomiting, and diffuse abdominal pain, all of which have worsened over the last five days. He describes the symptoms as much more severe and persistent than his prior episodes of pancreatitis.
His vital signs are HR 120, BP 100/60, RR 21, saturation 96% on RA, and a temperature of 38.8°C. He is in significant distress due to pain. Physical exam demonstrates severe abdominal tenderness with guarding, worst in the epigastrium.
After reading this article, learners should be able to:
- Recognize the signs and symptoms of severe acute pancreatitis
- Utilize evidence-based diagnostic tools (labs and imaging) to aid in the diagnosis of acute pancreatitis
- Justify a treatment plan for managing a patient with severe acute pancreatitis, including appropriate disposition
Background
Why should we care? Acute pancreatitis is a common emergency department (ED) diagnosis that often results in hospital admission, with an incidence of about 14 per 10,000 ED visits.1 However, the incidence is increasing, and the mortality from severe, necrotizing cases remains high.2 An estimated 20-30% of cases are severe, and those severe cases are associated with an in-hospital mortality rate of 15%, and as high as 50% in some studies.1,3 Initiating quality care from the ED maximizes the chances for good outcome as an inpatient.
What is it? Pancreatitis, or inflammation of the pancreas is characterized by abdominal pain, vomiting, and elevated pancreatic enzymes. The Atlanta criteria help clinicians classify pancreatitis as mild (no organ failure, no local/systemic complications), moderate (transient organ failure, exacerbation of comorbid disease), or severe (organ failure for > 48 h).4The Atlanta criteria also consider the presence of necrotizing pancreatitis: edematous or interstitial pancreatitis (without necrosis) is generally uncomplicated, while necrotizing pancreatitis carries a much higher risk of organ system failure and mortality.
What causes it? Pancreatitis has numerous etiologies. A commonly used acronym that may help learners remember some of these etiologies is GETSMASHED: gallstones, ethanol, trauma, steroids, mumps, autoimmunity, scorpion sting, hyperlipidemia (hypercalcemia, hyperparathyroidism), ERCP, and drugs. In the United States, gallstones and ethanol together cause most cases (approximately 45-55% and 25-35%, respectively).4 Of note, any of these causes can lead to severe, life-threatening disease.
Diagnostic Criteria (need 2 of 3):
- Signs and symptoms (epigastric pain, vomiting)4
- Lipase greater than three times the upper limit of normal4
- Imaging evidence of pancreatic inflammation (usually computed tomography4
Differential Diagnosis
Pancreatitis classically presents with the non-specific symptoms of abdominal pain (usually epigastric) and vomiting. Important considerations in patients with these symptoms include:
- Biliary disease, such as symptomatic cholelithiasis, cholecystitis, and ascending cholangitis5
- Gastric and small intestinal disease, such as gastritis or peptic ulcers, with or without perforation5
- Intra-abdominal infections, such as appendicitis5
- Bowel obstructions, with or without perforation5
- Viral infections such as enteritis5

Though certain historical or demographic factors may suggest pancreatitis (e.g., a 40-year-old alcoholic at the end of a week-long alcohol binge), all of these diagnoses need to be considered in the evaluation. If the patient is critically ill, presents in extremis, or has peritonitis, the differential narrows significantly. In this case, more emergent etiologies should be considered, to include perforated viscus, ruptured abdominal aortic aneurysm, bowel ischemia, and ectopic pregnancy.5 In these patients, resuscitation and early surgical consultation are indicated, while laboratory testing and imaging are obtained, if possible.
Role of Laboratory Studies
Labs play an important role in the diagnosis, treatment, prognostication, and monitoring of patients with severe pancreatitis. Laboratory markers that can aid in the diagnosis of patients with abdominal pain include lipase, white blood cell count, bilirubin level, and transaminases. Acute phase reactants such as the C-reactive protein (CRP) and procalcitonin, as well as serum triglycerides, are less commonly ordered in the ED setting, but can assist in the continued workup and management of patients who are admitted. A further discussion of these laboratory studies is below.
Lipase and Amylase
- These may become elevated several hours after the onset of symptoms.
- Amylase, which is generated in several tissues including the pancreas, is less specific (67-83%) than lipase (85-98%) and does not stay elevated in the serum as long as lipase does.6
- Most guidelines released by gastroenterological societies in recent years recommend lipase instead of amylase.8–10
- Lipase may be falsely elevated in alcoholics and those with renal dysfunction.7
- Current evidence recommends a lipase cutoff of three times the upper limit of normal. Levels beyond this have not been shown to be predictive of higher severity, except in children.11
White Blood Cell Count
- Nonspecific marker of inflammation and infection
- Level may be elevated due to third-spacing, fluid loss, generalized inflammation, or concomitant infection.
- Neutrophil-to-lymphocyte ratio (NLR) is a better predictor of pancreatitis severity than the absolute white blood cell count, with a value greater than 7.1 being 87% sensitive for severe disease.12 Thus, a complete blood cell count with differential can be helpful.
Liver Panel
- Elevated direct bilirubin should prompt investigation for biliary obstruction as a cause of pancreatitis.
- Elevated transaminases may also signify chronic alcohol use, concomitant liver inflammation, or underlying cirrhosis.
Acute Phase Reactants
- These are non-specific markers of inflammatory states.
- CRP can be a helpful marker for discriminating severe from mild acute pancreatitis; at 48 hours, 76% of pancreatitis patients with a CRP above 150 mg/dL had severe disease.13
- Procalcitonin is 80-90% specific and sensitive for severe disease if measured within 36 hours of symptom onset and elevated above 0.5 ng/mL.14
- Several other biomolecules have been studied (interleukins, macrophage-activating factors, serum amyloid, pentraxins, among many others) but have not proven to be useful in diagnosis or severity prediction.15
Triglycerides
- It may be reasonable to order this in the ED setting when a clear etiology of pancreatitis is unknown, or if the patient is obese, diabetic, or hyperlipidemic (especially due to its increasing incidence).
Role of Imaging
Computed tomography (CT) of the abdomen with contrast is the best modality to visualize pancreatic inflammation in the ED, but most cases of uncomplicated, mild pancreatitis do not need CT imaging.3 CT should be obtained, however, when there is suspicion for necrotizing pancreatitis, as imaging will guide surgical management and alter prognostication. However, the distinction between mild and severe pancreatitis, discussed above, is clinical. Pain out of proportion, failure to improve as expected, hemodynamic instability, an unclear diagnosis, and severe metabolic derangements are all factors which increase the utility of early imaging.
Findings on CT in severe cases may include abscess formation, gas formation suggestive of necrosis and superinfection, marked inflammation, or involvement of nearby structures.3 The lack of these features may be misleading, as necrosis and its associated radiographic changes are only seen after 3-4 days of symptoms.8 Therefore, CT may be repeated if the patient’s clinical status changes abruptly during admission.
Ultrasound (US) is another useful imaging modality that should be used as an adjunct in the management of acute pancreatitis. Ultrasound may reveal the cause of pancreatitis, as in gallstone pancreatitis, or suggest an alternative diagnosis, such as acute cholecystitis, which often presents with epigastric pain and vomiting. As with CT imaging, in patients with a clear etiology of pancreatitis or mild symptoms, ultrasound may not be necessary in the ED.
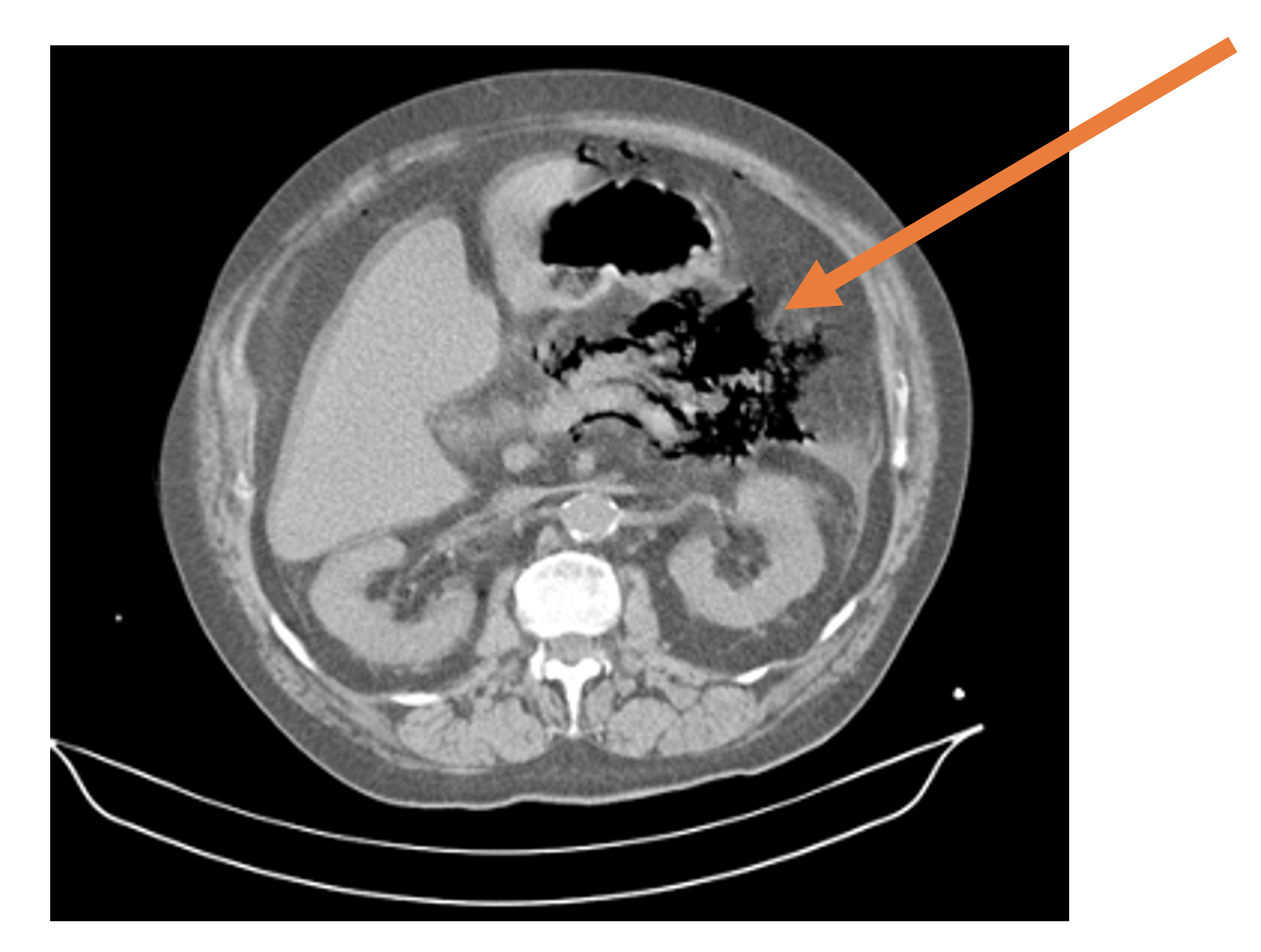
Figure 1. CT of the abdomen and pelvis demonstrating peripancreatic gas (orange arrow). Case courtesy of Dr Fabricio Machado, Radiopaedia.org, rID: 16029
Management
Severity Scores
Gauging the severity of the patient’s presentation is important in guiding management and disposition. There are two widely used sets of clinical criteria for making this assessment: Ranson’s criteria and the bedside index of severity in acute pancreatitis (BISAP).
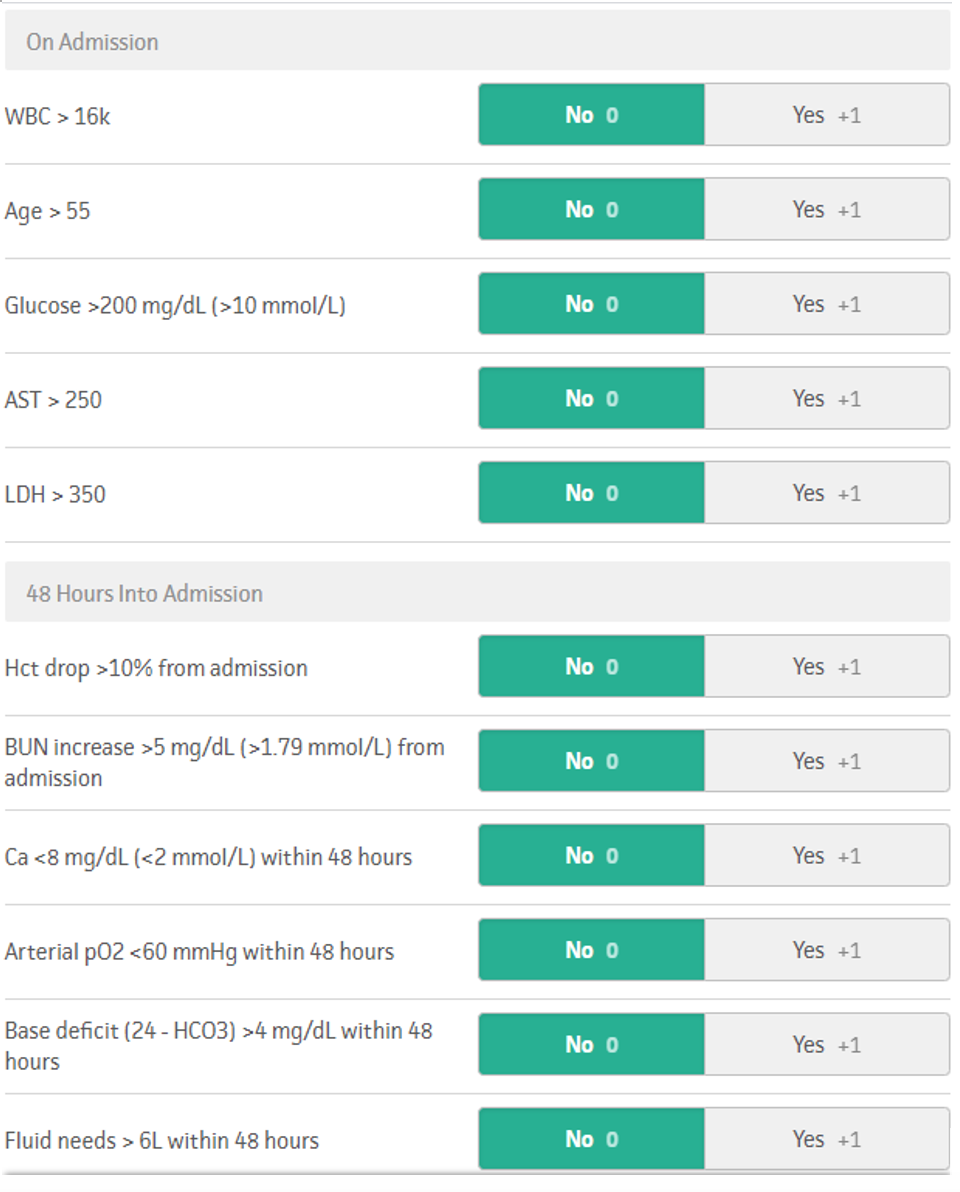
Ranson’s score was developed in the 1970s. It primarily utilizes laboratory markers to predict the severity of acute pancreatitis. Points are assigned in a binary fashion — either the marker is above (+1) or below (+0) a threshold value. A score of greater than or equal to three suggests severe pancreatitis with a predicted mortality of 15% at 3 points and 40% at 5 points. A total score of 7 predicts 100% mortality. Notably, the score must be calculated at admission and 48 hours after admission.16
Figure 2. Ranson’s score, adopted from MDCalc.com (https://www.mdcalc.com/ransons-criteria-pancreatitis-mortality).
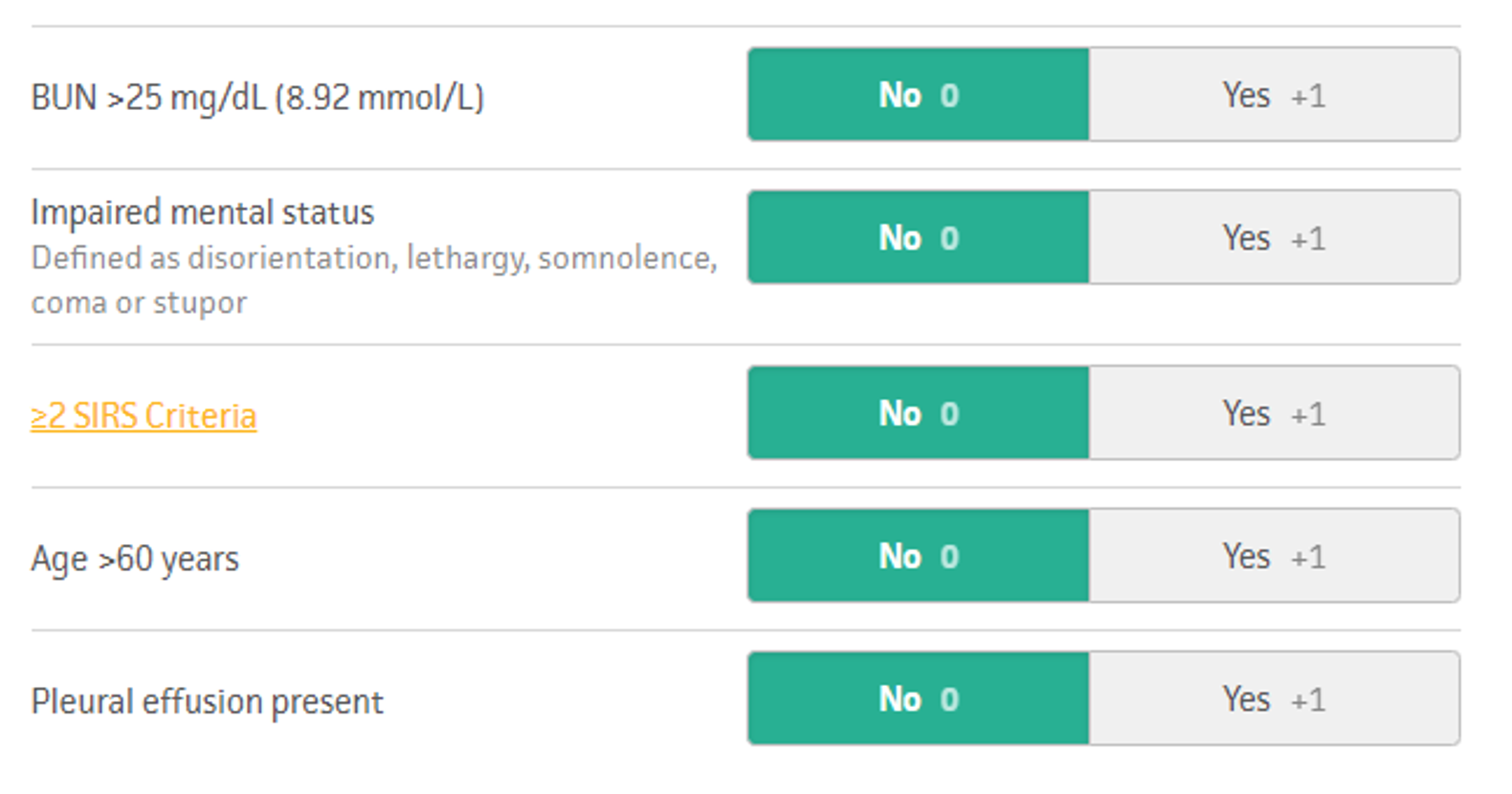
In 2008, the BISAP score was developed and validated for use in the prediction of severe acute pancreatitis.17 It utilizes both clinical data (such as altered mental status and the presence of a pleural effusion) in addition to laboratory markers. It performed similarly to Ranson’s criteria but is simpler and can be calculated at 24 hours after admission rather than at 48 hours. In the BISAP tool, a score of 0 is associated with <1% mortality, 2 with 1.9% mortality, and 5 with 22% mortality.17
Figure 3. BISAP score adopted from MDCalc.com (https://www.mdcalc.com/bisap-score-pancreatitis-mortality).
On the horizon is an even simpler decision-making tool, the ED-SAS score, which aims to predict severity based off information gleaned solely from the ED visit, at one point in time. It utilizes traditional systemic inflammatory response syndrome (SIRS) criteria, age, and peripheral oxygen saturation to predict 30-day mortality from pancreatitis. The rule was developed and validated within one hospital system, and additional external validation is required prior to wide-scale implementation of this rule.18
Volume expansion and vasopressors
Previously, aggressive fluid therapy was integral in the treatment of acute pancreatitis.19 Patients were not uncommonly given upwards of 10 liters of IV crystalloids over the first 24 hours of a hospital admission. However, abdominal compartment syndrome and acute respiratory distress syndrome (ARDS), once thought to be characteristic of acute pancreatitis may in fact be iatrogenic complications caused by the over-administration of intravenous fluid.20-21
Nevertheless, volume expansion remains important; volume depletion is almost universal in patients with pancreatitis, due to gastrointestinal losses from vomiting, insensible losses from fever and tachypnea, and vasodilation from concomitant infection and a systemic inflammatory response. There is not strong evidence on the ideal amount of fluid for the initial resuscitation, but some studies demonstrate higher rates of ARDS and abdominal fluid collections in patients that receive more than about four liters in the first 24 hours.20-21 The International Association of Pancreatology released guidelines that suggest this as a reasonable limit as well.8 Additionally, this is similar as the amount of fluid recommended in patients with severe sepsis, which has a similar pathophysiology to severe pancreatitis.22 Therefore, we recommend a bolus of one or two liters of crystalloids (20-30 mL/kg) with transition to vasopressors as necessary. This approach may decrease the risk of iatrogenic volume overload. However, high-quality, randomized trials are still needed to determine the optimal volume of fluid administration in patients with severe acute pancreatitis.
Lactated ringer’s (LR) is the preferred crystalloid. A meta-analysis composed of five studies and a total of 450 patients showed that the use of LR reduced the incidence of systemic inflammatory response syndrome (SIRS) at 24 hours after admission when compared to normal saline (NS).23 It did not, however, demonstrate a decrease in mortality. It seems reasonable to conclude that the timing and amount of crystalloid is more important than which solution is used.
Additionally, no strong studies exist on the ideal vasopressor in acute pancreatitis. It is reasonable to start with norepinephrine, as it is the currently recommended first-line vasopressor in sepsis, which has a similar pathophysiologic mechanism as acute pancreatitis.24
After this initial resuscitation, additional fluid may be given judiciously, using a clinical assessment of volume status. This approach is multifactorial:
- Adequate urine output (generally 0.5-1.0 cc/kg/hr)25
- Improvement in hemoconcentration, based on normalization of the hematocrit and blood urea nitrogen (BUN), if elevated at presentation25
- Improvement in lactate, though levels may not completely normalize despite adequate fluids if ongoing necrosis is present25
- Physical exam (moist mucus membranes, lack of skin tenting, etc.) 25
Antibiotics
Patients with necrotizing pancreatitis are at high risk for superimposed infection, which can lead to multisystem organ failure and severe acute pancreatitis. Antibiotics should be avoided when features consistent with superimposed infection are not present (fever, abscess, peripancreatic gas, hemodynamic instability), as this may cause the development of multidrug resistant organisms.26 Superimposed infection is usually due to translocation from gut microbes and is often polymicrobial.27 The initial agents should be broad, cover anaerobic organisms, and be selected in conjunction with the hospital’s antibiogram. The typical clinical course of a bacterial superimposed infection is improvement in symptoms or clinical status followed by an abrupt worsening, usually about a week after symptoms first began.3
Nausea and pain control
Opioid analgesia is often required in patients with severe pancreatitis. Fentanyl and hydromorphone are generally preferred due to their safer side effect profiles; morphine is less preferred, especially in patients with kidney disease, as it is renally cleared. Morphine may also cause sphincter of Oddi dysfunction.28
Ketamine infusions at 0.2-0.3 mg/kg are becoming increasingly popular for pain relief in many settings and have shown promise in patients with acute pancreatitis.29-30 One important consideration of ketamine infusions that must be kept in mind is the large dose of fluid required for infusion; this may be beneficial or deleterious (see section on IV fluids, above).
Acetaminophen may be considered if available intravenously, although used cautiously if any significant hepatic dysfunction is present. Non-steroidal anti-inflammatory drugs (NSAIDs), such as ketorolac, are less preferred due to the increased risk of kidney injury and gastrointestinal bleeding in patients already under significant systemic stress.
Nausea should be treated with repeated doses of centrally-acting antiemetics such as ondansetron or metoclopramide. A baseline electrocardiogram (EKG) may be useful to establish the QTc interval in the case that repeated doses of these medications become necessary.
Feeding
For context, all patients with pancreatitis historically were made nothing by mouth (NPO) status until symptoms were essentially resolved. Since 2008-2010, this practice has shifted in patients with mild pancreatitis–early oral refeeding, if tolerated, is now encouraged, as some studies have shown a reduction in hospital length-of-stay with this approach.31-33 In patients with severe pancreatitis, this same paradigm shift has not occurred, as there are no studies in that population to support this practice. In patients with severe disease, this is a decision best left to the inpatient team that will be managing the patient; generally, patients with severe disease should be kept NPO in the ED.
Cause-specific treatments and adjuncts
- Insulin is used for the treatment of hypertriglyceridemia-induced pancreatitis. Insulin lowers triglyceride levels by reducing their synthesis and also by accelerating their metabolism. Plasmapheresis can be considered in hypertriglyceridemia-induced severe pancreatitis but may result in worse clinical outcomes.34
- ERCP may be required in patients with pancreatitis caused by obstruction of the pancreatic duct with gallstones or biliary sludge. Most guidelines only recommend urgent ERCP when there is concomitant cholangitis present.11 One recent randomized trial showed no reduction in mortality or severe complications in patients with predicted severe biliary pancreatitis that underwent urgent ERCP.35 Research in this area is still ongoing.
- Enteral probiotics seem to increase mortality and are currently not recommended.36
- Steroids have been proposed as a treatment to reduce the severity of the massive inflammatory response commonly seen in non-autoimmune acute pancreatitis. A meta-analysis from 2015 concluded that there may be some benefit to steroids. However, the trial designs were heterogeneous, the dosages varied, and none of the trials in the study were large, randomized, double-blinded, placebo-controlled studies; more high-quality evidence is needed before this becomes standard-of-care.37 However, autoimmune pancreatitis, which is generally more chronic, should be treated with corticosteroids.
- Supportive care for affected organ systems should be provided as they would be in any critically-ill patient (e.g. dialysis for renal failure, mechanical ventilation for respiratory failure, etc.).
Disposition from the Emergency Department
Patients for which you have a strong suspicion for severe pancreatitis should be admitted to a monitored bed (preferably an intensive care unit). This is true even in patients who are hemodynamically stable, as patients may deteriorate days after presenting. Surgical consultation should be strongly considered in patients with hemodynamic instability, evidence of superinfection, or necrosis on imaging.
Pearls
- As with any patient with abdominal pain, always keep a broad differential and include those life-threateningconditions: AAA, bowel ischemia, ectopic, perforated viscus, etc.
- Ultrasound and CT of the abdomen and pelvis can aid in the etiology of pancreatitis or undifferentiatedabdominal pain.
- If concerned for concomitant infection or necrotizing pancreatitis, initiate broad-spectrum antibiotics.
- IVF like in sepsis; avoid overly aggressive fluid resuscitation and start pressors earlier to reduce the risk of iatrogenic volume overload.
- Aggressive, multimodal pain control.
Case resolution
Laboratory evaluation revealed a lipase 566, procalcitonin 1.33, CRP 210, leukocytosis 18,500, and acute kidney injury with Cr 2.1. Contrast-enhanced CT was obtained, which showed emphysematous pancreatitis. While in the ED, the patient became hypotensive and increasingly somnolent. He was given multiple liters of lactated Ringer’s, two rounds of antiemetics, and was started on broad spectrum IV antibiotics. Ultimately, a norepinephrine infusion was required to maintain adequate perfusion. He was admitted to the surgical ICU; during his hospital course, he was taken to the operating room by the acute care surgery service for pancreatic debridement. He required vasopressors and intensive care for several days but was ultimately discharged in stable condition after a thirteen-day hospital course.
References/Further Reading
- Garg SK, Sarvepalli S, Campbell JP, et al. Incidence, Admission Rates, and Predictors, and Economic Burden of Adult Emergency Visits for Acute Pancreatitis. J Clin Gastroenterol. 2019;53(3):220-225.
- Krishna SG, Kamboj AK, Hart PA, Hinton A, Conwell DL. The Changing Epidemiology of Acute Pancreatitis Hospitalizations: A Decade of Trends and the Impact of Chronic Pancreatitis. Pancreas. 2017;46(4):482-488.
- Leppäniemi A, Tolonen M, Tarasconi A, et al. 2019 WSES guidelines for the management of severe acute pancreatitis. World J Emerg Surg. 2019;14:27.
- Banks PA, Bollen TL, Dervenis C, et al. Classification of acute pancreatitis–2012: revision of the Atlanta classification and definitions by international consensus. Gut. 2013;62(1):102-111.
- Natesan S, Lee J, Volkamer H, Thoureen T. Evidence-Based Medicine Approach to Abdominal Pain. Emerg Med Clin North Am. 2016;34(2):165-190.
- Yadav D, Agarwal N, Pitchumoni CS. A critical evaluation of laboratory tests in acute pancreatitis. Am J Gastroenterol. 2002;97(6):1309-1318.
- Ismail OZ, Bhayana V. Lipase or amylase for the diagnosis of acute pancreatitis? Clin Biochem. 2017;50(18):1275-1280.
- Working Group IAP/APA Acute Pancreatitis Guidelines. IAP/APA evidence-based guidelines for the management of acute pancreatitis. Pancreatology. 2013;13(4 Suppl 2):e1-e15.
- Pancreatitis UKWP on A, UK Working Party on Acute Pancreatitis. UK guidelines for the management of acute pancreatitis. Gut. 2005;54(suppl_3):iii1-iii9. doi:10.1136/gut.2004.057026
- Yokoe M, Takada T, Mayumi T, et al. Japanese guidelines for the management of acute pancreatitis: Japanese Guidelines 2015. J Hepatobiliary Pancreat Sci. 2015;22(6):405-432.
- Leal C, Almeida N. Predicting Severity in Acute Pancreatitis: A Never-Ending Quest…. GE Port J Gastroenterol. 2019;26(4):232-234.
- Kokulu K, Günaydın YK, Akıllı NB, et al. Relationship between the neutrophil-to-lymphocyte ratio in acute pancreatitis and the severity and systemic complications of the disease. Turk J Gastroenterol. 2018;29(6):684-691.
- Larvin M. Assessment of severity and prognosis in acute pancreatitis. Eur J Gastroenterol Hepatol. 1997;9(2):122-130.
- Mofidi R, Suttie SA, Patil PV, Ogston S, Parks RW. The value of procalcitonin at predicting the severity of acute pancreatitis and development of infected pancreatic necrosis: systematic review. Surgery. 2009;146(1):72-81.
- Staubli SM, Oertli D, Nebiker CA. Laboratory markers predicting severity of acute pancreatitis. Crit Rev Clin Lab Sci. 2015;52(6):273-283.
- Aggarwal A, Manrai M, Kochhar R. Fluid resuscitation in acute pancreatitis. World J Gastroenterol. 2014;20(48):18092-18103.
- Ranson JH, Rifkind KM, Roses DF, Fink SD, Eng K, Spencer FC. Prognostic signs and the role of operative management in acute pancreatitis. Surg Gynecol Obstet. 1974;139(1):69-81.
- Wu BU, Johannes RS, Sun X, Tabak Y, Conwell DL, Banks PA. The early prediction of mortality in acute pancreatitis: a large population-based study. Gut. 2008;57(12):1698-1703.
- Miller J, Wu Y, Safa R, et al. Derivation and validation of the ED-SAS score for very early prediction of mortality and morbidity with acute pancreatitis: a retrospective observational study. BMC Emerg Med. 2021;21(1):16.
- de-Madaria E, Soler-Sala G, Lopez-Font I, et al. 719 Influence of Fluid Therapy on the Prognosis of Acute Pancreatitis: A Prospective Cohort Study. Gastroenterology. 2010;138(5):S – 96. doi:10.1016/s0016-5085(10)60440-4
- Eckerwall G, Olin H, Andersson B, Andersson R. Fluid resuscitation and nutritional support during severe acute pancreatitis in the past: What have we learned and how can we do better? Clin Nutr. 2006;25(3):497-504.
- Semler MW, Rice TW. Sepsis Resuscitation: Fluid Choice and Dose. Clin Chest Med. 2016;37(2):241-250.
- Iqbal U, Anwar H, Scribani M. Ringer’s lactate versus normal saline in acute pancreatitis: A systematic review and meta-analysis. Journal of Digestive Diseases. 2018;19(6):335-341. doi:10.1111/1751-2980.12606
- Avni T, Lador A, Lev S, Leibovici L, Paul M, Grossman A. Vasopressors for the Treatment of Septic Shock: Systematic Review and Meta-Analysis. PLoS One. 2015;10(8):e0129305.
- Mackenzie DC, Noble VE. Assessing volume status and fluid responsiveness in the emergency department. Clin Exp Emerg Med. 2014;1(2):67-77.
- Vege SS, DiMagno MJ, Forsmark CE, Martel M, Barkun AN. Initial Medical Treatment of Acute Pancreatitis: American Gastroenterological Association Institute Technical Review. Gastroenterology. 2018;154(4):1103-1139.
- Mowbray NG, Ben-Ismaeil B, Hammoda M, Shingler G, Al-Sarireh B. The microbiology of infected pancreatic necrosis. Hepatobiliary Pancreat Dis Int. 2018;17(5):456-460.
- Wu S-D. Effects of narcotic analgesic drugs on human Oddi’s sphincter motility. World Journal of Gastroenterology. 2004;10(19):2901. doi:10.3748/wjg.v10.i19.2901
- Agerwala SM, Sundarapandiyan D, Weber G. Ketamine Use for Successful Resolution of Post-ERCP Acute Pancreatitis Abdominal Pain. Case Rep Anesthesiol. 2017;2017:7845358.
- Motov S, Drapkin J, Likourezos A, et al. Continuous Intravenous Sub-Dissociative Dose Ketamine Infusion for Managing Pain in the Emergency Department. West J Emerg Med. 2018;19(3):559-566.
- Eckerwall GE, Tingstedt BBÅ, Bergenzaun PE, Andersson RG. Immediate oral feeding in patients with mild acute pancreatitis is safe and may accelerate recovery—A randomized clinical study. Clin Nutr. 2007;26(6):758-763.
- Li J, Xue G-J, Liu Y-L, et al. Early oral refeeding wisdom in patients with mild acute pancreatitis. Pancreas. 2013;42(1):88-91.
- Sathiaraj E, Murthy S, Mansard MJ, Rao GV, Mahukar S, Reddy DN. Clinical trial: oral feeding with a soft diet compared with clear liquid diet as initial meal in mild acute pancreatitis. Aliment Pharmacol Ther. 2008;28(6):777-781.
- He W-H, Yu M, Zhu Y, et al. Emergent Triglyceride-lowering Therapy With Early High-volume Hemofiltration Against Low-Molecular-Weight Heparin Combined With Insulin in Hypertriglyceridemic Pancreatitis: A Prospective Randomized Controlled Trial. J Clin Gastroenterol. 2016;50(9):772-778.
- Schepers NJ, Hallensleben NDL, Besselink MG, et al. Urgent endoscopic retrograde cholangiopancreatography with sphincterotomy versus conservative treatment in predicted severe acute gallstone pancreatitis (APEC): a multicentre randomised controlled trial. Lancet. 2020;396(10245):167-176.
- Besselink MGH, van Santvoort HC, Buskens E, et al. Probiotic prophylaxis in predicted severe acute pancreatitis: a randomised, double-blind, placebo-controlled trial. Lancet. 2008;371(9613):651-659.
- Dong L-H, Liu Z-M, Wang S-J, et al. Corticosteroid therapy for severe acute pancreatitis: a meta-analysis of randomized, controlled trials. Int J Clin Exp Pathol. 2015;8(7):7654-7660.