Authors: Cassandra Schandel (EM Resident Physician, Carilion Clinic), Sarah Chrabaszcz (EM Resident Physician, Carilion Clinic), Rahul Nadendla (EM Resident Physician, Carilion Clinic), and Michelle Clinton (EM Attending Physician, Assistant Program Director, Carilion Clinic) // Reviewed by: Marina Boushra (EM Attending Physician, Clinical Assistant Professor, Vidant Health); Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit)
Case:
A 7-year-old male with no PMH presents to the ED with a chief complaint of abdominal pain. His mother states that he has had diarrhea, nausea, and vomiting. His vital signs on arrival are BP 87/45 mmHg, HR 122 bpm, RR 35/min, SpO2 97% on room air. Physical exam shows a child in mild distress with generalized abdominal tenderness without guarding or rebound. The POC glucose is 222 mg/dL.
Background:
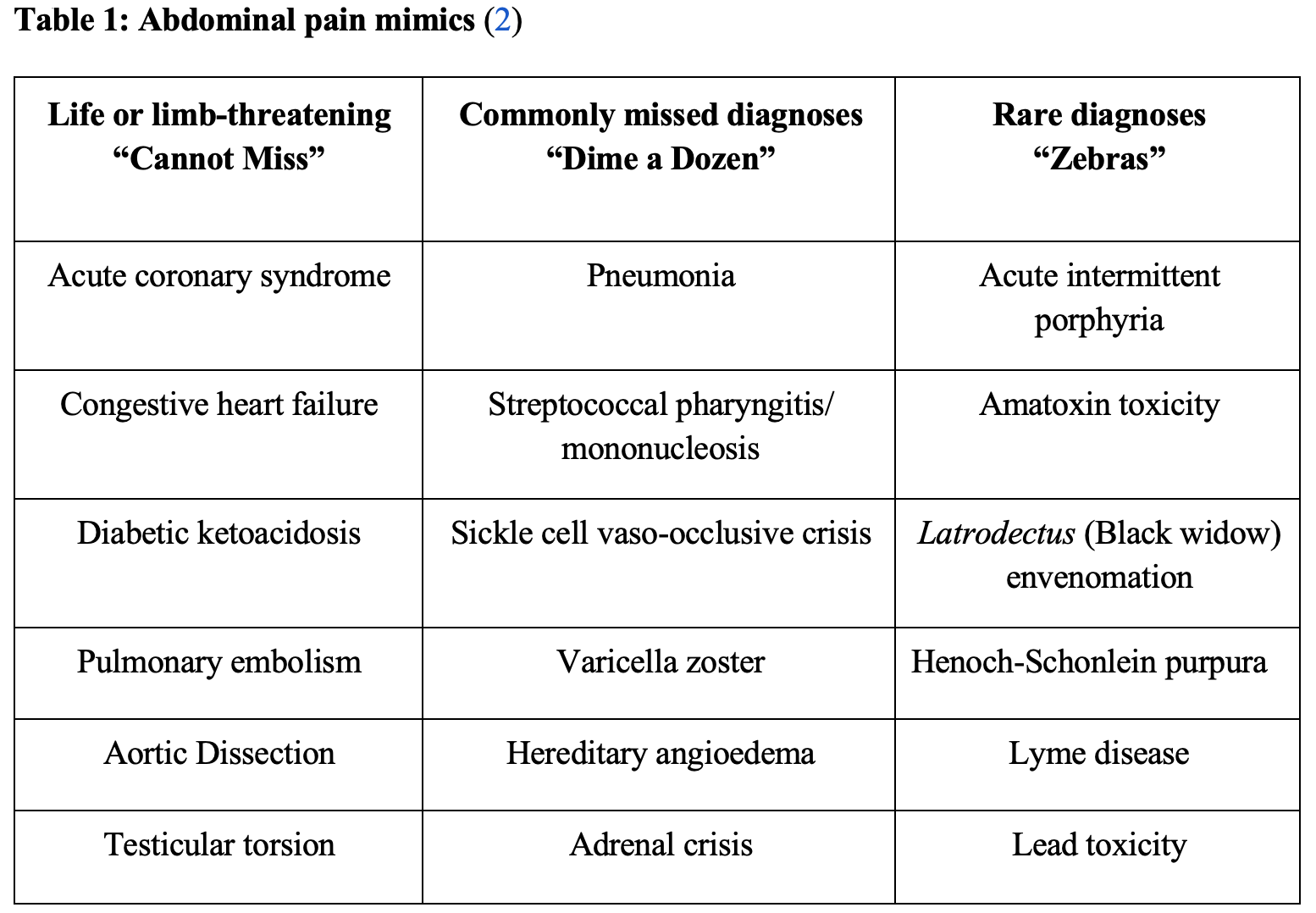
Abdominal pain comprises approximately 5% of emergency department encounters (1). This number is drastically higher in children, elderly, pregnant and immunocompromised patients, who may present atypically and are prone to misdiagnosis (1). A presenting complaint of abdominal pain requires comprehensive evaluation starting with a detailed history and physical exam. While the typical differential diagnosis for abdominal pain should include the usual suspects of intra-abdominal pathology based on location and associated symptoms, extra-abdominal mimics and rare conditions should be considered as well. These diagnoses are easily categorized into “cannot miss,” “dime a dozen,” and “zebras” (Table 1).
“Can’t Miss” Diagnoses
Acute coronary syndrome (ACS):
ACS is an umbrella term of cardiac syndromes that consists of unstable angina, non-ST elevation myocardial infarction (NSTEMI), and ST-elevation myocardial infarction (STEMI). It typically presents with chest pain with variable radiation, shortness of breath, nausea or vomiting, and diaphoresis, often in the setting of known risk factors (smoking, hypertension (HTN), hyperlipidemia (HLD), diabetes mellitus (DM), sedentary lifestyle, etc.). However, it can present atypically, especially in women, elderly, and patients with diabetes (3). Up to 33% of patients diagnosed with ACS do not have chest pain on presentation to the hospital (4). Alarmingly, 2% of acute myocardial infarctions are discharged from the emergency department with an incorrect diagnosis (5). Physicians cite atypical presentation as the most common reason for missed diagnosis (5). Literature shows that these atypical presentations have up to twice the mortality of their counterparts who presented with typical symptoms (6,7). A study in 2004 demonstrated that nausea/vomiting was the dominant presenting symptom in roughly 24% of ACS patients (8). Specifically, women presenting with ACS were more likely to have nausea/vomiting compared to men presenting with ACS (9). Physicians should therefore be wary of delays in diagnosis, especially in these atypical presentation patient populations. Consider cardiac evaluation in patients with nondescript abdominal pain or nausea/vomiting. Evaluation should include serial ECGs, cardiac biomarkers, and a chest radiograph. It is important to remember that only 50% of patients with ACS will have a diagnostic ECG, and a normal ECG cannot be used to rule out ACS (10).
Resources for further reading:
http://www.emdocs.net/em3am-acute-coronary-syndrome/
https://rebelem.com/tag/acute-coronary-syndrome/
http://www.emdocs.net/page/2/?s=acute+coronary+syndrome
Congestive heart failure (CHF):
CHF can be categorized as systolic (also called heart failure with reduced ejection fraction (HFrEF), diastolic (also called heart failure with preserved ejection fraction (HFpEF), or combined heart failure. It can also be divided into classes by using the New York Heart Association, which takes into account functional status (11). CHF typically presents with chest pain, palpitations, or worsening shortness of breath in the setting of known risk factors (CAD, previous MI, HTN, valve disease, congenital heart disease, cardiomyopathy, DM, etc.). In addition, heart failure can present atypically with either localized or generalized abdominal pain, abdominal distention, anorexia, and nausea/vomiting, especially in right heart failure (12). The decreased vascular capacitance in the splanchnic vasculature of patients with CHF can lead to these abdominal symptoms and worsen intravascular volume status (13). Furthermore, increasing abdominal distension can lead to non-focal pain and be masked by obesity. Heart failure can also cause intra-abdominal injury. Cardiogenic liver injury or cardiohepatic syndrome can occur from increased hepatic venous pressure and impaired perfusion due to heart failure (14,15). Heart failure can cause cardiorenal syndrome as well. Kidney dysfunction results from inadequate renal perfusion and/or elevated renal venous pressure and intra-abdominal pressure (16). Heart failure in the ED can initially be evaluated by looking for physical exam findings suggestive of CHF including rales, jugular venous distension (JVD), an S3 or S4 on cardiac auscultation, hepatojugular reflux, and leg edema. Further diagnostic studies should include obtaining a brain natriuretic peptide (BNP), complete metabolic panel, bedside echocardiogram, thoracic ultrasound, and a chest radiograph.
Resources for further reading:
http://www.emdocs.net/ed-management-heart-failure-pearls-pitfalls/
http://www.emdocs.net/heart-failure-sneaky-triggers-and-clinical-pearls/
https://coreem.net/core/congestive-heart-failure/
Diabetic ketoacidosis (DKA):
DKA presents with hyperglycemia and acidosis due to insulin deficiency. Diagnostic findings include: glucose >200 mg/dL in pediatric patients and >250 mg/dL in adults, pH <7.3 or bicarbonate <15 mEq/L, and ketosis (acetoacetate, beta-hydroxybutyrate) (17, 18). Common presentations include polydipsia, polyuria, generalized weakness, abdominal pain, and nausea/vomiting. Physical exam can demonstrate tachypnea, hypotension, tachycardia, altered mental status, and abdominal tenderness. One study in 2002 found that 46% of patients with DKA experienced abdominal pain (19). In approximately 35% of those patients, the cause of pain was secondary to metabolic decompensation. (19) Abdominal pain is not related to severity of hyperglycemia or dehydration, but this study did find a strong association between pain and metabolic acidosis (19). The foundations of treatment are intravenous fluids, insulin, and electrolyte repletion (especially potassium as patients often have total body potassium depleted despite normal or elevated serum levels) (20).
Resources for further reading:
http://www.emdocs.net/em3am-diabetic-ketoacidosis/
http://www.emdocs.net/diabetic-ketoacidosis-sneaky-triggers-clinical-pearls/
https://rebelem.com/diabetic-ketoacidosis-dka-myths/
Pulmonary embolism (PE):
PEs can be categorized as massive (with hypotension), sub-massive (without hypotension, but with right ventricular strain, evidence of heart failure, or elevated cardiac biomarkers), and lower risk PEs (21). Patients typically present with pleuritic chest pain, shortness of breath, and/or calf asymmetry in the setting of known risk factors (obesity, hormonal therapy, long periods of immobilization, smoking, etc.). Consequences of a missed PE can be catastrophic, and this disease process is believed to be responsible for 50,000-100,000 deaths annually (22). Nearly 7% of patients with PE present with gastrointestinal complaints ranging from vague pain to an acute abdomen (23). Patients with a low pre-test probability for PE can be evaluated using clinical decision-making rules such as the pulmonary embolism rule-out criteria (PERC), Wells’ criteria and D-dimer (24). In patients with risk factors for PE, do not use these rules as these are not validated in high-risk populations (25). These patients warrant definitive testing with CT- angiography. CT is the overwhelmingly preferred imaging technique, but you may need to consider other modalities if the patient is pregnant, has a contrast allergy, or chronic kidney disease (26). For pregnant patients, guidelines from multiple societies vary on performing a CT-PA vs V/Q scan; some studies even look at bilateral leg compression US with a low threshold to treat patients empirically to avoid radiation. Ultimately your choice will depend on what is available at your institution and its guidelines (27). For patients with contrast allergies and kidney disease, VQ studies are the preferred modality (28).
Resources for further reading:
http://www.emdocs.net/pulmonary-embolism-management-of-the-unstable-patient/
http://www.emdocs.net/r-e-b-e-l-em-the-critical-pulmonary-embolism-patient/
Aortic dissection:
This is estimated to occur in approximately 5 to 30 persons per million, and is a “cannot miss” diagnosis for emergency providers (29). Aortic dissection occurs secondary to disruption of layers of the aortic wall, most commonly from a primary tear that propagates under high pressure and causes hypoperfusion to the true lumen. Risk factors include known aortic aneurysm, hypertension (up to 70%), atherosclerosis, connective tissue disorders such as Marfan and Ehlers-Danlos syndromes, previous history of dissection, Turner syndrome, bicuspid aortic valve, or abrupt, transient increase in blood pressure (strenuous lifting, sympathomimetic drug use, energy drinks, etc.), and pregnancy/delivery (29). The Stanford classification categorizes aortic dissection as Type A, which involves the ascending aorta, and Type B, which involves only the descending aorta. Patients classically present with chest pain that is described as tearing with radiation into the back; however, pain can occur anywhere along the dissection and may mimic other common etiologies. High-risk presentations include chest, back, or abdominal pain with concerning physical exam findings such as a pulse deficit, high systolic blood pressure, or a neurologic deficit (30). The International Registry of Acute Aortic Dissection found that 30% of patients with a Type A or B dissection present with abdominal pain (31). Abdominal pain occurs more commonly in Type B dissections (32). There is an increased mortality in patients presenting with abdominal pain because there is no specific biomarker to rule out aortic dissection, and imaging only the abdomen can miss Type A or B dissection in the chest (33). Further diagnostics should include a bedside ultrasound to evaluate the aorta and subsequent advanced imaging if warranted. TTE is especially useful in identifying pericardial effusion from proximal dissection, severe aortic root dilatation, regional wall motion abnormality, aortic regurgitation, or intimal aortic flap (34).Treatment focuses on reduction of shear force on the aorta through blood pressure and heart rate control and surgical consultation.
Resources for further reading:
http://www.emdocs.net/core-em-aortic-dissection/
http://www.emdocs.net/acute-aortic-dissection-3/
http://coreem.net/core/aortic-dissection/
https://litfl.com/acute-aortic-dissection/
Testicular torsion:
Testicular torsion is a true urologic emergency and is a time-sensitive diagnosis. Torsion occurs when the spermatic cord twists, causing venous congestion which can progress to arterial compromise and eventually testicular ischemia. Testicular torsion occurs in a bimodal distribution in the first year of life and adolescence (35). The classic presentation includes acute unilateral pain, swelling, and erythema of the testicle, absent cremasteric reflex, abnormal lie, and nausea/vomiting (36). A 2013 retrospective study from the Journal of Pediatric Urology found that 9 of 84 children diagnosed with testicular torsion presented with only abdominal pain (37). In 6 of those patients, the scrotum was not evaluated, and 5 children were subsequently unable to have testicular salvage (37). If clinical suspicion for torsion is high, diagnostic evaluation should not delay surgical management. Imaging is not always accurate and can miss torsion especially if it is intermittent; do not be lulled into a false sense of security based on a normal ultrasound. Ultrasound is the preferred modality to evaluate for potential testicular torsion. Key findings include twisting of spermatic cord (whirlpool sign), altered blood flow, and increased size of testis (38).
Success rates for testicular salvage have been reported as greater than 90% with time to salvage of less than 6 hours, but this quickly drops off to 0-10% after 12-24 hours (39). A 2017 study was able to further prognosticate based on degree of torsion and found that patients with 15 hours of symptoms and >860 degrees of torsion have a 50% salvage rate, but also noted a 25% rate of atrophy following orchiopexy due to suspected reperfusion injury (39).
Resources for further reading:
http://www.emdocs.net/testicular-torsion-pearls-and-pitfalls/
https://coreem.net/core/testicular-torsion/
“A Dime a Dozen” Diagnoses
Pneumonia:
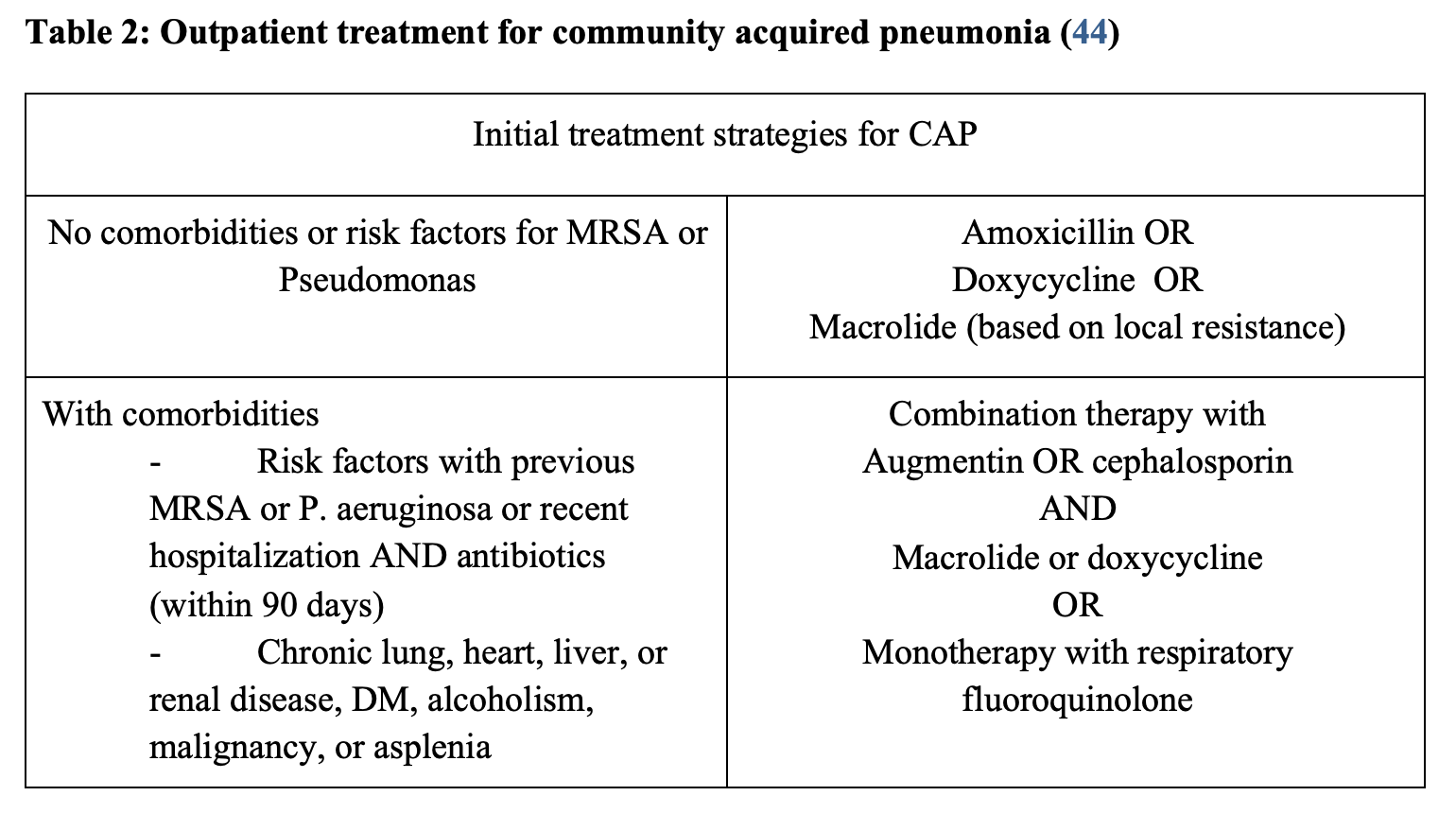
Pneumonia is a clinical diagnosis that typically presents with fever, productive cough, shortness of breath and chest pain. It can be categorized into community acquired (CAP), hospital acquired (HAP), and ventilator associated (VAP). The term HCAP (healthcare-associated pneumonia) is no longer used (40). Pneumonia can be caused by a variety of infectious agents including bacteria, fungi, and viruses. In pediatric patients and those with lower lobar or retrocardiac pneumonias, upper abdominal pain can be the main presenting symptom. In fact, pneumonia is the most common extra-abdominal cause of abdominal pain in pediatric patients (41,42). Diaphragmatic irritation can precipitate symptoms due to the innervation of the diaphragm and peritoneum (43). Chest radiographs can aid in differentiating abdominal pain that is due to a pulmonary cause, especially in patients with fever and respiratory complaints. Treatment is based on local resistance patterns and disease severity. Please refer to the Society of Infectious Diseases for the most recent guideline on therapies. A noteworthy change in the outpatient treatment recommendation for CAP is that single agent macrolide treatment is no longer recommended, as greater than 25% of Streptococcus pneumoniae demonstrate resistance. The local antibiogram may assist with antibiotic choice. Treatment recommendations for CAP per the 2019 IDSA guidelines are summarized below (Table 2).
Resources for further reading:
https://www.emrap.org/episode/c3pneumoniain/c3pneumoniain
http://www.emdocs.net/emdocs-cases-ed-evaluation-community-acquired-pneumonia/
Streptococcal pharyngitis/Mononucleosis:
Most cases of pharyngitis are viral, benign, and self-limiting; however, pharyngitis caused by Group A Streptococcus (GAS) and Epstein-Barr virus (EBV) are two noteworthy diagnoses because they can present with abdominal pain.
Streptococcal pharyngitis is a specific pharyngitis caused by GAS. Patients typically present with sore throat, fever, headache, and cervical lymphadenopathy; however, up to 10% of children with new onset abdominal pain and fever have streptococcal pharyngitis (45). It can be clinically difficult to diagnose because of the time and cost associated with testing. Interestingly, only 10-15% of adult patients with sore throat have streptococcal pharyngitis, but up to 75% receive antibiotics (46) One study used gastrointestinal symptoms as a diagnostic criteria but did not find a significant difference between pediatric and adult patients (47). Typically, the modified Centor criteria (Table 3) are used to guide testing and treatment.
Potential non-suppurative complications of untreated GAS infections include rheumatic fever and subsequent rheumatic heart disease. This disease can also progress to several soft tissue infections (suppurative complications) that include otitis media and sinusitis as well as peritonsillar abscess, retropharyngeal abscess, or epiglottis. An additional complication includes post-streptococcal glomerulonephritis which cannot be prevented with antibiotics. It typically occurs in 1-2 weeks following sore throat and 6 weeks following a skin infection, and is found incidentally on a routine urine analysis (48). The exact mechanism of this disease is not fully determined, but is a type II hypersensitivity reaction. Usually this disease has an excellent prognosis especially in children, while one in two adults will have reduced renal function or persistent proteinuria secondary to this (48).
A 2015 Rebel EM review of the literature on antibiotic use and complications in the treatment of streptococcal pharyngitis asked three specific questions: 1) do antibiotics reduce symptoms, 2) do antibiotics reduce suppurative complications, and 3) do antibiotics reduce non-suppurative complications. Overall, they found the literature supports that complications of antibiotics generally outweigh benefits in developed countries (49). If the decision is made to treat with antibiotics, penicillin or amoxicillin provide adequate coverage. Corticosteroids are controversial but can be considered for symptomatic relief. No current recommendations exist for use of steroids in acute pharyngitis, but studies in adults and children show that corticosteroids in combination with antibiotic treatment provide symptomatic relief of pain and faster recovery (50).

Mononucleosis: Mononucleosis is a multisystem disorder caused by Epstein-Barr virus (EBV). Manifestations include headache, fevers, and exudative pharyngitis. In contrast to the anterior cervical lymphadenopathy seen in streptococcal pharyngitis, posterior cervical chain lymphadenopathy is typical with mononucleosis. Fifty percent of patients with symptomatic EBV infection have splenomegaly, of whom up to 0.5% will have spontaneous or secondary splenic rupture (52). Patients with mononucleosis should receive strict discharge instructions recommending no contact sports for 6-8 weeks. Testing for mononucleosis can be tricky as the Monospot test (heterophile antibody) has low sensitivity during the first 1-2 weeks of illness but increases after those initial 2 weeks. The Monospot test has a specificity near 100% and a sensitivity of roughly 85% (53). The sensitivity is decreased early in the disease course as previously discussed and also in children less than 2 years old (with some sources saying less than 4 years old) (53). If the Monospot is negative, which is likely if testing in the first 2 weeks, then clinical findings such as fevers, fatigue, exudates, posterior lymphadenopathy or axillary lymphadenopathy, and an absolute lymphocyte count greater than 4000 or greater than 10% atypical lymphocytosis should make mononucleosis a consideration (54).Treatment for mononucleosis is supportive and amoxicillin administration for a mistaken diagnosis of streptococcal pharyngitis may lead to a characteristic morbilliform rash that improves with antibiotic discontinuation (55).
Resources for further reading:
http://www.emdocs.net/strep-throat-mimics-pearls-pitfalls/
https://www.emrap.org/corependium/chapter/recbXWUYBqyjwLr10/Pharynx-and-Throat-Emergencies
https://www.emrap.org/episode/c3sorethroat1/c3sorethroat3
Sickle cell vaso-occlusive crisis:
Sickle cell hemoglobin (HbSC) forms rigid polymers when deoxygenated and cannot quickly transition between oxygenated and deoxygenated forms like normal hemoglobin. Stressors such as illness, dehydration, hypoxia, or acute changes in environment/temperature can lead to crisis in which sickled B-globulin molecules obstruct capillaries and may cause pain, ischemia and/or organ damage.
Sickle cell crises are further broken down into vaso-occlusive crisis, splenic sequestration, aplastic crises, and hemolytic crises. In a vaso-occlusive crisis, pain is triggered by an inflammatory cascade. Patients usually present with pain in the extremities, back, and chest but can have pain anywhere. Obtain lab work to assess for anemia, thrombocytopenia, elevated LDH, and low reticulocyte count to assist with diagnosis. Consider obtaining LFTs, lipase, lactate, and abdominal imaging if there is clinical concern as complications of these crises can include cholecystitis, liver disease, ischemic colitis, and pancreatitis (56). A study from Africa in 2011 found that patients in crisis typically had single site abdominal pain, particularly in the epigastric region (57). The abdominal pain in sickle cell is generally attributed to vaso-occlusive crisis but can also be due to enlarged lymph nodes, bone marrow hyperplasia, cholelithiasis, or splenic pathology such as splenic hemorrhage or splenic thrombosis (57).
First-line treatment is opioids, particularly longer acting ones such as morphine or hydromorphone. Assessing dehydration and administering intravenous fluids is important as well. However, beware that administration of normal saline may lead to hyperchloremic metabolic acidosis and increased sickling, as well as increased risk for atelectasis, pulmonary edema, and acute chest syndrome (58, 59). If giving intravenous fluids, administer D5 ½ normal saline infusion at maintenance rate.
Resources for further reading:
https://litfl.com/sickle-cell-crisis/
https://www.ncbi.nlm.nih.gov/books/NBK526064/
Shingles:
Varicella zoster virus (VZV) is the infectious agent which causes chickenpox in children. When reactivated, it results in shingles. This virus resides in the dorsal root ganglia after initial infection. Approximately 1 in 3 people in the United States will develop shingles in their lifetime (60). High-risk populations include immunocompromised patients (cancer, HIV, transplant, or medications such as steroids or chemotherapy), age greater than 50 years, and patients with previous chickenpox (60). The classic shingles rash occurs in a dermatomal pattern, and patients can present with pain prior to the tell-tale rash. The rash typically occurs in a dermatomal pattern with a single stripe of blisters that can present with pain and itching; multiple dermatomes can be involved in immunocompromised patients. The rash typically lasts 2-4 weeks, but patients can have post-herpetic neuralgia long after it disappears. When examining a patient with abdominal pain, it is very important to expose the abdomen and inspect the skin. In a case study from 2009, a fourteen year-old patient presented to the emergency department for abdominal pain and was diagnosed with peritonitis; however, he subsequently developed a maculopapular rash to the left inguinal region (61). A rare presentation of infection is visceral zoster, and patients can present with symptoms mimicking an acute abdomen (62). While skin findings are expected with zoster infection, there are case reports of visceral zoster in the absence of cutaneous eruptions (63). Visceral zoster without skin findings would be a clinical diagnosis if made in the ED, and treatment includes antivirals.
Resources for further reading:
https://coreem.net/core/herpes-zoster/
https://litfl.com/varicella-zoster/
Hereditary angioedema:
Hereditary angioedema is an autosomal dominant deficiency of C1 esterase inhibitor that results in vasodilation and edema. The upper respiratory and gastrointestinal tract are most commonly affected (64). Symptoms include facial swelling, shortness of breath, voice changes, vague abdominal pain, abdominal distention, and nausea/vomiting. The abdominal pain is described as colicky and rated as severe in 87% of cases (65). Angioedema can lead to intestinal obstruction. Initial examination should be prioritized to assess the patient’s airway. This is a clinical diagnosis, but diagnostic testing may be required to evaluate for other causes such as surgical pathology based on the patient’s clinical presentation. Treatment includes C1 esterase inhibitor or fresh frozen plasma which includes C1 esterase inhibitor.
Resources for further reading:
http://www.emdocs.net/emdocs-cases-angioedema-evaluation-and-management/
https://coreem.net/core/angioedema/
“Zebra” Diagnoses:
Adrenal crisis:
Adrenal crisis occurs secondary to acute deficiency of adrenocortical hormones, particularly cortisol and aldosterone. Primary adrenal insufficiency (Addison’s disease) occurs with partial or complete destruction of the adrenal cortex. In secondary adrenal insufficiency, lack of stimulation of the adrenal gland leads to low levels of hormones. This is the most common cause overall (66). Both primary and secondary adrenal insufficiency can present with hypotension that can be severe and refractory to intravenous fluids and vasopressors, as well as non-specific symptoms of weakness, confusion, nausea/vomiting, and abdominal pain. Up to 22% of patients present with abdominal pain that mimics an acute abdomen (67). Acute stress situations such as surgery, infections, burns, trauma, and cardiovascular or metabolic events can precipitate an adrenal crisis. Patients previously on prolonged steroid therapy or with a history of autoimmune disease are also prone to adrenal crisis. In primary adrenal insufficiency, electrolyte abnormalities such as hyponatremia, hyperkalemia, and metabolic acidosis can be seen (68). Hyperpigmentation is also expected in chronic primary insufficiency due to increased ACTH production. In secondary adrenal insufficiency, dehydration, hypotension, and hyperkalemia are less evident as mineralocorticoid production is mostly preserved, but hypoglycemia is more commonly seen compared to primary insufficiency (68). Imaging can show calcification in the adrenal gland or renal tuberculosis (69). Treatment involves steroid replacement with intravenous hydrocortisone, intravenous fluid boluses, and correction of hypoglycemia.
Resources for further reading:
https://emcrit.org/ibcc/adrenal-crisis/
http://www.emdocs.net/adrenal-crisis-in-the-ed/
Acute intermittent porphyria:
Acute intermittent porphyria is an inherited disorder of heme biosynthesis. Its prevalence has been cited as 1 in 20,000 in European studies (70). Patients present with neurologic symptoms, hyponatremia, in addition to abdominal pain. They can also present with nausea, vomiting, diarrhea or constipation, and dark red urine. It has been reported that up to 20% of patients will present with seizures (71). Symptoms are due to an accumulation of metabolites in the heme biosynthesis pathway. Attacks can be triggered by stress, hormonal changes, drugs, alcohol, diet, etc. Typically, patients are young healthy females presenting with a premenstrual elevation in progesterone or oral contraceptive use as their inciting factor (72). Physical examination and imaging tends to be to be non-specific. In order to diagnose acute intermittent porphyria, obtain an elevated random urine or plasma porphobilinogen. Treatment includes intravenous heme and supportive care. On a historical side note, acute intermittent porphyria is called vampire disease and is possibly the origin of Dracula. Patients with this disease tend to have photosensitive skin and red urine, which was thought to be secondary to ingestion human blood ingestion.
Resources for further reading:
https://first10em.com/rapid-review-acute-intermittent-porphyria/
http://www.signavitae.com/2015/06/porphyrias-diagnostic-challenge-in-emergency-room/
Amatoxin toxicity:
Amatoxins are found in several species of mushrooms including Amanita, Lepiota, and miscellaneous species in the Pacific Northwest, Ohio, Pennsylvania, and New Jersey. Over 95% of deaths due to mushroom ingestions are due to these species, with Amanita phalloides having the highest mortality overall (73). Toxicity causes a protein deficit and cellular death resulting in multiorgan failure (74). The liver is the most commonly affected organ, so appropriate work-up includes obtaining LFTs, coagulation profile, ammonia and electrolytes. Poisoning occurs in three stages: initial/incubation, gastrointestinal, and cytotoxic. Symptoms usually occur at 6-12 hours during the gastrointestinal stage which include abdominal pain, nausea/vomiting, and diarrhea. The cytotoxic phase presents as jaundice, seizures, coma and ultimately death (74). Diagnosis can be made by clinical evaluation and confirmed by urinary amanitin analysis (75). Treatment includes supportive care, activated charcoal for decontamination, amatoxin update inhibitors (intravenous silibinin, intravenous penicillin G), antioxidants (intravenous N-acetylcysteine, intravenous cimetidine, intravenous vitamin C), and possibly liver transplantation (76,77). As with any toxic ingestion, Poison Control can provide recommendations and guide management, so they should be contacted early in the patient’s clinical course.
Resources for further reading:
https://www.ncbi.nlm.nih.gov/books/NBK431052/
http://www.emdocs.net/tox-cards-mushrooms/
Black widow envenomation:
This is a rare toxidrome, but an important one to know, as a black widow spider bite can present with generalized abdominal pain. Interestingly, it is only the female spiders that have the tell-tale red hourglass shape. Typically, patients are unaware of the bite but will report a history of working in cool, dark environments such as a basement or shed. Black widow spiders usually inhabit warmer climates. Symptoms develop approximately 30 to 120 minutes following the bite and include neurologic and autonomic dysfunction such as weakness, paresthesias, diffuse muscle pain, agitation, tachycardia, hypertension, diaphoresis, and nausea/vomiting. The spider produces a protein venom, α-Latrotoxin that attacks the presynaptic neurons resulting in these symptoms. Due to the intermittent muscle cramping, patients can present with abdominal rigidity mimicking an acute abdomen. Laboratory testing is nonspecific but can include hematuria, leukocytosis and elevation in creatine kinase (CK), glucose and LFTs. Treatment is supportive care including analgesia, benzodiazepines, muscle relaxants and tetanus prophylaxis. There is an anti-venom that can be given to patients with moderate to severe symptoms, but it is in short supply. One study found administering the anti-venom significantly reduced the duration of symptoms, and complete resolution of symptoms at a mean time of 31 minutes (78). While there have been no reported deaths from envenomation, there are case reports of anaphylaxis from anti-venom (79,80).This is another instance where Poison Control and local toxicologists can help guide management.
Resources for further reading:
https://www.poison.org/articles/2012-jun/black-widow-spiders
https://www.mayoclinic.org/diseases-conditions/spider-bites/diagnosis-treatment/drc-20352377
Henoch- Schonlein purpura (HSP):
HSP is an IgA vasculitis and is most common in children (ages 4-6 years). It most commonly occurs in Caucasian and Asian males. Most patients report a history of upper respiratory infection prior to the onset of a rash and abdominal pain. Gastrointestinal symptoms occur in 50-75% of patients (81). Abdominal pain commonly precedes the classic palpable rash by up to 2 weeks in approximately 20% of people (81). For diagnosis, patients need a purpuric/petechial rash with a minimum of 1 of the 4 criteria as outlined in the chart below (Table 4) (82). Typically, this disease is self-limiting and resolves within 4 weeks. One long-term complication, however, is chronic kidney disease and can advance to end-stage renal disease requiring hemodialysis. Renal failure is more common in adults as compared to pediatric patients (83). Diagnosis is based on clinical manifestations. Treatment is typically supportive; steroids for the treatment of renal disease or severe abdominal pain remain controversial (84).

Resources for further reading:
http://www.emdocs.net/em3am-henoch-schonlein-purpura-hsp/
Lyme Disease:
Lyme disease is a tick-borne illness caused by the spirochete Borrelia burgdorferi and is spread by the deer tick, Ixodes scapularis. Approximately 95% of cases occur in the Midwest and Northeast United States (86). While the history of a tick bite can help clue you in on a diagnosis, one study found only 25% of patients were able to recall a known tick bite (87). Early symptoms include fever, fatigue, malaise, headache, myalgias, arthralgias, and the characteristic target-like erythema migrans rash. According to the CDC, up to 30% of people, however, do not report a rash with Lyme disease (88). Later stages of the disease involve multiple organ systems such as neurologic, musculoskeletal, cutaneous, cardiovascular, hepatic, ocular, and renal. Patients can present with generalized abdominal pain due to neurologic involvement. Criteria for diagnosis includes the presence of an erythema migrans rash or at least one later clinical manifestation such as neurologic, musculoskeletal or cardiovascular plus confirmatory laboratory testing. Treatment is based on the stage of the disease and includes amoxicillin, cefuroxime, or doxycycline; more severe symptoms require ceftriaxone or penicillin G (89).
Further resources:
http://www.emdocs.net/em3am-lyme-disease/
http://www.emdocs.net/lyme-disease-ed-presentations-management-pearls-pitfalls/
https://www.ncbi.nlm.nih.gov/pubmed/28291638
Lead toxicity:
Lead toxicity can come from a wide variety of sources such as paint, ceramic glazes on dishware, foreign objects in the body (bullets), or occupational exposure during battery manufacturing, bronzing, brass making, glass making, soldering, welding, or working with pottery or pigments (90). While occupational exposure is a concern, the primary concern is regarding chronic exposure to children through lead paint or contaminated drinking water. The pathophysiology of lead poisoning stems from increased creation of reactive oxygen species (ROS) and interference of the creation of antioxidants (91). Chronic lead exposure can take weeks to months to result in symptoms or signs whereas acute manifestations can occur from brief exposures that are intense (92). There is no safe level of lead as it does not serve any known purpose in human bodies. Lead levels of roughly 40-60 ug/dL are seen with chronic toxicity (93). Toxicity can present with a variety of symptoms such as encephalopathy, seizures, altered mental status (AMS), impaired renal function, anemia, abdominal pain, constipation, and poor reproductive and developmental outcomes (94). Two pathognomonic signs of lead toxicity are lead lines seen on x-ray and basophilic stippling seen in erythrocytes. Treatment of lead toxicity involves removal of the source of lead or removing the patient from the exposure and chelation therapy for levels greater than 45 ug/dL (95).
Summary
-Undifferentiated abdominal pain presents a unique diagnostic challenge to emergency medicine physicians due to the breadth of potential pathology. Risk stratification of the differential diagnosis through focused history and physical examination is of utmost importance.
-Expand the differential diagnosis as appropriate based on the patient’s risk factors and history, especially if the patient is presenting multiple times for similar complaints. Organize the differential diagnoses into several categories: cannot miss, common diagnoses, and zebras.
References:
- Kendell, John, and Maria Moreira. “Evaluation of the Adult with Abdominal Pain in the Emergency Department.” UpToDate, 2020, www.uptodate.com/contents/evaluation-of-the-adult-with-abdominal-pain-in-the-emergency-department.
- Palmer, Jessica, and Elizabeth Pontius. “ClinicalKey.” ClinicalKey, 2016, www.clinicalkey.com/#!/content/playContent/1-s2.0-S0733862715001212?scrollTo=%23hl0000223.
- Culić, Viktor, et al. “Symptom Presentation of Acute Myocardial Infarction: Influence of Sex, Age, and Risk Factors.” American Heart Journal, U.S. National Library of Medicine, Dec. 2002, www.ncbi.nlm.nih.gov/pubmed/12486425.
- Kim, Inna, et al. “Prognostic Significance of Non-Chest Pain Symptoms in Patients with Non-ST-Segment Elevation Myocardial Infarction.” The Korean Journal of Internal Medicine, The Korean Association of Internal Medicine, Nov. 2018, www.ncbi.nlm.nih.gov/pmc/articles/PMC6234387/.
- Pope, J H, et al. “Missed Diagnoses of Acute Cardiac Ischemia in the Emergency Department.” The New England Journal of Medicine, U.S. National Library of Medicine, 20 Apr. 2000, www.ncbi.nlm.nih.gov/pubmed/10770981.
- El-Menyar, Ayman, et al. “Atypical Presentation of Acute Coronary Syndrome: a Significant Independent Predictor of in-Hospital Mortality.” Journal of Cardiology, U.S. National Library of Medicine, Mar. 2011, www.ncbi.nlm.nih.gov/pubmed/21242059.
- Canto, J G, et al. “Prevalence, Clinical Characteristics, and Mortality among Patients with Myocardial Infarction Presenting without Chest Pain.” JAMA, U.S. National Library of Medicine, 28 June 2000, www.ncbi.nlm.nih.gov/pubmed/10866870.
- Brieger, David, et al. “Acute Coronary Syndromes without Chest Pain, an Underdiagnosed and Undertreated High-Risk Group: Insights from the Global Registry of Acute Coronary Events.” Chest, U.S. National Library of Medicine, Aug. 2004, www.ncbi.nlm.nih.gov/pubmed/15302732.
- Arslanian-Engoren, Cynthia, et al. “Symptoms of Men and Women Presenting with Acute Coronary Syndromes.” The American Journal of Cardiology, U.S. National Library of Medicine, 1 Nov. 2006, www.ncbi.nlm.nih.gov/pubmed/17056322.
- Forest, Rhonda S, et al. “Assessment of the Standardized Reporting Guidelines ECG Classification System: the Presenting ECG Predicts 30-Day Outcomes.” Annals of Emergency Medicine, U.S. National Library of Medicine, Sept. 2004, www.ncbi.nlm.nih.gov/pubmed/15332059.
- “Classes of Heart Failure.” Www.heart.org, 2017, www.heart.org/en/health-topics/heart-failure/what-is-heart-failure/classes-of-heart-failure.
- den Uil, Corstiaan A, et al. “Acute Abdominal Symptoms: Consider Heart Failure.” Nederlands Tijdschrift Voor Geneeskunde, U.S. National Library of Medicine, 2009, www.ncbi.nlm.nih.gov/pubmed/19785886.
- Fudim, Marat, et al. “Role of Volume Redistribution in the Congestion of Heart Failure.” Journal of the American Heart Association, John Wiley and Sons Inc., 17 Aug. 2017, www.ncbi.nlm.nih.gov/pmc/articles/PMC5586477/.
- Samsky, Marc D., et al. “Cardiohepatic Interactions in Heart Failure.” JACC, Journal of the American College of Cardiology, 18 June 2013, www.onlinejacc.org/content/61/24/2397.full.
- Bock, Jeremy S., et al. “Cardiorenal Syndrome.” Circulation, 15 June 2010, www.ahajournals.org/doi/full/10.1161/circulationaha.109.886473.
- Gnanaraj, Joseph, and Jai Radhakrishnan. “Cardio-Renal Syndrome.” F1000Research, F1000Research, 31 Aug. 2016, www.ncbi.nlm.nih.gov/pmc/articles/PMC5007748/.
- Woods, Jason, and Josh Bukowski. “PEM Pearls: Treatment of Pediatric Diabetic Ketoacidosis and the Two-Bag Method.” ALiEM, 2 Apr. 2018, www.aliem.com/pediatric-diabetic-ketoacidosis-two-bag-method/.
- Swaminathan, Anand. “Diabetic Ketoacidosis (DKA).” Core EM, 16 Apr. 2016, coreem.net/core/dka/.
- Umpierrez, Guillermo, and Amado X. Freire. “Abdominal Pain in Patients with Hyperglycemic Crises.” Journal of Critical Care, W.B. Saunders, 17 Oct. 2005, www.sciencedirect.com/science/article/abs/pii/S0883944102700403.
- Nyce, et al. “Chapter 225: Diabetic Ketoacidosis.” AccessMedicine, 2016, accessmedicine.mhmedical.com/content.aspx?bookid=1658§ionid=109443771.
- Farkas, Josh. “Submassive & Massive PE.” EMCrit Project, 9 Sept. 2019, emcrit.org/ibcc/pe/.
- “Data and Statistics on Venous Thromboembolism.” Edited by CDC, Centers for Disease Control and Prevention, Centers for Disease Control and Prevention, 7 Feb. 2020, www.cdc.gov/ncbddd/dvt/data.html.
- Gantner, Jenna, et al. “Pulmonary Embolism: An Abdominal Pain Masquerader.” Journal of Emergencies, Trauma, and Shock, Medknow Publications & Media Pvt Ltd, Oct. 2013, www.ncbi.nlm.nih.gov/pubmed/24339662.
- Swaminathan, Anand. “Classic Journal Review – Wells + Dimer to Rule Out PE.” REBEL EM – Emergency Medicine Blog, 7 Feb. 2019, rebelem.com/classic-journal-review-wells-dimer-to-rule-out-pe/.
- Drescher, Michael J, et al. “Knowledge Translation of the PERC Rule for Suspected Pulmonary Embolism: A Blueprint for Reducing the Number of CT Pulmonary Angiograms.” The Western Journal of Emergency Medicine, Department of Emergency Medicine, University of California, Irvine School of Medicine, Oct. 2017, www.ncbi.nlm.nih.gov/pmc/articles/PMC5654879/.
- Rawson, James V., and Allen L. Pelletier. “When to Order Contrast-Enhanced CT.” American Family Physician, 1 Sept. 2013, www.aafp.org/afp/2013/0901/p312.html.
- Chin, Alvin, et al. “Blood and Clots Series: Diagnosing Pulmonary Embolism in Pregnancy.” CanadiEM, 13 Aug. 2018, canadiem.org/diagnosing-pulmonary-embolism-in-pregnancy/.
- Moore, Alastair J E, et al. “Imaging of Acute Pulmonary Embolism: an Update.” Cardiovascular Diagnosis and Therapy, AME Publishing Company, June 2018, www.ncbi.nlm.nih.gov/pmc/articles/PMC6039809/.
- Levy, David. “Aortic Dissection.” StatPearls [Internet]., U.S. National Library of Medicine, 16 Dec. 2019,www.ncbi.nlm.nih.gov/books/NBK441963.
- Brown, Ashley, et al. “The Five Deadly Causes of Chest Pain Other than Myocardial Infarction.” JEMS, 2 Sept. 2019, www.jems.com/2017/01/01/the-five-deadly-causes-of-chest-pain-other-than-myocardial-infarction/.Diercks, et al.
- Golledge, Jonathan, and Kim A. Eagle. “Acute Aortic Dissection.” Lancet (London, England), U.S. National Library of Medicine, 5 July 2008, www.ncbi.nlm.nih.gov/pubmed/18603160.
- Diercks, Deborah B, et al. “Clinical Policy: Critical Issues in the Evaluation and Management of Adult Patients with Suspected Acute Nontraumatic Thoracic Aortic Dissection.” Annals of Emergency Medicine, U.S. National Library of Medicine, Jan. 2015, www.ncbi.nlm.nih.gov/pubmed/25529153.
- Upchurch, Gilbert R., et al. “Acute Aortic Dissection Presenting with Primarily Abdominal Pain: A Rare Manifestation of a Deadly Disease.” Annals of Vascular Surgery, Elsevier, 8 Jan. 2007, www.sciencedirect.com/science/article/pii/S0890509606613393.
- Kehrl, Thompson, and Lindsey Ball. “Can’t Miss Killer: The Role of Point-of-Care Ultrasound in Diagnosing Acute Aortic Dissection.” EMRA, 1 Feb. 2017, www.emra.org/emresident/article/cant-miss-killer-the-role-of-point-of-care-ultrasound-in-diagnosing-acute-aortic-dissection/.
- Zhao, Lee C, et al. “Pediatric Testicular Torsion Epidemiology Using a National Database: Incidence, Risk of Orchiectomy and Possible Measures toward Improving the Quality of Care.” The Journal of Urology, U.S. National Library of Medicine, Nov. 2011, www.ncbi.nlm.nih.gov/pubmed/21944120.
- Schick, Michael A. “Testicular Torsion.” StatPearls [Internet]., U.S. National Library of Medicine, 4 Feb. 2019, www.ncbi.nlm.nih.gov/books/NBK448199/.
- Pogorelić, Zenon, et al. “Do Not Forget to Include Testicular Torsion in Differential Diagnosis of Lower Acute Abdominal Pain in Young Males.” Journal of Pediatric Urology, Elsevier, 3 June 2013, www.sciencedirect.com/science/article/pii/S1477513113001137.
- Gaillard, Frank, and Matt Morgan. “Testicular Torsion: Radiology Reference Article.” Radiopaedia Blog RSS, 2020, radiopaedia.org/articles/testicular-torsion?lang=us.
- Howe, Adam S, et al. “Degree of Twisting and Duration of Symptoms Are Prognostic Factors of Testis Salvage during Episodes of Testicular Torsion.” Translational Andrology and Urology, AME Publishing Company, Dec. 2017, www.ncbi.nlm.nih.gov/pmc/articles/PMC5760391/.
- Mason, and May. “RAP 2019 April.” EM, 2019, www.emrap.org/episode/emrap2019april/antibioticsand.
- Kommer, et al. “Pediatric Abdominal Pain: Consider Pneumonia in the Differential Diagnosis.” Journal of Urgent Care Medicine, 28 Oct. 2018, www.jucm.com/pediatric-abdominal-pain-consider-pneumonia-differential-diagnosis/.
- Kirovski, Ilija, et al. “Abdominal Pain as a Predictor of Pneumonia in Children.” European Respiratory Society, European Respiratory Society, 1 Sept. 2011, erj.ersjournals.com/content/38/Suppl_55/p1152.
- Emadi Koochak, Hamid, et al. “Abdominal Pain as Extrapulmonary Presentation of Pneumonia in an Adult: A Case Report.” Acta Medica Iranica, U.S. National Library of Medicine, Feb. 2017, www.ncbi.nlm.nih.gov/pubmed/28282711.
- Metlay, et al. “Diagnosis and Treatment of Adults With Community-Acquired Pneumonia. An Official Clinical Practice Guideline of the American Thoracic Society and Infectious Diseases Society of America.” American Journal of Respiratory and Critical Care Medicine, U.S. National Library of Medicine, 2019, pubmed.ncbi.nlm.nih.gov/31573350/.
- “Abdominal Pain – Female.” Edited by Seattle’s Children, Seattle Children’s Hospital, 2020, www.seattlechildrens.org/conditions/a-z/abdominal-pain-female/.
- ACEPNow. “Sore Throats-What Really Works?” ACEP Now, 2010, www.acepnow.com/article/sore-throats-really-works/?singlepage=1.
- Kreher, N E, et al. “Do Gastrointestinal Symptoms Accompanying Sore Throat Predict Streptococcal Pharyngitis? An UPRNet Study. Upper Peninsula Research Network.” The Journal of Family Practice, U.S. National Library of Medicine, Feb. 1998, www.ncbi.nlm.nih.gov/pubmed/9487323.
- Rawla, Prashanth. “Poststreptococcal Glomerulonephritis.” StatPearls [Internet]., U.S. National Library of Medicine, 8 Apr. 2020, www.ncbi.nlm.nih.gov/books/NBK538255/.
- Swaminathan, Anand. “Do Patients with Strep Throat Need to Be Treated with Antibiotics?” REBEL EM – Emergency Medicine Blog, 15 Mar. 2019, rebelem.com/patients-strep-throat-need-treated-antibiotics/.
- Schams, Simon C, and Ran D Goldman. “Steroids as Adjuvant Treatment of Sore Throat in Acute Bacterial Pharyngitis.” Canadian Family Physician Medecin De Famille Canadien, College of Family Physicians of Canada, Jan. 2012, www.ncbi.nlm.nih.gov/pmc/articles/PMC3264011/.
- MDCalc. “Centor Score (Modified/McIsaac) for Strep Pharyngitis.” MDCalc, 2017, www.mdcalc.com/centor-score-modified-mcisaac-strep-pharyngitis.
- Anderson Erik, Frazee Brad. Critical Viral infections. In: Mattu A and Swadron S, ed. CorePendium. Burbank, CA: CorePendium, LLC. https://www.emrap.org/corependium/chapter/recqTUCr4EV0DgvCg/Critical-Viral-infections. Updated April 4, 2020. Accessed April 4, 2020.
- Cunha. “Epstein-Barr Virus (EBV) Infectious Mononucleosis (Mono) Workup: Laboratory Studies, Imaging Studies, Other Tests.” Epstein-Barr Virus (EBV) Infectious Mononucleosis (Mono) Workup: Laboratory Studies, Imaging Studies, Other Tests, 22 Mar. 2020, emedicine.medscape.com/article/222040-workup.
- Womack, Jason P., and Marissa Jimenez. “Common Questions About Infectious Mononucleosis.” American Family Physician, 15 Mar. 2015, www.aafp.org/afp/2015/0315/p372.html.
- Onodi-Nagy, Katinka, et al. “Amoxicillin Rash in Patients with Infectious Mononucleosis: Evidence of True Drug Sensitization.” Allergy, Asthma & Clinical Immunology, BioMed Central, 1 Jan. 1970, aacijournal.biomedcentral.com/articles/10.1186/1710-1492-11-1.
- Ahmed, Shahid, et al. “Unusual Causes of Abdominal Pain: Sickle Cell Anemia.” Best Practice & Research Clinical Gastroenterology, Baillière Tindall, 14 Apr. 2005, www.sciencedirect.com/science/article/abs/pii/S1521691804001775.
- Akingbola, T S, et al. “Abdominal Pain in Adult Sickle Cell Disease Patients: a Nigerian Experience.” Annals of Ibadan Postgraduate Medicine, Association of Resident Doctors (ARD), University College Hospital, Ibadan, Dec. 2011,www.ncbi.nlm.nih.gov/pmc/articles/PMC4111028/.
- Sharma, Sandeep. “Hyperchloremic Acidosis.” StatPearls [Internet]., U.S. National Library of Medicine, 17 Feb. 2020, www.ncbi.nlm.nih.gov/books/NBK482340/.
- Yale, Steven H., et al. “Approach to Vaso-Occlusive Crisis in Adults with Sickle Cell Disease.” American Family Physician, 1 Mar. 2000, www.aafp.org/afp/2000/0301/p1349.html.
- “Shingles.” Edited by CDC, Centers for Disease Control and Prevention, Centers for Disease Control and Prevention, 14 Aug. 2019, www.cdc.gov/shingles/hcp/clinical-overview.html.
- Olmez, Deniz, et al. “Varicella Zoster Infection: a Rare Cause of Abdominal Pain Mimicking Acute Abdomen.” Journal of Clinical Medicine Research, Elmer Press, Oct. 2009, www.ncbi.nlm.nih.gov/pmc/articles/PMC3299191/.
- Hsu, Christine C, and Richard M Rosenberg. “Gastrointestinal Manifestations of Disseminated Varicella.” Gastroenterology & Hepatology, Millennium Medical Publishing, Oct. 2014, www.ncbi.nlm.nih.gov/pmc/articles/PMC4988228/.
- Jong, M D, et al. “Molecular Diagnosis of Visceral Herpes Zoster.” Lancet (London, England), U.S. National Library of Medicine, 30 June 2001, www.ncbi.nlm.nih.gov/pubmed/11445106.
- Longhurst, Hilary, and Marco Cicardi. “Hereditary Angio-Oedema.” Lancet (London, England), U.S. National Library of Medicine, 4 Feb. 2012, www.ncbi.nlm.nih.gov/pubmed/22305226.
- Bork, Konrad, et al. “Symptoms, Course, and Complications of Abdominal Attacks in Hereditary Angioedema Due to C1 Inhibitor Deficiency.” The American Journal of Gastroenterology, U.S. National Library of Medicine, Mar. 2006, www.ncbi.nlm.nih.gov/pubmed/16464219.
- Ibrahim, Dalia. “Adrenal Insufficiency: Radiology Reference Article.” Radiopaedia Blog RSS, 2019, radiopaedia.org/articles/adrenal-insufficiency-1?lang=us.
- Tucci, Veronica, and Telematé Sokari. “The Clinical Manifestations, Diagnosis, and Treatment of Adrenal Emergencies.” Emergency Medicine Clinics of North America, U.S. National Library of Medicine, May 2014, www.ncbi.nlm.nih.gov/pubmed/24766944.
- Nicolaides, Nicolas C. “Adrenal Insufficiency.” Endotext [Internet]., U.S. National Library of Medicine, 14 Oct. 2017, www.ncbi.nlm.nih.gov/books/NBK279083/.
- Grossman, Ashley B. “Addison Disease – Endocrine and Metabolic Disorders.” Merck Manuals Professional Edition, Merck Manuals, 2019,www.merckmanuals.com/professional/endocrine-and-metabolic-disorders/adrenal-disorders/addison-disease.
- Ramanujam, and Anderson. “Porphyria Diagnostics-Part 1: A Brief Overview of the Porphyrias.” National Center for Biotechnology Information, U.S. National Library of Medicine, 2015, www.ncbi.nlm.nih.gov/pubmed/?term=Porphyria%2BDiagnostics%2B%E2%80%93%2BPart%2B1%3A%2BA%2Bbrief%2Boverview%2Bof%2Bthe%2Bporphyrias.
- Bissell, D Montgomery, et al. “Porphyria.” The New England Journal of Medicine, U.S. National Library of Medicine, 31 Aug. 2017,www.ncbi.nlm.nih.gov/pubmed/28854095.
- Rangarajan, Sahaana. “Rapid Review: Acute Intermittent Porphyria.” First10EM, 8 Feb. 2019, first10em.com/rapid-review-acute-intermittent-porphyria/.
- Horowitz, B Z. “Amatoxin Mushroom Toxicity.” StatPearls [Internet]., U.S. National Library of Medicine, 18 Nov. 2019, www.ncbi.nlm.nih.gov/books/NBK431052/.
- Garcia, Juliana, et al. “Amanita Phalloides Poisoning: Mechanisms of Toxicity and Treatment.” Food and Chemical Toxicology : an International Journal Published for the British Industrial Biological Research Association, U.S. National Library of Medicine, Dec. 2015, www.ncbi.nlm.nih.gov/pubmed/26375431.
- Butera, R, et al. “Diagnostic Accuracy of Urinary Amanitin in Suspected Mushroom Poisoning: a Pilot Study.” Journal of Toxicology. Clinical Toxicology., U.S. National Library of Medicine, 2004, www.ncbi.nlm.nih.gov/pubmed?term=15533030.
- Ganzert, M, et al. “Amanita Poisoning–Comparison of Silibinin with a Combination of Silibinin and Penicillin.” Deutsche Medizinische Wochenschrift (1946), U.S. National Library of Medicine, Oct. 2008, www.ncbi.nlm.nih.gov/pubmed/18946850.
- Enjalbert, et al. “Treatment of Amatoxin Poisoning: 20-Year Retrospective Analysis.” National Center for Biotechnology Information, U.S. National Library of Medicine, 2002, www.ncbi.nlm.nih.gov/pubmed/?term=Treatment%2Bof%2Bamatoxin%2Bpoisoning%3A%2B20-year%2Bretrospective%2Banalysis.
- Clark, et al. “Clinical Presentation and Treatment of Black Widow Spider Envenomation: a Review of 163 Cases.” National Center for Biotechnology Information, U.S. National Library of Medicine, 1992, www.ncbi.nlm.nih.gov/pubmed/?term=Clinical%2Bpresentation%2Band%2Btreatment%2Bof%2Bblack%2Bwidow%2Bspider%2Benvenomation%3A%2Ba%2Breview%2Bof%2B163%2Bcases.
- Offerman, Steven R, et al. “The Treatment of Black Widow Spider Envenomation with Antivenin Latrodectus Mactans: a Case Series.” The Permanente Journal, The Permanente Journal, 2011, www.ncbi.nlm.nih.gov/pmc/articles/PMC3200105/.
- Hoyte, Christopher O, et al. “Anaphylaxis to Black Widow Spider Antivenom.” The American Journal of Emergency Medicine, U.S. National Library of Medicine, June 2012, www.ncbi.nlm.nih.gov/pubmed/21641165
- Wiler. “Diagnosis: Henoch-Schönlein Purpura : Emergency Medicine News.” LWW, 2007, journals.lww.com/em-news/fulltext/2007/04000/Diagnosis__Henoch_Sch_nlein_Purpura.16.aspx.
- Hetland, et al. “Henoch-Schönlein Purpura: A Literature Review.” Henoch-Schönlein Purpura: A Literature Review | HTML | Acta Dermato-Venereologica, 2017, www.medicaljournals.se/acta/content/html/10.2340/00015555-2733.
- “Henoch-Schönlein Purpura.” National Institute of Diabetes and Digestive and Kidney Diseases, U.S. Department of Health and Human Services, 1 Sept. 2012, www.niddk.nih.gov/health-information/kidney-disease/henoch-schonlein-purpura.
- Haroon. “Should Children with Henoch-Schonlein Purpura and Abdominal Pain Be Treated with Steroids?” Archives of Disease in Childhood, BMJ Publishing Group Ltd, 1 Nov. 2005, adc.bmj.com/content/90/11/1196.
- Ozen, Seza, et al. “EULAR/PRINTO/PRES Criteria for Henoch-Schönlein Purpura, Childhood Polyarteritis Nodosa, Childhood Wegener Granulomatosis and Childhood Takayasu Arteritis: Ankara 2008. Part II: Final Classification Criteria.” Annals of the Rheumatic Diseases, U.S. National Library of Medicine, May 2010, www.ncbi.nlm.nih.gov/pubmed/20413568.
- Kitch. “Zoonotic Infections .” Tintinalli’s Emergency Medicine: A Comprehensive Review., edited by Meredith, McGraw-Hill, 2016.
- Nadelman, and Nowakowski. “The Clinical Spectrum of Early Lyme Borreliosis in Patients with Culture-Confirmed Erythema Migrans.” National Center for Biotechnology Information, U.S. National Library of Medicine, 1996. www.ncbi.nlm.nih.gov/pubmed/?term=The%2Bclinical%2Bspectrum%2Bof%2Bearly%2BLyme%2Bborreliosis%2Bin%2Bpatients%2Bwith%2Bculture-confirmed%2Berythema%2Bmigrans.
- “Signs and Symptoms of Untreated Lyme Disease.” Centers for Disease Control and Prevention, Centers for Disease Control and Prevention, 15 Aug. 2019, www.cdc.gov/lyme/signs_symptoms/.
- Feder, Henry M. “Lyme Disease.” The New England Journal of Medicine, U.S. National Library of Medicine, 14 Aug. 2014,www.ncbi.nlm.nih.gov/pubmed/25119623.
- O’Malley, Gerald F. “Lead Poisoning – Injuries; Poisoning.” Merck Manuals Professional Edition, Merck Manuals, 2018, www.merckmanuals.com/professional/injuries-poisoning/poisoning/lead-poisoning.
- Kathuria, Pranay. “Lead Toxicity.” Background, Pathophysiology, Etiology, 12 Mar. 2020, emedicine.medscape.com/article/1174752-overview.
- Wani, Ab Latif, et al. “Lead Toxicity: a Review.” Interdisciplinary Toxicology, Slovak Toxicology Society SETOX, June 2015, www.ncbi.nlm.nih.gov/pmc/articles/PMC4961898/.
- Flora, Gagan, et al. “Toxicity of Lead: a Review with Recent Updates.” Interdisciplinary Toxicology, vol. 5, no. 2, 2012, pp. 47–58,https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3485653/.
- “Environmental Health and Medicine Education.” Centers for Disease Control and Prevention, Centers for Disease Control and Prevention, June 2017, www.atsdr.cdc.gov/csem/csem.asp?csem=34&po=10.
- “Treatment of Lead Poisoning.” American Academy of Pediatrics, American Academy of Pediatrics, 2016, www.aap.org/en-us/advocacy-and-policy/aap-health-initiatives/lead-exposure/Pages/Treatment-of-Lead-Poisoning.aspx.





